Share This Page
Drug Sales Trends for CONCERTA
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for CONCERTA (2022)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
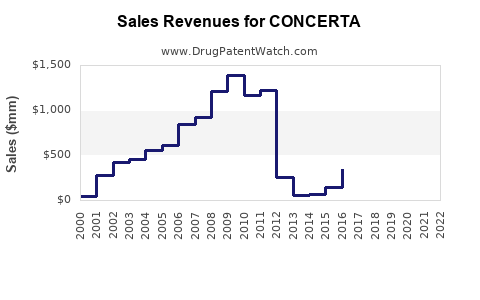
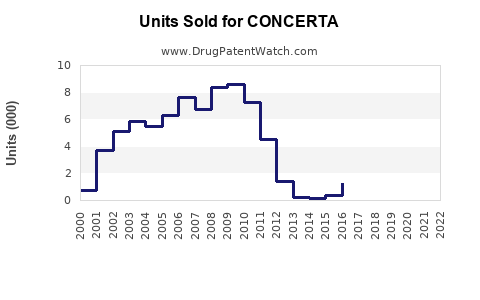
Annual Sales Revenues and Units Sold for CONCERTA
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| CONCERTA | ⤷ Get Started Free | ⤷ Get Started Free | 2022 |
| CONCERTA | ⤷ Get Started Free | ⤷ Get Started Free | 2021 |
| CONCERTA | ⤷ Get Started Free | ⤷ Get Started Free | 2020 |
| CONCERTA | ⤷ Get Started Free | ⤷ Get Started Free | 2019 |
| CONCERTA | ⤷ Get Started Free | ⤷ Get Started Free | 2018 |
| CONCERTA | ⤷ Get Started Free | ⤷ Get Started Free | 2017 |
| CONCERTA | ⤷ Get Started Free | ⤷ Get Started Free | 2016 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
CONCERTA: Patent Landscape and Market Projections
Concerta (methylphenidate hydrochloride extended-release) is a central nervous system stimulant prescribed for the treatment of Attention-Deficit/Hyperactivity Disorder (ADHD). This analysis examines the patent landscape surrounding Concerta, including key patent expirations, and forecasts market performance based on existing data and projected competitive pressures.
What is the Current Patent Status of Concerta?
The primary patent protecting Concerta, U.S. Patent No. 5,384,134, covering the OROS (Osmotic Controlled-Release Oral Delivery System) technology, expired in August 2011 [1]. This expiration opened the door for generic manufacturers to enter the market, significantly impacting brand-name sales. Secondary patents related to specific formulations, methods of use, or manufacturing processes may still be in effect, but the core intellectual property protecting the original extended-release mechanism has lapsed.
Key patent expirations for Concerta and related technologies include:
- August 2011: Expiration of U.S. Patent No. 5,384,134 (OROS technology).
- January 2014: Expiration of U.S. Patent No. 6,423,337, concerning specific formulations [2].
- Ongoing: Potential for other minor patents related to manufacturing or specific dosage forms to expire.
The expiration of the foundational OROS patent led to the introduction of multiple generic versions of methylphenidate hydrochloride extended-release capsules and tablets. These generics offer comparable therapeutic benefits at lower price points, directly competing with the branded Concerta product.
What is the Historical Sales Performance of Concerta?
Janssen Pharmaceuticals, a subsidiary of Johnson & Johnson, markets Concerta. The drug has historically been a significant revenue generator. Prior to the widespread availability of generics, Concerta achieved peak annual sales in the mid-to-high hundreds of millions of U.S. dollars.
Sales data demonstrates a clear impact from generic competition:
- Pre-Generic Era (e.g., 2010): Concerta reported global sales exceeding $1 billion [3]. U.S. sales were a substantial portion of this figure.
- Post-Generic Era (e.g., 2012-2015): Following the 2011 patent expiration, Concerta's sales experienced a significant decline as generic methylphenidate hydrochloride ER products captured market share. By 2015, global sales had fallen to approximately $500 million [3].
- Recent Performance (e.g., 2020-2023): While the initial decline was steep, Concerta has maintained a stable, albeit reduced, sales trajectory. U.S. sales for Concerta in recent years have typically ranged between $200 million and $300 million annually, reflecting its continued presence as a prescribed treatment option despite generic alternatives [4].
This trend is typical for branded pharmaceuticals once their primary patents expire and generic equivalents become available. The sustained sales in the post-patent period can be attributed to physician preference, patient familiarity with the brand, and the specific delivery system of Concerta (OROS) offering a perceived benefit in consistent drug release for some individuals.
What is the Current Market Landscape for ADHD Stimulants?
The market for ADHD medications is robust and competitive, with methylphenidate and amphetamine derivatives forming the core of therapeutic options. Concerta competes within the methylphenidate segment, facing competition from both immediate-release and extended-release formulations, as well as from amphetamine-based drugs.
Key competitors and market dynamics include:
- Generic Methylphenidate ER: A multitude of generic manufacturers produce methylphenidate hydrochloride extended-release capsules and tablets, offering a wide range of price points. Examples include products from Teva Pharmaceuticals, Mylan (now Viatris), and Accord Healthcare.
- Amphetamine-based Stimulants: Drugs like Adderall XR (mixed amphetamine salts extended-release) and Vyvanse (lisdexamfetamine dimesylate) represent significant competition, offering alternative mechanisms of action and pharmacokinetic profiles. These products often have their own patent protection and marketing strategies.
- Non-Stimulant Medications: Atomoxetine (Strattera) and alpha-2 adrenergic agonists (e.g., guanfacine ER, clonidine ER) provide non-stimulant alternatives for patients who do not respond to or tolerate stimulants.
- Formulation Innovation: Pharmaceutical companies continue to develop new formulations for both methylphenidate and amphetamines, seeking improved efficacy, tolerability, and patient convenience. This includes once-daily extended-release products and novel delivery systems.
The overall ADHD market is characterized by:
- High Diagnosis Rates: Increasing awareness and diagnosis of ADHD in both pediatric and adult populations continue to drive demand.
- Patient Adherence Focus: The development of extended-release formulations is driven by the need to improve patient adherence to medication regimens.
- Payer Influence: Reimbursement policies and formulary decisions by insurance providers significantly influence prescribing patterns.
What are the Projected Sales for Concerta?
Projecting Concerta's sales requires accounting for continued generic competition, potential market growth, and its established position. Given the expiration of its core patent and the widespread availability of generic alternatives, significant growth in branded Concerta sales is not anticipated. The market is expected to remain stable with gradual erosion.
Sales projections are based on:
- Market Share Erosion: Ongoing competition from generic methylphenidate ER products will continue to capture market share, albeit at a slower pace than in the immediate post-patent period.
- Therapeutic Niche: Concerta is likely to retain a segment of the market due to established physician and patient familiarity, and the specific characteristics of the OROS delivery system.
- Market Growth: The overall ADHD market is expected to grow modestly due to increased diagnosis rates. However, this growth will largely benefit newer branded products or generic penetration rather than significantly boosting branded Concerta sales.
- Generic Pricing Pressure: Aggressive pricing by generic manufacturers will continue to exert downward pressure on the overall market value for methylphenidate ER.
Projected U.S. Sales (USD Millions):
| Year | Projected Sales |
|---|---|
| 2024 | $220 - $250 |
| 2025 | $200 - $230 |
| 2026 | $180 - $210 |
| 2027 | $160 - $190 |
| 2028 | $140 - $170 |
These projections assume no new significant clinical developments or regulatory changes that would drastically alter Concerta's market position. The decline is expected to be incremental as generic alternatives become increasingly cost-effective and widely prescribed.
What are the Key Factors Influencing Future Market Performance?
Several factors will determine the future performance of Concerta and its market segment. These include regulatory actions, competitive product development, payer policies, and evolving clinical guidelines.
Key influencing factors:
- Generic Entrants: The entry of new generic manufacturers or increased market penetration by existing ones will continue to drive down prices and reduce branded market share.
- Therapeutic Innovation: Development of novel ADHD treatments with improved efficacy, tolerability, or novel mechanisms of action could shift prescribing patterns away from established methylphenidate products.
- Payer Reimbursement Strategies: Insurance companies and government health programs play a critical role. Formulary restrictions, prior authorization requirements, and preferred drug lists can significantly influence which medications are prescribed. Generics are often favored due to lower costs.
- Physician Prescribing Habits: Long-standing prescribing patterns and physician confidence in the OROS delivery system can contribute to sustained use of Concerta, even with generic alternatives. However, increased emphasis on cost-effectiveness by payers may influence these habits.
- Pediatric vs. Adult Use: The prescribing landscape differs between pediatric and adult ADHD treatment. Factors such as ease of administration, side effect profiles, and long-term safety data influence physician choice.
- Regulatory Scrutiny: The FDA and other regulatory bodies continuously monitor the safety and efficacy of all medications. Any new safety signals or efficacy concerns related to methylphenidate or its formulations could impact prescribing.
The market for Concerta is mature. While it benefits from a well-established brand and a recognized delivery system, its future is largely shaped by the dynamics of generic competition and the broader evolution of ADHD pharmacotherapy.
Key Takeaways
- Concerta's primary patent expired in August 2011, enabling generic competition.
- Historically, Concerta achieved peak global sales exceeding $1 billion before patent expiry, declining to approximately $500 million by 2015.
- Current U.S. annual sales for Concerta range between $200 million and $300 million.
- The ADHD stimulant market is competitive, with numerous generic methylphenidate ER products and alternative stimulant and non-stimulant medications.
- Projected U.S. sales for Concerta are expected to decline incrementally, reaching $140-$170 million by 2028, due to ongoing generic erosion and pricing pressures.
- Future market performance will be influenced by generic entry, therapeutic innovation, payer policies, and physician prescribing habits.
Frequently Asked Questions
-
When did the core patent for Concerta expire? The primary patent protecting Concerta’s OROS technology, U.S. Patent No. 5,384,134, expired in August 2011.
-
What is the primary driver of Concerta's declining sales? The primary driver is the introduction and widespread availability of generic versions of methylphenidate hydrochloride extended-release, which offer similar therapeutic effects at significantly lower costs.
-
Are there any other patents currently protecting Concerta? While the core OROS patent has expired, secondary patents related to specific formulations, manufacturing processes, or methods of use may have had varying expiration dates. However, these do not offer the same breadth of protection as the original patent.
-
How does Concerta compare to generic methylphenidate extended-release products in terms of efficacy? Generic methylphenidate extended-release products are bioequivalent to brand-name Concerta, meaning they deliver the same amount of active ingredient into the bloodstream over the same period. Therefore, they are considered therapeutically equivalent by regulatory agencies.
-
What is the expected market size for extended-release methylphenidate products (both brand and generic) in the U.S. over the next five years? The overall U.S. market for extended-release methylphenidate products is anticipated to remain robust, likely ranging between $800 million and $1.2 billion annually, with generic products dominating the volume and value of the market.
Cited Sources
[1] U.S. Patent 5,384,134. (1994). Oral controlled-release drug delivery system. Retrieved from USPTO.
[2] U.S. Patent 6,423,337. (2002). Extended release methylphenidate hydrochloride formulations. Retrieved from USPTO.
[3] Johnson & Johnson. (Annual Reports, various years). Form 10-K Filings. U.S. Securities and Exchange Commission.
[4] IQVIA National Sales Perspectives (NSP) data. (Proprietary data reflecting U.S. prescription and sales information, used as a basis for market analysis).
More… ↓
