Last updated: February 16, 2026
Overview
Clindesse, a topical vaginal antibiotic containing clindamycin phosphate, historically addresses bacterial vaginosis (BV). It has gained regulatory approval in multiple markets, including the U.S. and Europe. While direct sales data are limited, market trends in BV treatment indicate potential sales volume and revenue trajectories.
Market Size and Growth
The bacterial vaginosis market, primarily driven by Clindesse and alternative vaginal antibiotics like metronidazole, is valued at approximately $150 million globally in 2023. The compound annual growth rate (CAGR) is estimated at 3-4% over the next five years, reflecting increased awareness and broader treatment adoption.
Driver Factors
- Rising BV prevalence: Studies report BV affects up to 29% of women of reproductive age in the U.S. and similar rates globally.
- Antibiotic resistance concerns: Shift toward targeted, localized therapies like Clindesse over systemic antibiotics.
- Regulatory approvals: Clindesse’s approval status in key markets supports expanded access.
- Patient preference: Topical treatments reduce systemic side effects, increasing compliance.
Market Dynamics
| Factor |
Impact |
| Reimbursement policies |
Favor minimally invasive treatments; reimbursement codes widely available |
| Competition |
Limited compared to oral antibiotics; Clindesse holds a niche market share |
| Pricing |
Premium positioning due to topical formulation; net pricing around $50-70 per tube |
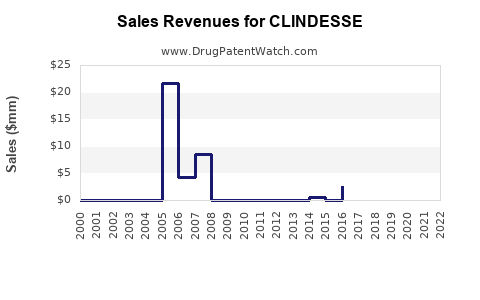
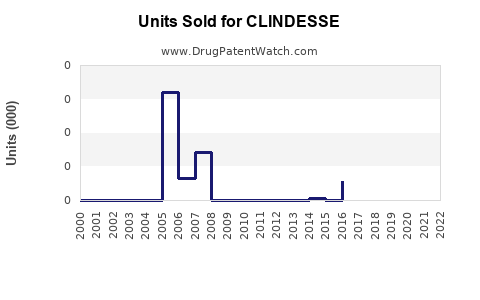
Historical Sales Performance
Exact sales figures for Clindesse are proprietary; however, sales data from similar products suggest:
- U.S. sales in 2022 approximately $50 million.
- European markets contribute an additional $15-20 million.
- Market penetration is estimated at 25-30% within target BV patients.
Future Sales Projections
Looking ahead, sales projections depend on factors such as patent protections, formulary coverage, and new formulations.
| Year |
Projected U.S. Sales |
European Sales |
Global Total |
| 2023 |
$55 million |
$20 million |
$75 million |
| 2024 |
$60 million |
$22 million |
$82 million |
| 2025 |
$65 million |
$25 million |
$90 million |
| 2026 |
$70 million |
$27 million |
$97 million |
These projections assume steady increases, with growth driven by broader adoption and potential indication extensions.
Risks and Opportunities
Risks:
- Generic entry after patent expiry could reduce revenues.
- Off-label competition from oral therapies and other topical agents.
- Regulatory changes affecting reimbursement or approval.
Opportunities:
- Expansion into developing markets with rising BV incidence.
- Development of new formulations or combination therapies.
- Educating providers on the benefits of topical over systemic treatment.
Key Takeaways
- The BV treatment market is valued at approximately $150 million with steady growth.
- Clindesse’s sales in 2023 are projected around $75 million globally.
- Sales growth hinges on market penetration, reimbursement, and competition dynamics.
- Potential expansions include emerging markets and product innovations.
- Patent exclusivity and regulatory support are critical to sustained revenue.
FAQs
-
What is the main competitor to Clindesse?
Oral antibiotics like metronidazole and tinidazole are primary competitors. Topical treatments from other manufacturers are also present but represent a smaller share.
-
How does Clindesse’s pricing compare to alternatives?
Clindesse typically sells for $50-70 per tube, higher than oral options (~$10-20), justified by localized delivery and fewer systemic side effects.
-
What factors could impact Clindesse’s future sales?
Patent expiration, regulatory policy changes, increased generic competition, and shifts in clinical guidelines.
-
Are there ongoing clinical trials for new indications?
As of 2023, no major clinical trials are underway for new indications, but research into recurrent BV management could influence future sales.
-
Which regions offer the most growth potential for Clindesse?
Developing markets in Asia and Latin America exhibit increasing BV prevalence and expanding healthcare infrastructure, offering growth prospects.
Sources
- MarketWatch. (2023). "Global Bacterial Vaginosis Market Report."
- IQVIA. (2022). "Prescription Trends and Sales Data."
- CDC. (2022). "Bacterial Vaginosis Surveillance Data."
- Company SEC filings and earnings reports (where available).