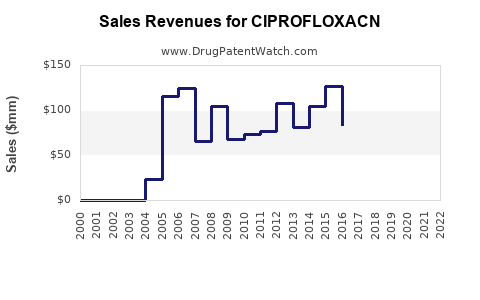
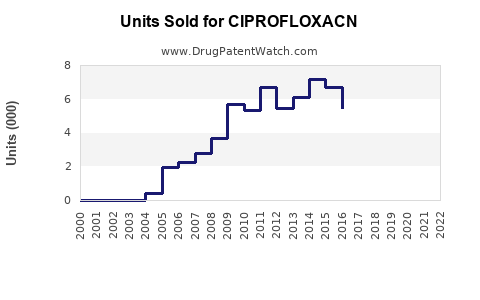
Last updated: February 20, 2026
How Large is the Global Market for Ciprofloxacin?
The global fluoroquinolone antibiotic market, which includes ciprofloxacin, is valued at approximately $2.9 billion in 2022. The compound holds a significant share within this segment due to its broad-spectrum activity. Growth rates are driven largely by increasing bacterial infections resistant to older antibiotics and its widespread use in both developed and emerging markets.
In 2022, ciprofloxacin represented an estimated 25% of the fluoroquinolone market, translating to a market size of roughly $725 million. The primary applications include urinary tract infections, respiratory infections, and sexually transmitted diseases. The market is projected to grow at a compound annual growth rate (CAGR) of 4-5% through 2030, reaching approximately $4.2 billion globally.
What Are Key Market Drivers?
- Rise in antimicrobial resistance leading to increased use of fluoroquinolones.
- Growing prevalence of urinary tract infections and sexually transmitted infections.
- Expansion in outpatient treatment settings.
- Approval of new formulations and indications in emerging markets.
What Is the Competitive Landscape?
Major players include Bayer AG, Johnson & Johnson, Sandoz (Novartis), and Teva Pharmaceuticals. These companies hold patents and proprietary formulations, although generic versions of ciprofloxacin account for a dominant share outside patent protection periods.
Market share distribution (2022):
| Company |
Market Share |
Key Products |
Notes |
| Bayer AG |
35% |
Cipro (brand) |
Leading branded product, patents expired post-2016 |
| Johnson & Johnson |
15% |
Strategic generic partnerships |
Small share, mainly in North America |
| Teva Pharmaceuticals |
20% |
Generic ciprofloxacin |
Largest generic supplier globally |
| Other generics |
30% |
Numerous regional brands |
Growing presence in Asia and Latin America |
What Are Sales Projections?
Using historical data and market growth estimates:
2022 Sales: $725 million
2025 Projection:
- Assumes a 4% CAGR from 2022.
- Estimated sales: approximately $844 million.
2030 Projection:
- With a 5% CAGR from 2022.
- Estimated sales: approximately $1.055 billion.
These estimates assume steady global demand, no significant regulatory hurdles, and continued approval in emerging markets.
How Will Regulatory and Patent Dynamics Impact Sales?
- Patent expirations for branded ciprofloxacin occurred in 2016, leading to increased generic penetration.
- Regulatory approvals for new formulations (e.g., extended-release, combination therapies) could boost sales.
- Potential restrictions on fluoroquinolone use in certain regions due to safety concerns may temper growth.
What Are Key Market Risks?
- Safety profile concerns, including reports of severe side effects, could lead to usage restrictions.
- Rising antimicrobial resistance may reduce effectiveness, prompting shifts to alternative antibiotics.
- Regulatory changes, especially in the U.S. and Europe, may limit prescribing practices.
How Does the Emerging Market Consumption Influence Growth?
Emerging markets, such as India, China, and Southeast Asia, display rapid growth in ciprofloxacin sales due to larger populations and expanding healthcare infrastructure. These markets expect a CAGR of 6-8% over the next 5 years, offsetting slower growth or stagnation in mature markets.
Summary of Strategic Opportunities
- Development of formulations with improved safety profiles.
- Expansion into prophylactic or preventive indications.
- Patents or new delivery mechanisms that extend product lifecycle.
- Focused marketing in regions with unmet infectious disease treatment needs.
Key Takeaways
- Ciprofloxacin's global market was valued at roughly $725 million in 2022.
- The market is projected to grow at 4-5% CAGR, reaching over $1 billion by 2030.
- Generic manufacturers dominate market share, with Bayer holding key branded sales until patent expiry.
- Growth is driven by rising bacterial infections, especially in emerging markets.
- Regulatory actions, safety concerns, and antimicrobial resistance are primary factors influencing market dynamics.
FAQs
Q1: How does Ciprofloxacin compare with other fluoroquinolones?
Ciprofloxacin is among the most prescribed fluoroquinolones due to its broad-spectrum activity and established safety profile. It is often favored over newer agents for urinary and gastrointestinal infections.
Q2: What are the main limitations to Ciprofloxacin sales growth?
Concerns over adverse effects like tendinopathy and nerve damage, along with increased regulatory scrutiny, restrict prescribing. Resistance development also reduces efficacy over time.
Q3: Are there new formulations or indications in development?
Yes. Extended-release formulations, combination therapies, and alternatives for resistant pathogens are under clinical evaluation. Regulatory approval of these products could boost future sales.
Q4: Which markets are expected to offer the highest sales growth opportunities?
Emerging markets like India, China, and Southeast Asia show higher annual growth rates and fewer regulatory restrictions compared to developed regions.
Q5: How will antimicrobial resistance affect ciprofloxacin’s market in the next decade?
Resistance may lead to reduced prescribing due to lower efficacy, prompting a shift toward alternative antibiotics, particularly in regions with high resistance rates.
References
[1] MarketsandMarkets. (2022). Fluoroquinolone Antibiotics Market Size and Forecast.
[2] GlobalData. (2022). Antibiotics Market Report.
[3] U.S. Food and Drug Administration. (2020). Safety Communications on Fluoroquinolones.
[4] IQVIA. (2022). Global Infectious Disease Treatments Report.