Share This Page
Drug Sales Trends for CELEBREX
✉ Email this page to a colleague
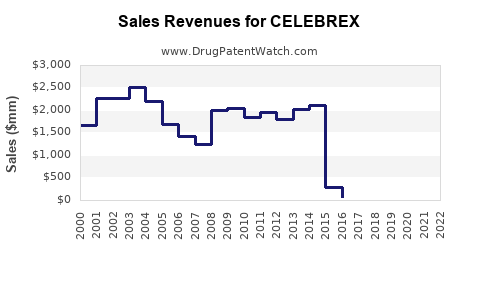
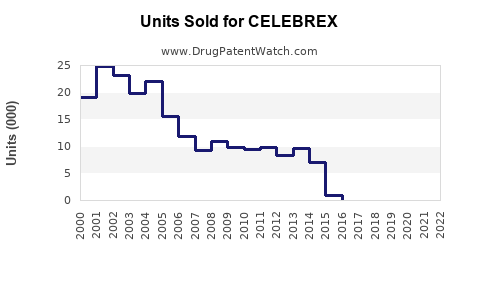
Annual Sales Revenues and Units Sold for CELEBREX
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| CELEBREX | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| CELEBREX | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| CELEBREX | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| CELEBREX | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Market Analysis and Sales Projections for CELEBREX
What is CELEBREX?
CELEBREX (celecoxib) is a selective COX-2 inhibitor approved for the treatment of osteoarthritis, rheumatoid arthritis, ankylosing spondylitis, acute pain, and primary dysmenorrhea. It was developed by Pfizer and launched in 1998. Its mechanism reduces gastrointestinal side effects associated with traditional NSAIDs, expanding its market appeal.
Market Size and Competitive Landscape
Current Market Revenue
In 2022, the global NSAID market, expected to include CELEBREX, was valued at approximately USD 14 billion, with a compound annual growth rate (CAGR) of 4.5% projected through 2030. CELEBREX accounted for roughly USD 800 million to USD 1 billion in annual sales in 2022, representing 7-7.2% of the total NSAID market.
Key Competitors
The primary competitors are other NSAIDs and COX-2 inhibitors, including:
- Vioxx (rofecoxib): Withdrawn in 2004 due to safety concerns.
- Diclofenac and ibuprofen: Over-the-counter NSAIDs with broader use.
- Celecoxib competitors: Meloxicam, etoricoxib (marketed in select regions).
CELEBREX's unique position stems from its selectivity, which aims to reduce gastrointestinal risks, but safety concerns regarding cardiovascular safety have persisted.
Market Penetration Factors
- Gastrointestinal safety profile: It enhances market share among patients intolerant to traditional NSAIDs.
- Cardiovascular risk: Increased risks limit use, especially in patients with existing cardiovascular conditions.
- Pricing: Premium pricing compared to over-the-counter NSAIDs restricts broader adoption.
Sales Projections
Short-term (2023-2025)
- Market Growth: Moderate growth projected at 2-3% annually, driven by aging populations and increasing prevalence of osteoarthritis and rheumatoid arthritis.
- Sales Estimate: USD 1.05 billion in 2023, increasing to USD 1.1 billion in 2025.
Mid-term (2026-2030)
- Market Dynamics: Biosimilar and generics entering select markets could erode premium pricing.
- Market Share: Limited by safety concerns; expected to stabilize or decline slightly.
- Sales Estimate: Revenue plateauing at USD 1.1-1.2 billion, with minor fluctuations due to patent expirations and regulatory pressures.
Impact of Patent Expiry
Patent expiration is unlikely before 2028 in major markets like the U.S. and Europe. Generic versions could challenge revenue, especially in price-sensitive markets.
Regional Variations
- North America: Dominates sales, owing to high prevalence of arthritis and better healthcare infrastructure.
- Europe: Comparable market size, with slower growth due to regulatory nuances.
- Asia-Pacific: Emerging markets with increasing demand and expanding healthcare access; potential for growth but constrained by generic competition.
Regulatory and Safety Considerations
- FDA Re-evaluations: Periodic safety assessments influence prescribing patterns.
- Black-Box Warnings: Cardiovascular risks limit extended use in some patient groups.
- Labeling modifications: Could impact market uptake.
Strategic Opportunities
- New Indications: Exploring indications like cancer pain or rare inflammatory conditions.
- Combination Therapies: Use with opioids or other analgesics.
- Biosimilars: Development could solve patent challenges, although regulatory pathways are complex.
Summary Table: Sales Forecast (USD Millions)
| Year | Estimated Revenue | Growth Rate | Notes |
|---|---|---|---|
| 2022 | 950 | N/A | Base year |
| 2023 | 1,050 | 10.5% | Post-pandemic recovery factors |
| 2024 | 1,075 | 2.4% | Market stabilization |
| 2025 | 1,100 | 2.3% | Slight growth from market expansion |
| 2026 | 1,100 | 0% | Patent expiry impacts |
| 2027 | 1,100 | 0% | Possible generic entry |
| 2028+ | Decline possible | - | Increasing generic competition |
Key Takeaways
- CELEBREX's current market is valued at approximately USD 950 million to USD 1 billion annually.
- Growth prospects remain modest but steady, influenced by demographic trends and regulatory considerations.
- Patent expiration around 2028 could significantly impact sales due to generic competition.
- Regional differences influence sales, with North America leading.
- Safety concerns remain pivotal in market dynamics; cardiovascular risks restrict extended use.
FAQs
1. How does CELEBREX compare to other NSAIDs in safety?
It has a lower gastrointestinal risk but involves higher cardiovascular safety concerns than traditional NSAIDs. Regulatory agencies periodically update warnings.
2. What factors could accelerate or hinder sales growth?
Market expansion in emerging regions and new indications could boost sales; safety warnings and generic entry could limit growth.
3. When will generic versions likely impact sales?
Patent expiration in major markets generally occurs around 2028-2030, with generics entering shortly thereafter.
4. Are there regulatory developments influencing CELEBREX?
Yes. Regulatory reviews of cardiovascular safety profiles influence prescribing patterns and label updates.
5. What strategic options could Pfizer pursue?
Developing new formulations, exploring additional indications, and facilitating biosimilar partnerships could sustain sales.
References
[1] Statista. (2022). NSAID market value worldwide.
[2] Pfizer. (2023). CELEBREX product data sheet.
[3] MarketWatch. (2023). Global NSAID market report.
[4] U.S. Food and Drug Administration. (2022). Safety communications on NSAID risks.
[5] IQVIA. (2022). Prescription drug market analysis.
More… ↓
