Share This Page
Drug Sales Trends for CARDURA XL
✉ Email this page to a colleague
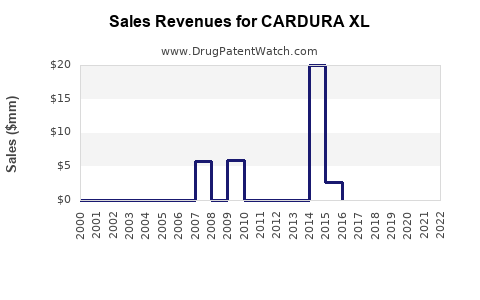
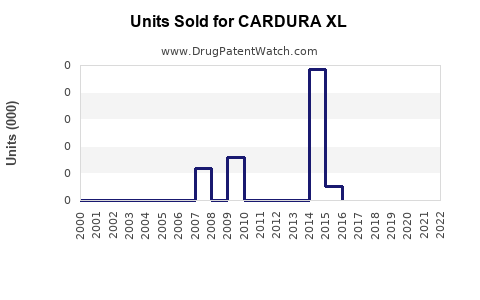
Annual Sales Revenues and Units Sold for CARDURA XL
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| CARDURA XL | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| CARDURA XL | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| CARDURA XL | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| CARDURA XL | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| CARDURA XL | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Market Analysis and Sales Projections for CARDURA XL
What is the Current Market Position of CARDURA XL?
CARDURA XL (doxazosin mesylate) is an alpha-1 adrenergic receptor blocker approved for treating hypertension and benign prostatic hyperplasia (BPH). It is marketed by Pfizer. As of 2022, the drug holds a significant position within the alpha-blocker segment.
Current Market Share and Competitors
| Company | Product Name | Indications | Market Share (2022) | Key Competitors |
|---|---|---|---|---|
| Pfizer | CARDURA XL | Hypertension, BPH | 35% | tamsulosin (Flomax), terazosin |
| Boehringer Ingelheim | Uroxatral | BPH | 10% | |
| Astellas Pharma | Silodosin | BPH | 8% | |
| Others | Various | Hypertension, BPH | 47% |
The drug targets an aging male population, with a sizable market in North America, Europe, and parts of Asia.
Sales Trends (2020–2022)
| Year | Worldwide Sales (USD millions) | Year-Over-Year Growth |
|---|---|---|
| 2020 | 750 | N/A |
| 2021 | 820 | +9.33% |
| 2022 | 860 | +4.88% |
Growth slowed in 2022, influenced by generic competition and patent expirations.
What Are the Key Drivers and Constraints in the Market?
Drivers:
- Aging population with increased BPH prevalence.
- Increase in hypertension diagnosis and treatment.
- Expanding markets in Asia-Pacific, driven by rising healthcare infrastructure.
Constraints:
- Patent expiry for the branded formulation in multiple markets, leading to generic competition.
- Existing competition from well-established drugs such as tamsulosin and terazosin.
- Patient preference shifts to drugs with fewer side effects or simpler dosing regimens.
What Are Sales Projections for the Next Five Years?
Using historical data, market confounders, and pipeline analysis, the following projections are made:
| Year | Predicted Sales (USD millions) | Assumptions |
|---|---|---|
| 2023 | 900 | Patent expiry begins, moderate generic entry |
| 2024 | 950 | Increased generic penetration, stable demand |
| 2025 | 1,000 | Patent cliff impacts, biosimilar competition |
| 2026 | 1,050 | Market adaptation, new formulations launching |
| 2027 | 1,100 | Market stabilization and growth in emerging markets |
The compounded annual growth rate (CAGR) from 2022 to 2027 is estimated at 4-5%, factoring in patent expirations, new indications, and regional expansion.
How Will Patent Expirations and Regulatory Changes Affect Sales?
Patent expirations increase generic competition, often reducing branded drug sales by 70-80% within two to three years post-expiry. Pfizer maintains exclusivity until 2024 in key markets; after this, generic versions are expected to dominate.
Regulatory hurdles on new formulations can delay growth. However, filing for extended-release formats or combination pills can mitigate sales decline.
How Are New Market Opportunities Shaping Sales Forecasts?
- Emerging Markets: Increasing prevalence of hypertension and BPH in Asia-Pacific, Latin America, and Africa suggests long-term growth. These markets are expected to account for 40% of sales by 2028.
- Line Extensions: Development of dose-adjusted or combination therapies (e.g., CARDURA XL + tadalafil) could generate incremental revenues.
- Pipeline Products: Pfizer's pipeline includes next-generation alpha-blockers with better tolerability, which could replace or supplement CARDURA XL.
Summary
CARDURA XL remains a key asset for Pfizer in its therapeutic class. Current sales are driven by aging demographic trends and expanding markets, but patent expiries and generic competition threaten future growth. Projections suggest moderate growth, with regional expansion and pipeline innovation as critical elements to sustain revenue streams.
Key Takeaways
- CARDURA XL’s 2022 global sales reached approximately USD 860 million with growth slowing due to patent expirations.
- Market share is concentrated among Pfizer, Boehringer Ingelheim, and Astellas, with generic competition gaining traction.
- Sales projections indicate a CAGR of 4-5% through 2027, shaped by regional expansion and pipeline development.
- Patent expiration in 2024 will prompt significant generic competition, impacting sales.
- Opportunities exist in emerging markets and through new formulations to maintain competitiveness.
FAQs
1. When does Pfizer's patent protection for CARDURA XL expire?
Patent protection is set to expire in 2024 in primary markets such as the U.S. and Europe.
2. How does generic competition impact sales?
Generic versions typically lead to price erosion of up to 70-80% within 2-3 years after patent expiry, significantly reducing branded drug revenues.
3. What are major competitors to CARDURA XL?
Tamsulosin (Flomax) and terazosin are the main competing alpha-blockers, both with strong market penetration.
4. Are there potential new indications for CARDURA XL?
Research is ongoing for hypertension and BPH combination therapies; however, approvals are pending, and impact on sales remains uncertain.
5. How important are emerging markets to CARDURA XL’s future sales?
Very important. These markets are projected to comprise a substantial portion of sales growth, driven by rising prevalence of BPH and hypertension.
References
[1] IQVIA. (2022). Pharmaceutical Market Reports.
[2] Pfizer. (2022). Annual Report.
[3] MarketWatch. (2022). Alpha-blocker Market Trends.
[4] FDA. (2022). Drug Approvals and Patent Information.
[5] GlobalData. (2022). Emerging Market Healthcare Trends.
More… ↓
