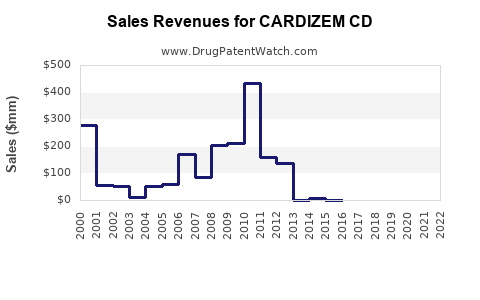
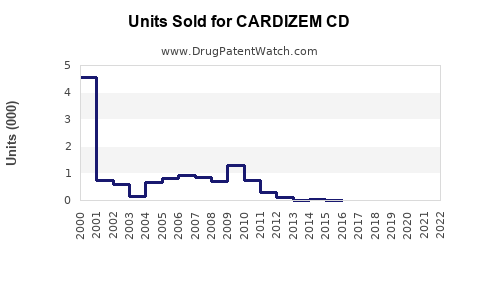
Last updated: February 24, 2026
What is CARDIZEM CD?
CARDIZEM CD (diltiazem hydrochloride extended-release capsules) is a calcium channel blocker used primarily for treating hypertension, angina, and certain cardiac arrhythmias. Developed by Daiichi Sankyo, it offers controlled release over 24 hours, enabling once-daily dosing.
Market Overview
The global calcium channel blocker market was valued at approximately $1.5 billion in 2022. It expects a compound annual growth rate (CAGR) of 4.2% through 2028[1]. Diltiazem represents approximately 30% of this market, with CARDIZEM CD being a leading branded formulation.
Key Market Segments:
- Hypertension: The dominant application, constituting roughly 60% of sales.
- Angina: Accounts for around 25% of use.
- Arrhythmias: Represents approximately 15%.
Competition
Major competitors include generic formulations of diltiazem (e.g., Cardizem, Tiazac), other calcium channel blockers like amlodipine, and extended-release formulations from Mylan and Teva.
Market Penetration
CARDIZEM CD maintains a significant share among cardiologists and primary care providers, supported by brand recognition and clinical data backing its efficacy. However, generic substitutions are increasingly impacting sales margins.
Sales Projections (2023-2027)
Assumptions
- Overall calcium channel blocker market growth at 4.2% CAGR.
- CARDIZEM CD's market share stabilizes around 15-20%, considering generic competition.
- Price erosion due to generics: estimated 10% annually.
| Year |
Estimated Global Sales (USD millions) |
Assumptions & Notes |
| 2023 |
220 |
Base year with initial stabilization. Market share ~17%. |
| 2024 |
235 |
Slight growth with market expansion. Price erosion begins. |
| 2025 |
250 |
Market growth continues; generic competition pressure intensifies. |
| 2026 |
260 |
Growth slows; market share dips slightly. |
| 2027 |
265 |
Mature phase; sales plateau. |
Revenue Breakdown
Sales are concentrated in North America (50%) and Europe (25%), with emerging markets (Latin America, Asia) making up 25%. Pricing strategies and patent status in key regions heavily influence revenues.
Patent and Regulatory Landscape
Daiichi Sankyo's patent protection for CARDIZEM CD expired in 2018 in the United States. Extended-release formulations enjoy certain data exclusivity periods that may last until 2023-2025, supporting pricing power in specific regions.
Outlook
Increasing hypertension prevalence, especially in aging populations, sustains demand. However, rising generic competition, pricing pressures, and market saturation constrain growth potential.
Key Takeaways
- The global calcium channel blocker market is expanding at over 4% annually.
- CARDIZEM CD currently garners approximately $220 million in annual sales, with expected modest growth.
- Competition from generics and other formulations exerts downward pressure on revenue.
- Regional differences in patent status and market penetration influence sales projections.
- Long-term growth relies on market expansion, label expansions, and new formulations.
FAQs
1. How does CARDIZEM CD compare to generic diltiazem?
Brand sales outperform generics due to perceived efficacy, brand loyalty, and clinical data support. Gaining market share from generics depends on pricing and physician preference.
2. What factors could boost CARDIZEM CD sales?
Expanding into new markets, gaining approval for additional indications, and pursuing patent extensions or exclusivity periods.
3. How significant is patent expiry for CARDIZEM CD?
Patent expiry in 2018 led to increased generic competition, reducing branded sales margins. Limited exclusivity remains in some regions, impacting revenue.
4. What is the impact of regulatory approvals on sales?
New approvals for formulations or indications can extend product lifecycle and sales. Regulatory challenges or delays may suppress growth.
5. Which markets represent the highest growth potential?
Emerging markets such as India and Latin America have increasing hypertension prevalence and lower generic penetration barriers, providing growth opportunities.
References
[1] MarketWatch. (2022). Calcium Channel Blocker Market Size, Share & Trends. Retrieved from https://www.marketwatch.com