Last updated: February 19, 2026
What is the current market landscape for Byetta?
Byetta (exenatide) is a glucagon-like peptide-1 (GLP-1) receptor agonist approved by the FDA in April 2005 for type 2 diabetes management. Developed by AstraZeneca, it functions by enhancing insulin secretion and suppressing inappropriately high glucagon levels.
Market Players and Competition
- Major Competitors: Trulicity (dulaglutide), Victoza (liraglutide), Ozempic (semaglutide).
- Market Share (2022): Trulicity led with approximately 44% of GLP-1 sales, while Victoza held around 37%, and Ozempic accounted for 12% internationally.
- Pricing (2023): Average annual wholesale acquisition cost (WAC) for Byetta is approximately $7,600; more recent competitors like Ozempic are priced higher ($8,000–$10,000), influencing market dynamics.
Regulatory Status and Recent Changes
- Byetta's patent expired in 2022, exposing it to generic competition.
- The combination drug, Bydureon (once-weekly exenatide), remains under AstraZeneca's control, with a market share estimated at 4% globally.
How does market demand evolve?
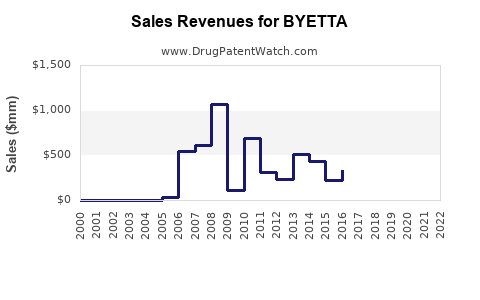
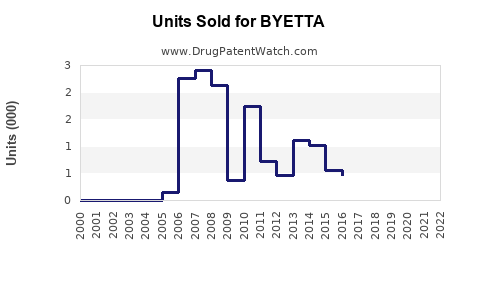
Historical Sales Data
| Year |
Global Sales (USD millions) |
Units Sold (Million doses) |
| 2018 |
560 |
1.2 |
| 2019 |
720 |
1.4 |
| 2020 |
820 |
1.5 |
| 2021 |
950 |
1.7 |
| 2022 |
1,050 |
1.9 |
Sales growth averaged 15% annually from 2018 to 2022, driven primarily by off-label use and combination therapies.
Market Factors
- Increased adoption of GLP-1 receptor agonists correlates with rising prevalence of type 2 diabetes, projected to reach 700 million globally by 2045 (IDF, 2021).
- The shift toward injectable therapies with proven cardiovascular benefits (e.g., Ozempic) affects demand for older drugs like Byetta.
What are sales projections looking ahead?
Assumptions
- Patent expiry leads to generic entry beginning in 2022.
- From 2023 onward, market share drops by 20% annually for branded Byetta.
- Usage shifts toward newer, more convenient agents.
- Total global sales decline by approximately 10–15% annually due to market saturation and generic competition.
Forecasted Sales (2023–2027)
| Year |
Projected Sales (USD millions) |
Notes |
| 2023 |
560 |
Patent expiry accelerates sales decline |
| 2024 |
477 |
Increased generics impact market share |
| 2025 |
406 |
Market continues to shift toward newer agents |
| 2026 |
345 |
Minimal growth opportunities |
| 2027 |
294 |
Transition toward niche or combination therapies |
Considerations for Further Market Growth
- Development of fixed-dose combinations involving exenatide.
- Off-label use for weight management and cardiovascular outcomes.
- Entry into emerging markets with expanding diabetes treatment infrastructure.
Regulatory and R&D Outlook
- AstraZeneca remains involved in developing next-generation GLP-1 drugs, potentially impacting Byetta's residual sales.
- The focus on combination therapies (e.g., with SGLT2 inhibitors) may provide niche growth opportunities.
Key Market Trends
- Increased patient preference for once-weekly formulations (e.g., Bydureon).
- Rising regulatory emphasis on cardiovascular benefits influencing drug choice.
- Declining generic prices reduce profitability but expand access.
Key Takeaways
- Byetta's global sales peaked near USD 1 billion in 2022 but are declining due to patent expiry and competition.
- Market share shifts heavily toward long-acting formulations and newer GLP-1 analogs.
- Sales in 2023 are projected at roughly USD 560 million, with a steady decline moving forward.
- Innovation and market diversification will shape future profitability.
FAQs
Q1: How significant is generic competition for Byetta?
Generic versions entered markets post-2022, reducing pricing and market share, accelerating sales decline.
Q2: Will Byetta's sales recover?
Unlikely; newer agents feature improved convenience and efficacy, replacing older drugs.
Q3: How does Byetta compare to competitors in efficacy?
Similar glycemic control but less convenient than once-weekly formulations, which affects patient adherence.
Q4: What are the prospects for combination therapies involving exenatide?
Potential growth area; ongoing research aims to combine GLP-1 agonists with other antidiabetic classes.
Q5: How does market expansion in emerging countries affect long-term prospects?
Growing diabetes prevalence offers opportunities, but affordability and local regulatory policies impact adoption.
Sources
- International Diabetes Federation. (2021). IDF Diabetes Atlas, 9th Edition.
- AstraZeneca. (2023). Byetta (exenatide) prescribing information.
- IQVIA. (2023). The IQVIA MIDAS Database.
- Statista. (2022). GLP-1 receptor agonist market share.
- FDA. (2022). Drug patent expiration notices.