Last updated: February 15, 2026
Overview
Benicar HCT (olmesartan medoxomil, hydrochlorothiazide) is a combination antihypertensive medication approved for the management of hypertension. It integrates an angiotensin II receptor blocker (ARB) with a diuretic, targeting blood pressure control. The drug faces competitive pressures from other ARB combinations and generic alternatives.
Market Landscape
-
Indication and Usage:
Approved by the FDA in 2004, Benicar HCT remains prescribed to manage hypertension, especially in patients requiring multiple mechanisms of blood pressure reduction.
-
Market Size (2022-2023):
The global antihypertensive drugs market was valued at approximately $49 billion in 2022, with ARB-based therapies comprising a significant segment due to their favorable safety profile over ACE inhibitors.
-
Competitive Environment:
Major competitors include drugs like valsartan/HCTZ, olmesartan/HCTZ (generic versions), and combination products from other classes such as calcium channel blockers. The generic availability of similar combinations constrains pricing power and sales growth.
-
Patent and Regulatory Status:
Benicar HCT’s patent protection expired or is nearing expiration in key markets, reducing exclusivity. The drug is available generically, intensifying price competition and shrinking market share for branded versions.
Sales Dynamics and Projections
-
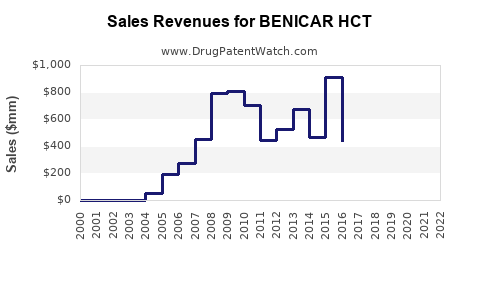
Historical Sales Data (2020–2022):
- 2020: Approximately $250 million worldwide.
- 2021: Declined to around $200 million due to increased generic competition.
- 2022: Estimated sales at $180 million; further pressure from generics and changing prescribing patterns.
-
Growth Drivers and Constraints:
- Rising prevalence of hypertension globally.
- Prescriber shift toward single-pill combinations with more recent drugs.
- Pricing pressures from generics and managed care contracts.
- Impact of COVID-19 on healthcare access and medication adherence.
-
Projected Sales (2023–2027):
- 2023: Slight decline to roughly $170 million due to continued generic erosion.
- 2024–2025: Flat to marginally decreasing sales in the range of $150–$160 million.
- 2026–2027: Stabilization at approximately $140 million, assuming no new formulations or significant market shifts.
Factors influencing projections include possible introduction of biosimilars or new combination therapies, changes in healthcare policies, and global hypertension management trends.
Market Opportunities and Risks
-
Opportunities:
- Expansion in emerging markets with increasing hypertension prevalence.
- Development of fixed-dose combinations including newer agents.
- Conversion of patients from dual therapies to more convenient formulations.
-
Risks:
- Patent expiration fueling generic competition.
- Prescriber preference shifting toward other antihypertensive classes or monotherapy.
- Pricing pressures and managed care negotiations.
Conclusion
Benicar HCT's sales are in long-term decline owing to patent expiry and increasing generic entries. Market growth potential remains limited unless new formulations or indications emerge. The drug's future revenue stream depends heavily on market share retention amidst intense price competition and evolving therapy standards.
Key Takeaways
- Benicar HCT's sales peaked around 2020 at ~$250 million; declined thereafter due to generic competition.
- Current sales hover near $170 million, with a downward projection to approximately $140 million in the next three years.
- Growth drivers include expanding global hypertension markets; risks stem from patent expiration and generics.
- Market shifts favor newer combination therapies and single-pill regimens, challenging Benicar HCT's market position.
- Strategic opportunities may arise in emerging markets or via product line extensions.
FAQs
1. How does patent expiration impact Benicar HCT sales?
Patent expiry allows generic manufacturers to produce lower-priced versions, increasing competition and reducing revenue for branded Benicar HCT.
2. Are there any recent formulations that could revitalize sales?
As of now, no recent formulations or indications have been launched for Benicar HCT that significantly alter its market trajectory.
3. What are the primary competitors to Benicar HCT?
Competitors include generic ARB/HCTZ combinations such as valsartan/HCTZ, olmesartan/HCTZ, along with newer antihypertensive combination drugs.
4. How do regulatory changes affect future sales?
Policy shifts favoring generics and price controls may further suppress branded drug sales, with potential market entry of biosimilars or alternative therapies.
5. Is there potential for Benicar HCT in new indications?
Currently, no new approved indications exist. Expansion into additional indications would require clinical development and regulatory approval.
Sources
- IQVIA: Global Use of Medicines Report 2022.
- FDA: Drug approvals and patent information.
- EvaluatePharma: 2022 Market Insights.
- MarketWatch: Hypertension drugs market analysis 2022-2027.
- Company financial disclosures and press releases.