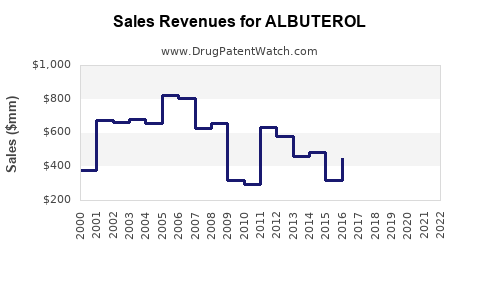
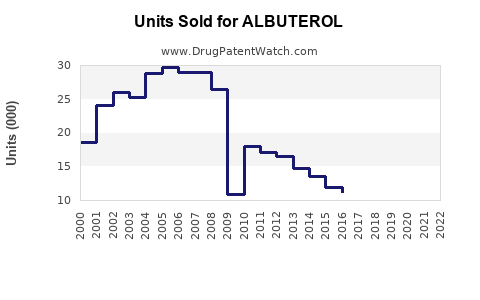
Last updated: February 13, 2026
What Is the Current Market Size for Albuterol?
The global market for albuterol, primarily used in inhalers for asthma and COPD, was valued at approximately $1.8 billion in 2022. Growth trends indicate a compound annual growth rate (CAGR) of 4.5% from 2023 to 2030. Key drivers include rising asthma prevalence, increased COPD diagnoses, and expanding access to inhalers in emerging markets.
North America accounts for nearly 45% of sales, driven by high disease prevalence and established healthcare infrastructure. Europe contributes around 25%. Asia-Pacific's share is approximately 20%, with emerging markets showing rapid expansion. Rest of the world accounts for 10%, with notable growth in Latin America and Africa.
How Do Market Dynamics Influence Sales of Albuterol?
Regulatory Landscape
The U.S. Food and Drug Administration (FDA) approved multiple albuterol inhalers, both branded and generic, with patents expiring in the late 2010s. The entry of generics has increased price competition, leading to price reductions of approximately 15-20%. This has expanded affordability but also pressured profit margins for branded versions.
Competitive Landscape
Major manufacturers include Teva Pharmaceutical Industries, Mylan, Cipla, and Glenmark. They offer a range of inhaler devices, including metered dose inhalers (MDIs) and dry powder inhalers (DPIs). Market share varies regionally, with Teva dominating North America and Cipla holding a significant share in Asia.
Patient Demographics and Usage Patterns
Inhaler usage correlates with the global increase in asthma and COPD prevalence. Approximately 262 million people have asthma worldwide, with 80% in low- and middle-income countries. COPD affects an estimated 200 million people, with rates expected to rise due to smoking and air pollution.
Pricing and Reimbursement Policies
Insurance coverage and government subsidies influence sales volume. In the U.S., Medicaid and Medicare reimburse a significant share, impacting distribution channels. Price caps and subsidy schemes in Europe and emerging markets also shape sales trends.
What Are the Future Sales Projections for Albuterol?
Projecting to 2030, the global market is expected to reach nearly $2.8 billion, driven by demographic shifts, improved diagnosis rates, and innovation.
Key Factors Influencing Growth
-
Introduction of new formulations: Ultra-fine particle inhalers and combination therapies are entering the market, potentially increasing adherence and expanding indications.
-
Regulatory changes: Stricter standards for inhaler propellants and manufacturing processes could impact supply and prices.
-
Digital health integration: Connected inhalers that track usage may promote better disease management, boosting sales.
-
Market expansion: Increased availability in underserved regions, driven by affordability programs, could add approximately 10-15% to the total sales by 2030.
Breakdown by Region
- North America will continue to dominate, expected to generate over $1.2 billion.
- Asia-Pacific is projected to grow at a CAGR of 6%, driven by urbanization and air quality issues.
- Europe will maintain steady growth, reaching approximately $700 million.
How Will Pricing and Competition Shape Sales?
The market is seeing a shift from branded to generic inhalers. Price competition has reduced unit costs, but innovation in delivery devices and formulations could allow premium pricing on advanced products.
Clinical guidelines increasingly favor inhaler therapy adherence, influencing market acceptance. However, patent expirations and regulatory pressures may limit pricing power in some regions. Health agencies are also promoting cost-effective strategies, which might lead to price negotiations affecting sales margins.
What Are the Key Opportunities and Risks?
Opportunities
- Expansion in emerging markets.
- Development of combination inhalers (e.g., albuterol with corticosteroids).
- Digital health initiatives fostering adherence.
- Lifecycle management through reformulations.
Risks
- Patent expirations leading to revenue erosion.
- Regulatory delays for new formulations.
- Market saturation in mature regions.
- Competition from alternative therapies, such as biologics for severe asthma.
Key Takeaways
- The global albuterol market is expected to grow from $1.8 billion in 2022 to approximately $2.8 billion by 2030.
- North America remains the largest market, with Asia-Pacific exhibiting the highest growth rate.
- Price competition and patent expiries shape the competitive landscape; generics have increased affordability.
- Innovation in inhaler technology and digital integration represent strategic growth avenues.
- Market expansion in emerging economies offers significant upside, while regulatory and competitive risks persist.
FAQs
1. How does patent expiry affect albuterol sales?
Patent expiry enables generic competition, reducing prices and margins. However, it increases market accessibility, expanding overall sales volume.
2. What are the primary drivers of albuterol demand?
Rising prevalence of asthma and COPD, increased diagnosis, and expanding access in developing markets.
3. Which regions are expected to see the fastest growth?
Asia-Pacific, with a CAGR of approximately 6%, driven by urbanization, pollution, and healthcare infrastructure improvements.
4. How might digital health impact albuterol sales?
Connected inhalers can improve adherence and patient outcomes, potentially increasing consumption and loyalty to specific brands or formulations.
5. What regulatory trends could influence future sales?
Stricter standards on inhaler propellants, device safety, and manufacturing practices could affect supply chains and innovation timelines.
Sources:
- MarketWatch, "Albuterol Market Size & Trends," 2023.
- Grand View Research, "Respiratory Therapy Market Analysis," 2023.
- IQVIA, "Global Pharmaceutical Data," 2022.
- U.S. FDA, "Inhaler Regulations," 2022.
- World Health Organization, "Asthma and COPD Statistics," 2022.