Share This Page
Drug Sales Trends for ALBUTEROL SULFATE
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for ALBUTEROL SULFATE (2022)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
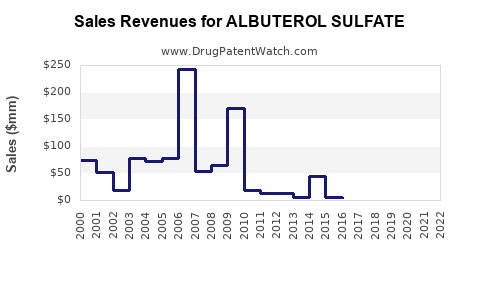
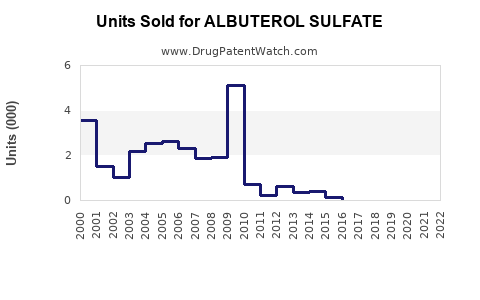
Annual Sales Revenues and Units Sold for ALBUTEROL SULFATE
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| ALBUTEROL SULFATE | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| ALBUTEROL SULFATE | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| ALBUTEROL SULFATE | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| ALBUTEROL SULFATE | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| ALBUTEROL SULFATE | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| ALBUTEROL SULFATE | ⤷ Start Trial | ⤷ Start Trial | 2017 |
| ALBUTEROL SULFATE | ⤷ Start Trial | ⤷ Start Trial | 2016 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Albuterol Sulfate Market Analysis and Sales Projections
Albuterol sulfate, a short-acting beta-agonist (SABA), is a cornerstone therapy for managing bronchospasm in conditions like asthma and chronic obstructive pulmonary disease (COPD). Its efficacy in rapidly opening airways makes it an indispensable medication. This analysis details the current market landscape, key market drivers, competitive environment, and projected sales for albuterol sulfate.
What is the Current Market Size and Growth Rate for Albuterol Sulfate?
The global albuterol sulfate market was valued at approximately $1.7 billion in 2023. The market is projected to grow at a compound annual growth rate (CAGR) of 3.2% from 2024 to 2030, reaching an estimated $2.1 billion by the end of the forecast period. This growth is primarily fueled by the increasing prevalence of respiratory diseases and greater accessibility to generic medications.
Key Market Segments
- By Product Type:
- Metered-dose inhalers (MDIs)
- Nebulizer solutions
- Tablets
- Syrups
- By Application:
- Asthma
- COPD
- Exercise-induced bronchospasm
- Other respiratory disorders
- By Distribution Channel:
- Hospital pharmacies
- Retail pharmacies
- Online pharmacies
- By Region:
- North America
- Europe
- Asia-Pacific
- Latin America
- Middle East & Africa
The MDI segment holds the largest market share due to its convenience and widespread prescription. Nebulizer solutions are also significant, particularly for severe cases or patients unable to use inhalers effectively.
What are the Primary Drivers of the Albuterol Sulfate Market?
Several factors are contributing to the sustained demand for albuterol sulfate.
Increasing Prevalence of Respiratory Diseases
The incidence of asthma and COPD continues to rise globally. Factors contributing to this include environmental pollution, urbanization, and an aging population. The World Health Organization (WHO) estimates that 235 million people worldwide have asthma, and COPD is projected to become the third leading cause of death globally by 2030 [1]. This growing patient population directly translates to increased demand for effective bronchodilators like albuterol sulfate.
Growing Demand for Generic Medications
Albuterol sulfate is largely a genericized drug, with its patent expiring decades ago. The availability of multiple generic manufacturers has led to significant price reductions, making the drug more affordable and accessible. This affordability is crucial for broad patient access, especially in developing economies. The market for generic respiratory inhalers is robust, with significant market penetration.
Advancements in Drug Delivery Systems
While albuterol sulfate is an established molecule, ongoing developments in inhaler technology aim to improve drug delivery and patient adherence. Innovations in MDI design, such as breath-actuated inhalers and those with dose counters, enhance ease of use and reduce the risk of incorrect administration. Nebulizer devices are also becoming more portable and efficient, improving treatment for patients at home.
Government Initiatives and Healthcare Policies
Various government initiatives aimed at improving respiratory healthcare access and affordability further bolster the market. Subsidies for essential medicines, public health campaigns focusing on respiratory disease awareness, and reimbursement policies that favor generic drugs contribute to market expansion.
Who are the Key Players in the Albuterol Sulfate Market?
The albuterol sulfate market is highly competitive, characterized by numerous generic manufacturers and a few branded product holders. Key players include:
- Teva Pharmaceutical Industries Ltd.
- Hikma Pharmaceuticals PLC
- Mylan N.V. (now Viatris Inc.)
- Sun Pharmaceutical Industries Ltd.
- Cipla Ltd.
- Sandoz International GmbH (a Novartis company)
- Boehringer Ingelheim International GmbH (primarily for branded products and combinations)
- AstraZeneca PLC (for combination therapies and branded albuterol products)
This competitive landscape drives pricing pressures but also ensures a consistent supply chain. The market has seen consolidation, with larger pharmaceutical companies acquiring smaller generic players to expand their respiratory portfolios.
What are the Major Challenges Facing the Albuterol Sulfate Market?
Despite its strong market position, albuterol sulfate faces several challenges.
Regulatory Hurdles and Patent Expirations
While the primary patents for albuterol sulfate have long expired, manufacturers face ongoing regulatory scrutiny for product quality, manufacturing processes, and labeling. Obtaining and maintaining approvals in different global markets requires significant investment and adherence to strict guidelines.
Competition from Newer Therapies
While albuterol sulfate remains a first-line rescue medication, advancements in long-acting bronchodilators (LABAs) and inhaled corticosteroids (ICS) for maintenance therapy, as well as newer biologics for severe asthma, present competition. These therapies offer different mechanisms of action and improved long-term disease control for certain patient populations. However, albuterol sulfate's role as an acute relief medication is unlikely to be fully replaced.
Supply Chain Disruptions and Manufacturing Issues
As a widely used generic drug, albuterol sulfate is susceptible to supply chain disruptions caused by raw material shortages, manufacturing plant issues, or geopolitical events. Shortages of albuterol inhalers have occurred periodically in the past, highlighting the vulnerability of the supply chain for essential medicines.
Environmental Concerns with MDIs
Traditional MDIs utilize propellants, some of which are hydrofluorocarbons (HFCs), potent greenhouse gases. There is increasing regulatory and environmental pressure to transition to more environmentally friendly propellants or alternative delivery methods. Companies are actively researching and developing HFC-free MDIs [2].
What are the Sales Projections for Albuterol Sulfate?
Based on the current market dynamics, growth drivers, and challenges, the sales projections for albuterol sulfate are as follows:
| Year | Estimated Market Value (USD Billion) | CAGR (2024-2030) |
|---|---|---|
| 2024 | 1.76 | 3.2% |
| 2025 | 1.82 | 3.2% |
| 2026 | 1.88 | 3.2% |
| 2027 | 1.94 | 3.2% |
| 2028 | 2.00 | 3.2% |
| 2029 | 2.06 | 3.2% |
| 2030 | 2.12 | 3.2% |
These projections assume a consistent demand driven by the prevalence of respiratory diseases and the continued reliance on albuterol sulfate as a primary rescue medication. The growth rate is moderate, reflecting the mature nature of the drug and the competitive generic market.
How is Albuterol Sulfate Used in Different Regions?
Regional market dynamics for albuterol sulfate vary based on healthcare infrastructure, economic development, and disease prevalence.
North America
This region represents a significant market due to a high prevalence of asthma and COPD, a well-established healthcare system, and widespread access to generic medications. The demand is driven by both MDI and nebulizer formulations. Regulatory bodies like the FDA closely monitor product quality and supply.
Europe
Similar to North America, Europe has a substantial market for albuterol sulfate, supported by national healthcare systems and high disease burden. Market access and reimbursement policies play a crucial role. Environmental regulations concerning propellants are becoming more influential in this region.
Asia-Pacific
This region is experiencing the fastest growth due to increasing respiratory disease incidence, expanding healthcare access, and a growing middle-class population with greater purchasing power. Countries like China and India represent large and rapidly developing markets for albuterol sulfate. Generic drug penetration is high.
Latin America, Middle East & Africa
These regions present significant growth potential, driven by an increasing focus on public health initiatives and improved access to essential medicines. Affordability is a critical factor, making generic albuterol sulfate highly sought after. Challenges include underdeveloped healthcare infrastructure in some areas and varying regulatory frameworks.
What are the Future Trends in the Albuterol Sulfate Market?
Several trends are likely to shape the future of the albuterol sulfate market.
Transition to HFC-Free Propellants
The industry is actively working towards developing and launching albuterol MDIs that use hydrofluoroalkane (HFA) propellants with lower global warming potential or entirely new propellant systems [2]. This transition will require significant investment in R&D and manufacturing reformulation. Regulatory mandates or incentives could accelerate this shift.
Combination Therapies
While albuterol sulfate is primarily used as a single agent for rescue, research into novel delivery methods or its use in combination with other active pharmaceutical ingredients (APIs) for specific patient subgroups might emerge. However, its established role as a stand-alone rescue inhaler is unlikely to change dramatically in the near term.
Digital Health Integration
The integration of smart inhaler technology, which tracks usage patterns and provides adherence feedback, could indirectly impact albuterol sulfate usage by improving patient management. This technology enhances the effectiveness of treatment plans involving rescue medications.
Supply Chain Resilience
Given past disruptions, there will be an increased focus on building more resilient and diversified supply chains for albuterol sulfate and its key raw materials. This could involve multiple manufacturing sites, strategic stockpiling, and greater transparency in the supply chain.
Key Takeaways
The albuterol sulfate market is stable and projected to grow moderately, driven by the persistent and increasing burden of respiratory diseases globally. Its position as a critical, affordable rescue medication for asthma and COPD ensures continued demand. The market is characterized by intense competition among generic manufacturers, leading to price sensitivity. Key future trends include the transition to more environmentally friendly propellants for MDIs, a focus on supply chain resilience, and the potential for smart inhaler integration to improve patient adherence.
Frequently Asked Questions
1. What is the primary therapeutic use of albuterol sulfate?
Albuterol sulfate is primarily used to treat bronchospasm associated with asthma, COPD, and other respiratory conditions. It acts as a short-acting bronchodilator, rapidly opening the airways for easier breathing.
2. What is the difference between albuterol sulfate and salbutamol?
Albuterol sulfate and salbutamol are the same active pharmaceutical ingredient. "Albuterol" is the United States adopted name (USAN) for the drug, while "salbutamol" is the International Nonproprietary Name (INN). The drug is marketed under both names in different regions.
3. What are the main types of albuterol sulfate delivery devices?
The most common delivery devices for albuterol sulfate are metered-dose inhalers (MDIs) and nebulizer solutions. Oral tablets and syrups are also available but are less common for acute symptom relief.
4. Are there any significant safety concerns associated with albuterol sulfate?
While generally safe and effective when used as prescribed, albuterol sulfate can cause side effects such as tremors, nervousness, headache, and increased heart rate. Overuse can lead to paradoxical bronchospasm or diminished effectiveness. It is crucial for patients to use it as a rescue medication and not as a daily maintenance therapy, which is typically managed with other drug classes.
5. What impact will the move to HFC-free propellants have on the market?
The transition to hydrofluorocarbon (HFC)-free propellants for metered-dose inhalers is driven by environmental concerns. This transition will require significant investment from manufacturers in research, development, and manufacturing line changes. It may also lead to temporary price adjustments for new formulations and could influence market share as companies roll out new products.
Cited Sources
[1] World Health Organization. (2023). Asthma. https://www.who.int/news-room/fact-sheets/detail/asthma [2] U.S. Food and Drug Administration. (2021). FDA Statement on Metered-Dose Inhalers (MDIs) and Environmental Impact. https://www.fda.gov/drugs/news-events-human-drugs/fda-statement-metered-dose-inhalers-mds-and-environmental-impact
More… ↓
