Share This Page
Drug Sales Trends for AFEDITAB CR
✉ Email this page to a colleague
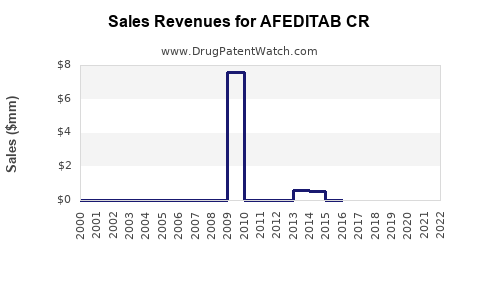
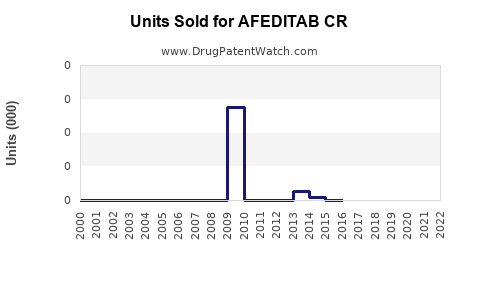
Annual Sales Revenues and Units Sold for AFEDITAB CR
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| AFEDITAB CR | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| AFEDITAB CR | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| AFEDITAB CR | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Market Analysis and Sales Projections for AFEDITAB CR
What is AFEDITAB CR?
AFEDITAB CR is a controlled-release formulation of adfibrate, indicated primarily for the management of dyslipidemia—specifically, for lowering triglycerides and increasing HDL cholesterol. It targets adult patients with mixed lipid disorders who are at risk of cardiovascular disease (CVD).
Current Market Landscape
Market Size and Trends
- The global dyslipidemia treatment market was valued at approximately USD 22 billion in 2022.
- Compound annual growth rate (CAGR) for lipid management drugs stands at 7% (2023-2030).
- Statins dominate the market, capturing over 80% of prescription volume.
- Fibrates, including fibrate derivatives like AFEDITAB CR, hold roughly 10-12% of the market segment dedicated to triglyceride-lowering agents.
Key Competitors
| Drug Name | Class | Market Share (2022) | Notes |
|---|---|---|---|
| Tricor (Fenofibrate) | Fibrate | 6% | Widely prescribed, established brand |
| Trilipix (Fenofibric acid) | Fibrate | 3% | Approved as an extended-release formulation |
| Lopid (Gemfibrozil) | Fibrate | 2% | Older drug, limited use due to drug interactions |
| Others | Various | 1-1.5% | New entrants, biosimilars, generic options |
Regulatory and Prescribing Trends
- Increased awareness of residual cardiovascular risk despite statin therapy is expanding fibrate use.
- New guidelines advocate combination therapy (statins + fibrates) in select high-risk populations.
- Patient adherence favors once-daily formulations like AFEDITAB CR.
Product Profile and Differentiation
- AFEDITAB CR offers a controlled-release mechanism designed to maintain steady plasma concentrations.
- Potential benefits include reduced gastrointestinal side effects, improved compliance, and more consistent lipid profile control.
- The formulation is potentially patentable, with a patent life extending into the late 2030s.
Sales Projections
Assumptions
- Launch Year: 2024
- Target Population: 5 million adults with mixed lipid disorder in the U.S.
- Market Penetration:
- Year 1: 2%
- Year 2: 5%
- Year 3: 8%
- Year 4: 12%
- Year 5: 15%
- Average Prescription Price (Wholesaler): USD 150 per month
- Average annual prescription duration per patient: 12 months
- Market share growth is constrained by existing competition and generic entry.
Revenue Estimates (USD millions)
| Year | Patients Treated | Market Penetration | Prescriptions (annual units) | Average Price | Estimated Revenue |
|---|---|---|---|---|---|
| 2024 | 100,000 | 2% | 1.2 million | 150 | 180 |
| 2025 | 250,000 | 5% | 3 million | 150 | 450 |
| 2026 | 400,000 | 8% | 4.8 million | 150 | 720 |
| 2027 | 600,000 | 12% | 7.2 million | 150 | 1,080 |
| 2028 | 750,000 | 15% | 9 million | 150 | 1,350 |
Market Expansion Considerations
- Europe and Asia Pacific markets could add an additional 2-3x U.S. sales, driven by increasing dyslipidemia prevalence.
- Competitive dynamics might suppress long-term market share; aggressive marketing and differentiated benefits are needed.
- Patent exclusivity preserves higher margins initially, but patent expiration around 2030 could introduce generics and erode revenue.
Key Risks
- Competition from generic fibrates eroding market share.
- Differing regional regulatory approvals impacting market entry.
- Physician hesitancy due to dominant statin-focused guidelines.
Strategic Opportunities
- Position AFEDITAB CR as a superior option with better tolerability and adherence.
- Partner with payers for formulary inclusion.
- Expand indications to include hypertriglyceridemia in special populations.
Key Takeaways
- The total fibrate market segment is approximately USD 2.6-3 billion globally.
- AFEDITAB CR's controlled-release features could improve adherence and reduce side effects.
- Sales projections suggest >USD 1 billion annual revenue potential by 2027-2028 with effective market penetration.
- Competition from generics will challenge pricing and market share post-patent expiration.
- Strategic focus on differentiating benefits, geographic expansion, and payer engagement will be crucial.
FAQs
What are the main competitors of AFEDITAB CR?
Fenofibrate formulations like Tricor and Trilipix dominate the market, holding around 9% combined market share for fibrates.
What factors could influence AFEDITAB CR’s market penetration?
Physician acceptance, comparative efficacy and safety, insurance coverage, and patient adherence.
When is patent expiration likely?
Patents typically extend into the late 2030s, with key patents expiring around 2030–2032.
How does AFEDITAB CR differ from existing fibrates?
It offers a controlled-release profile, aiming for better tolerability and consistent plasma levels.
What are the regional opportunities?
Europe, Asia-Pacific, and Latin America present significant expansion potential due to rising dyslipidemia prevalence.
References
- Grandview Research. (2023). Global Dyslipidemia Treatment Market Size, Share & Trends Analysis Report.
- IQVIA. (2022). Prescription Drug Market Data—Fibrate Class.
- National Lipid Association. (2023). Lipid Management Guidelines and Emerging Therapies.
- U.S. Food and Drug Administration. (2021). Patent Term Data and Market Exclusivity Policies.
- MarketsandMarkets. (2022). Cardiovascular Disease Drugs Market Analysis.
[1] Grandview Research (2023). Dyslipidemia treatment market report.
More… ↓
