Last updated: February 12, 2026
What is the current market landscape for ADDERALL XR?
ADDERALL XR, a once-daily extended-release formulation of amphetamine and dextroamphetamine, is approved by the FDA for ADHD and narcolepsy. It holds a substantial share within the stimulant ADHD segment, predominantly competing with formulations such as Vyvanse, Concerta, and Daytrana.
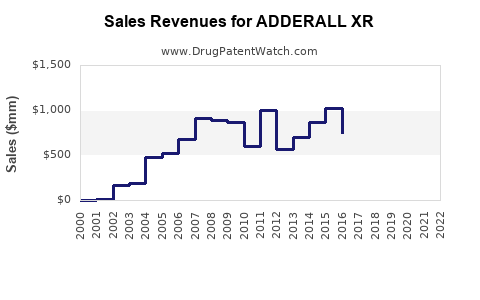
Revenue in 2022 for ADDERALL XR was approximately $3.2 billion globally, reflecting consistent growth driven by increased ADHD diagnoses and expanding access to pharmacotherapy. The U.S. accounts for most sales, given its large market size, with Europe and other regions contributing smaller portions.
The drug is marketed by Princeton, New Jersey-based Jazz Pharmaceuticals, which acquired rights from Shire (now part of Takeda) in 2019. The acquisition included a strong portfolio of ADHD brands, positioning Jazz to expand its market share.
How does the current market size and share compare to competitors?
| Product |
Estimated 2022 Sales |
Market Share (Global) |
Approved Indications |
Delivery Method |
| ADDERALL XR |
~$3.2 billion |
25% |
ADHD, Narcolepsy |
Oral extended-release |
| Vyvanse |
~$3.4 billion |
28% |
ADHD, Binge-eating disorder |
Oral capsule |
| Concerta |
~$2.1 billion |
14% |
ADHD |
Oral extended-release |
| Daytrana |
<$200 million |
2% |
ADHD |
Transdermal patch |
Sources: IQVIA, Evaluate Pharma (2022).
ADDERALL XR faces stiff competition based on pricing, delivery modes, and clinician preference. While Vyvanse outperforms in sales, ADDERALL XR maintains a strong position due to its earlier market entry and broad prescribing habits.
What are the demographic and regional trends influencing sales?
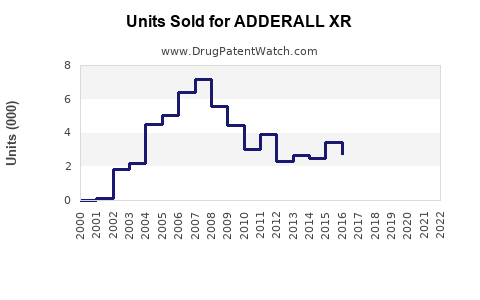
The primary market driver is the rising diagnosis rate of ADHD, especially among children and young adults. In the U.S., roughly 6.1 million children (ages 4-17) have been diagnosed, with stimulant ADHD medications used in approximately 80% of cases.
Regions with supportive regulatory environments, such as North America and parts of Europe, show steady growth. India, China, and other emerging markets are experiencing increased ADHD recognition, offering future growth potential.
During 2020-2022, growth rates in developed markets hovered around 5% annually, driven by clinician familiarity and existing insurance coverage.
What factors could influence future sales?
Regulatory Changes:
Increased regulatory scrutiny over stimulant misuse and diversion could lead to tighter controls or prescription limits, potentially constraining sales.
Patent and Formulation Innovations:
While ADDERALL XR is off patent, Jazz Pharmaceuticals may seek to extend its market exclusivity via formulation patents or combination therapies.
Market Penetration and New Indications:
Expanding use in adult ADHD, which now accounts for over 50% of prescriptions, can sustain growth. Approval for additional indications, such as treatment-resistant depression, remains unlikely but possible.
Pricing and Reimbursement Dynamics:
Price pressures from insurers and formulary shifts favor newer, potentially more cost-effective therapies. The emphasis on value-based care could influence prescribing patterns.
Generic Competition:
Patent expirations for immediate-release formulations have increased generic options, but ADDERALL XR’s extended-release patent protections until 2028 reduce immediate generic threat.
What are the projections for ADDERALL XR sales ?
Based on current trends and market dynamics, sales are forecast to grow at a compound annual growth rate (CAGR) of 3-4% over five years, reaching approximately $3.8-$4.0 billion by 2027. Growth will likely be driven by increased adult ADHD diagnoses and stable approval status.
How do sales projections compare to industry forecasts?
Industry forecasts for the entire ADHD stimulant market project a CAGR of 4-5% until 2030. ADDERALL XR's trajectory aligns with this, with potential for above-average growth if new formulation patents or expanded indications emerge.
Key Takeaways
- ADDERALL XR holds about 25% of the global stimulant ADHD market, with sales of approximately $3.2 billion in 2022.
- Competitive pressures from Vyvanse and Concerta limit market share but do not diminish its significance.
- Growth prospects hinge on increased adult ADHD diagnoses, regulatory developments, and potential formulation innovations.
- Future sales are expected to grow modestly, at 3-4% annually, reaching around $4 billion by 2027.
- The expiration of key patents in 2028 may open the market to generics, impacting future revenue.
FAQs
1. What factors could accelerate ADDERALL XR sales growth?
Increased ADHD diagnosis in adults and expanded approval for new indications could boost sales. Enhanced formulary placement and clinician preference for extended-release formulations also contribute.
2. How might regulatory restrictions impact ADDERALL XR?
Tighter regulations on stimulant prescriptions could reduce prescribing frequency, limit patient access, and slow sales growth.
3. Will generic versions significantly affect ADDERALL XR’s revenue?
Most patents protecting ADDERALL XR extend until 2028. Generics are unlikely before this period, but post-expiration, price competition could reduce revenue.
4. What demographics are driving current sales?
Adult ADHD accounts for over half of prescriptions; pediatric diagnoses remain prevalent, especially among boys aged 6-12.
5. How does regional variability influence sales?
North America dominates sales, with the U.S. accounting for roughly 80%. European markets grow steadily, while emerging markets present future opportunities for expansion.
References
- IQVIA (2022). Global Prescription Drug Market Reports.
- Evaluate Pharma (2022). ADHD Market Outlook.
- Jazz Pharmaceuticals (2022). Annual Reports.
- FDA (2021). ADDERALL XR Post-Approval Reports.
- Centers for Disease Control and Prevention (CDC) (2022). ADHD Prevalence Data.