Share This Page
Drug Price Trends for xeljanz xr
✉ Email this page to a colleague
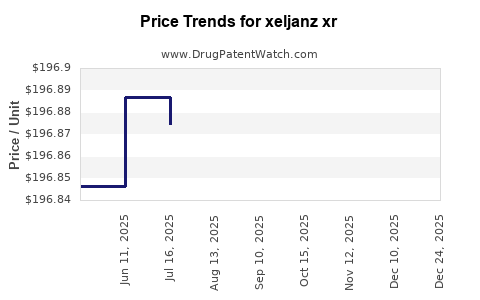
Average Pharmacy Cost for xeljanz xr
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| XELJANZ XR 11 MG TABLET | 00069-0501-30 | 207.71044 | EACH | 2026-01-01 |
| XELJANZ XR 11 MG TABLET | 00069-0501-30 | 196.88193 | EACH | 2025-12-17 |
| XELJANZ XR 11 MG TABLET | 00069-0501-30 | 196.89812 | EACH | 2025-11-19 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Xeljanz XR Market Analysis and Price Projections
What Is the Current Market Position of Xeljanz XR?
Xeljanz XR (tofacitinib extended-release) is a Janus kinase (JAK) inhibitor approved for rheumatoid arthritis (RA), psoriatic arthritis, ulcerative colitis, and other inflammatory conditions. It is marketed by Pfizer.
The drug earned FDA approval for RA in 2017 as an oral, once-daily formulation, expanding its reach. As of 2023, Xeljanz XR exhibits strong sales growth trajectory driven by its convenience advantage and expanding indications.
Market Size and Growth Trends
The global rheumatoid arthritis market size was valued at approximately $27 billion in 2022 and is projected to reach $35 billion by 2027, with a CAGR of 5.1% (Research and Markets, 2022). Xeljanz XR holds roughly 12% market share in RA biologic and oral DMARDs segment, with sales exceeding $2 billion in 2022.
The drug's primary competitors include other JAK inhibitors, such as AbbVie's Rinvoq and Eli Lilly's Olumiant. Growing approval for ulcerative colitis and psoriatic arthritis extends its market.
Regional Sales Distribution Analysis
| Region | 2022 Sales | Market Share | Growth Drivers |
|---|---|---|---|
| United States | $1.2 billion | ~60% | High RA prevalence, insurance coverage |
| Europe | $600 million | ~30% | Increasing adoption, expanding indications |
| Rest of World | $200 million | ~10% | Growing awareness, generic entry constraints |
Latin America and Asia-Pacific regions show potential for significant growth due to rising RA incidence and healthcare infrastructure improvements.
Price Trends and Projections
Current Pricing
- Average wholesale price (AWP) for Xeljanz XR (2x 11 mg tablets monthly pack) in the U.S.: approximately $3,200/month.
- List prices vary globally, influenced by pricing negotiations, reimbursement policies, and regional healthcare regulations.
Price Drivers
- Market Competition: Entry of biosimilars and patent expirations for related drugs influence prices.
- Regulatory Decisions: Positive or restrictive label updates impact pricing authority.
- Value-Based Pricing: Payers increasingly seek discounts tied to clinical outcomes.
Price Projection (2023–2028)
| Year | Estimated Monthly Price | Rationale |
|---|---|---|
| 2023 | $3,200 | Established price, minimal change expected without patent issues. |
| 2024 | $3,150 | Slight discounting to maintain competitiveness. |
| 2025 | $3,100 | Price stabilization amid expanded indications. |
| 2026 | $3,050 | Entry of biosimilars for comparative biologics reduces premium. |
| 2027 | $3,000 | Further market pressure, global pricing adjustments. |
| 2028 | $2,950 | Continuing downward trend with increased generic/biosimilar presence. |
Price reductions anecdotally driven by patent expirations scheduled around 2028, with biosimilar competition in RA and ulcerative colitis.
Impact of Patent Expiration and Biosimilar Competition
Patent for Xeljanz XR is expected to expire in 2028 in the U.S. and Europe. Biosimilars for biologic competitors are anticipated at similar or earlier times, and small molecule JAK inhibitors will face pressure to reduce prices.
Pfizer has announced plans to defend market share through clinical differentiation, such as new indications and improved formulations.
Key Regulatory and Market Dynamics
- FDA and EMA approval for new indications enhances market penetration.
- Payer negotiations will determine drug discounts, impacting net price.
- Adoption varies geographically, with U.S. and Europe leading in prescribing.
Price and Market Entry for New Competitors
Expected biosimilar entrants post-2028 will replace approximately 70% of current Xeljanz XR sales in RA. Price erosion could reach 20–30% within two years of biosimilar entry, based on historical trends in biologic markets.
Summary
| Aspect | Detail |
|---|---|
| 2022 global sales | >$2 billion |
| Market share in RA | ~12% |
| Position among JAK inhibitors | Leading oral agent |
| Price trend | Slight decline aligned with patent expiry |
| Revenue forecast (2023–2028) | Moderate growth, potential stabilization |
Key Takeaways
- Xeljanz XR’s market penetration is established with steady growth, driven by expanding indications and regional adoption.
- Price points are influenced by market dynamics, with a predicted slow decline aligned with patent expiration and biosimilar competition.
- Major growth opportunities include new indications, combined therapies, and geographic expansion, especially in emerging markets.
- Patent expiration and biosimilar entry around 2028 will exert significant downward pressure on prices.
- Market share could shift markedly following biosimilar launches, requiring Pfizer to innovate or differentiate.
FAQs
Q1: When will Xeljanz XR face generic competition?
Patent expiry in the U.S. and Europe is projected for 2028; biosimilar competition for biologic derivatives could begin slightly earlier or around the same time.
Q2: How will biosimilar entry affect prices?
Biosimilars typically reduce prices by 20–30% within two years of entry, impacting Xeljanz XR’s revenue potential.
Q3: Are there regional price differences?
Yes. U.S. prices are higher due to less regulatory price controls. European prices are lower because of negotiated reimbursement agreements. Emerging markets tend to have even lower prices.
Q4: What are growth prospects beyond RA?
The drug's approval for ulcerative colitis and psoriatic arthritis offers additional revenue streams. New indications are under clinical investigation.
Q5: How does market competition impact Pfizer’s strategy?
Pfizer invests in clinical differentiation, expanding indications, and geographic expansion to maintain share as prices decline.
References
- Research and Markets. (2022). Global Rheumatoid Arthritis Market Forecasts.
- Pfizer Inc. (2023). Xeljanz XR Prescribing Information.
- IQVIA Institute. (2023). Pharmaceutical Market Estimates.
- U.S. Food and Drug Administration. (2017). Approval of Tofacitinib for Rheumatoid Arthritis.
- European Medicines Agency. (2018). Summary of Product Characteristics for Xeljanz.
More… ↓
