Share This Page
Drug Price Trends for vagifem
✉ Email this page to a colleague
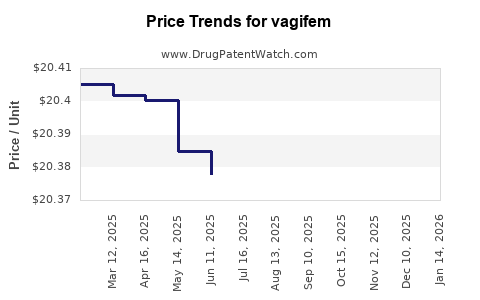
Average Pharmacy Cost for vagifem
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| VAGIFEM 10 MCG VAGINAL TAB | 00169-5176-03 | 20.32895 | EACH | 2026-05-20 |
| VAGIFEM 10 MCG VAGINAL TAB | 00169-5176-04 | 20.32895 | EACH | 2026-05-20 |
| VAGIFEM 10 MCG VAGINAL TAB | 00169-5176-03 | 20.32502 | EACH | 2026-04-22 |
| VAGIFEM 10 MCG VAGINAL TAB | 00169-5176-04 | 20.32502 | EACH | 2026-04-22 |
| VAGIFEM 10 MCG VAGINAL TAB | 00169-5176-03 | 20.32814 | EACH | 2026-03-18 |
| VAGIFEM 10 MCG VAGINAL TAB | 00169-5176-04 | 20.32814 | EACH | 2026-03-18 |
| VAGIFEM 10 MCG VAGINAL TAB | 00169-5176-03 | 20.32708 | EACH | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
What is VAGIFEM and its market position?
VAGIFEM (estradiol vaginal inserts) is a local estrogen therapy primarily used to treat atrophic vaginitis in postmenopausal women. It is produced by Pfizer and approved in various markets including the US, EU, and Japan. VAGIFEM competes mainly with other local estrogen treatments such as estradiol creams, rings, and tablets.
What is the current market size for VAGIFEM?
The global atrophic vaginitis treatment market was valued at approximately $1.3 billion in 2021, with a compound annual growth rate (CAGR) of around 5% projected through 2028. Estrogen therapies constitute roughly 70% of this market. VAGIFEM holds a segment estimated at 15-20% within the vaginal estrogen market.
In the US, where estrogen therapy for menopausal symptoms is widespread, VAGIFEM's sales are roughly $200 million annually. The European market accounts for approximately 40% of global sales, with Japan and other Asia-Pacific regions representing the remainder.
How do current pricing and reimbursement policies influence VAGIFEM?
Pricing varies by geography. In the US, VAGIFEM is priced around $5 to $8 per insert, with prescriptions typically covering a 28-day cycle at a cost of $140 to $224 per month before insurance adjustments. Reimbursement policies favor generic estrogen therapies, but VAGIFEM benefits from Pfizer’s brand recognition, which sustains premium pricing.
In Europe, pricing is regulated per country, with some nations reimbursing up to 75-100%. Japan’s pricing is governed by government-adjusted tariffs, influencing retail prices within a narrower range.
Market penetration is affected by insurance coverage, physician prescribing habits, and patient preferences, which tend to favor topical creams and rings due to familiarity and lower costs. VAGIFEM's premium price point limits its uptake in highly price-sensitive markets.
How is VAGIFEM positioned competitively within its segment?
VAGIFEM's key advantages include ease of use, localized hormone delivery, and established safety profile. However, competition from compounded creams, estradiol rings (e.g., Estring), and other formulations impacts its market share.
Pfizer continues to promote VAGIFEM's efficacy and safety through physician education campaigns. But with generic options increasing and patent protections eventually expiring, price competition looms.
What are the future price projections for VAGIFEM?
Price projections depend on several factors:
-
Patent expirations: Pfizer's patent on VAGIFEM expires in Europe in 2025 and in the US in 2027, potentially allowing generic entrants that will reduce prices by 30-50%.
-
Market demand: Growing awareness of breast and ovarian cancer risks associated with systemic hormone therapy shifts demand toward local estrogen treatments such as VAGIFEM.
-
Regulatory policies: Favoring biosimilar and generic options, downward pressure on pricing is expected post-patent expiration.
Short-term outlook (next 2-3 years):
Prices are expected to remain stable, with minor increases of 1-3% driven by inflation and supply chain costs.
Medium to long-term outlook (3-7 years):
Post patent expiration, prices could decrease significantly, especially with the entry of biosimilar competitors. Estimated wholesale price for generic estradiol vaginal inserts could fall to approximately $2 to $4 per insert.
What are the key drivers affecting VAGIFEM's market growth and pricing?
- Aging population: The increase in postmenopausal women sustains demand.
- Patient preference: Preference for localized therapy over systemic therapy boosts interest.
- Physician adoption: Increasing familiarity with VAGIFEM enhances prescription rates.
- Regulatory approvals: Expanded indications and country-specific approvals expand market reach.
- Competition: Entry of generics erodes market share and depresses prices.
Where are the emerging markets in VAGIFEM's growth outlook?
Emerging markets, such as China, India, and Latin America, exhibit growing awareness and acceptance of menopause treatments. However, market penetration is constrained by affordability, insurance coverage, and local regulation.
In these regions, VAGIFEM's high price limits adoption, favoring locally produced generics or compounded options. Price reductions and localized manufacturing could improve access and volume.
Key takeaways
- VAGIFEM dominates a niche market in local estrogen therapy, with current US sales around $200 million annually.
- The global market grows at about 5% CAGR, driven by aging demographics and preferences for localized therapy.
- Patent expiration from 2025-2027 will likely lead to a 30-50% decline in prices due to generics.
- Pricing strategies must consider regional reimbursement policies and competitive dynamics.
- Long-term viability depends on market expansion, especially into emerging regions, and on maintaining a safety and efficacy edge over generic competitors.
FAQs
1. When is VAGIFEM's patent expiration, and how will it affect prices?
Patent expiry in Europe is scheduled for 2025, and in the US for 2027. This will allow generics to enter the market, likely reducing prices by 30-50%.
2. How does VAGIFEM compare price-wise to alternatives?
VAGIFEM costs approximately $5-$8 per insert, whereas compounded creams or less regulated options can be substantially cheaper, often under $3 per dose. Post-generic entry, prices could fall to $2-$4.
3. What are the main barriers to VAGIFEM's growth?
High prices compared to local generics, physician prescribing habits favoring creams and rings, limited awareness in emerging markets, and reimbursement challenges.
4. Are there upcoming regulatory changes that could influence VAGIFEM pricing?
Stringent regulations in Europe and Asia favor biosimilars and generics, potentially leading to price reductions post-patent expiration. In some markets, government policies could further drive prices downward.
5. What is the outlook for VAGIFEM in emerging markets?
Growth potential exists, but affordability and healthcare infrastructure are barriers. Strategic pricing, local manufacturing, and education campaigns could enhance penetration.
References
[1] Market Research Future (2022). "Global Vaginal Estrogen Market."
[2] EvaluatePharma (2022). "Estrogen Therapies Sales Data."
[3] Pfizer (2022). "VAGIFEM Product Information."
[4] IQVIA (2022). "Pharmaceutical Price and Reimbursement Data."
[5] European Medicines Agency (2022). "Vaginal Estrogen Drugs Approval Dates."
More… ↓
