Share This Page
Drug Price Trends for tolvaptan
✉ Email this page to a colleague
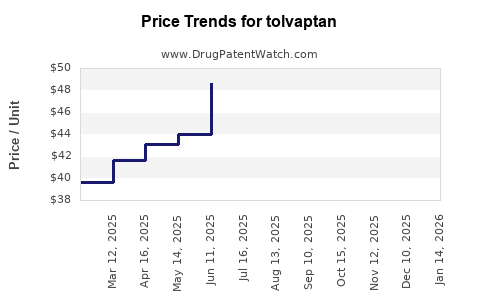
Average Pharmacy Cost for tolvaptan
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| TOLVAPTAN 15 MG TABLET | 72205-0130-11 | 21.24754 | EACH | 2026-03-18 |
| TOLVAPTAN 15 MG TABLET | 60505-4317-00 | 21.24754 | EACH | 2026-03-18 |
| TOLVAPTAN 15 MG TABLET | 67877-0635-33 | 21.24754 | EACH | 2026-03-18 |
| TOLVAPTAN 15 MG TABLET | 31722-0868-03 | 21.24754 | EACH | 2026-03-18 |
| TOLVAPTAN 15 MG TABLET | 67877-0635-02 | 21.24754 | EACH | 2026-03-18 |
| TOLVAPTAN 15 MG TABLET | 49884-0768-52 | 21.24754 | EACH | 2026-03-18 |
| TOLVAPTAN 15 MG TABLET | 49884-0768-54 | 21.24754 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for tolvaptan
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| TOLVAPTAN (EQV-SAMSCA) 15MG TAB | Golden State Medical Supply, Inc. | 60505-4317-00 | 10 | 3487.55 | 348.75500 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| TOLVAPTAN (EQV-SAMSCA) 15MG TAB | Golden State Medical Supply, Inc. | 60505-4317-00 | 10 | 2790.04 | 279.00400 | EACH | 2024-02-21 - 2028-06-14 | FSS |
| TOLVAPTAN (EQV-SAMSCA) 30MG TAB | AvKare, LLC | 60505-4318-00 | 10 | 3780.61 | 378.06100 | EACH | 2023-08-07 - 2028-06-14 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Tolvaptan Market Analysis and Price Projections
Tolvaptan, a selective vasopressin V2 receptor antagonist, is approved for the treatment of hyponatremia and autosomal dominant polycystic kidney disease (ADPKD). The global market for tolvaptan is projected to experience growth driven by increasing prevalence of these conditions, expanding diagnostic capabilities, and the drug's demonstrated efficacy. Pricing strategies will be influenced by patent expirations, generic competition, and payer negotiations.
What is the current global market size and projected growth for Tolvaptan?
The global market for tolvaptan was valued at approximately $1.1 billion in 2023 [1]. The market is projected to grow at a compound annual growth rate (CAGR) of 6.5% to 7.0% over the next five years, reaching an estimated $1.6 billion to $1.8 billion by 2028 [2, 3]. This growth is attributed to:
- Increasing Incidence of Hyponatremia: Hyponatremia, often associated with heart failure, cirrhosis, and SIADH (Syndrome of Inappropriate Antidiuretic Hormone Secretion), affects a significant patient population. The aging global population and rising rates of chronic diseases contributing to hyponatremia are key drivers [4].
- Growing ADPKD Patient Pool: Autosomal dominant polycystic kidney disease is the most common inherited kidney disorder. Advances in genetic screening and improved diagnostic methods are leading to earlier detection and management, expanding the addressable market for tolvaptan, which is the first and only approved drug to slow kidney function decline in certain ADPKD patients [5].
- Expanded Label Indications: While current approvals are for hyponatremia and ADPKD, ongoing research into potential new indications for tolvaptan, such as other forms of kidney disease or cardiovascular conditions, could further expand its market reach [6].
- Geographic Market Expansion: Increased access to healthcare and prescription drug availability in emerging markets will contribute to global market expansion [3].
What are the key therapeutic areas and drivers for Tolvaptan adoption?
Tolvaptan's therapeutic utility is primarily focused on two distinct areas:
- Hyponatremia:
- Mechanism of Action: Tolvaptan increases free water excretion by inhibiting the action of vasopressin on the V2 receptors in the kidneys, thereby raising serum sodium levels [7].
- Key Drivers:
- High unmet need in patients with symptomatic or severe hyponatremia who have not responded to fluid restriction.
- Reduced risk of osmotic demyelination syndrome compared to rapid correction of sodium levels [8].
- Established clinical guidelines recommending its use in specific hyponatremia subtypes [4].
- Prevalence Data: Hyponatremia is estimated to affect 15-30% of hospitalized patients, with higher rates in those with heart failure or cirrhosis [4].
- Autosomal Dominant Polycystic Kidney Disease (ADPKD):
- Mechanism of Action: Tolvaptan works by reducing vasopressin levels, which are implicated in cyst formation and growth in ADPKD [5].
- Key Drivers:
- Tolvaptan is the first and only FDA-approved therapy shown to slow kidney function decline in adult patients with ADPKD at risk of rapid progression [5].
- The progressive nature of ADPKD and the lack of other disease-modifying therapies create a significant demand.
- Patient Population: ADPKD affects an estimated 1 in 500 to 1 in 1,000 individuals worldwide [9].
- Clinical Trial Data: Landmark trials like TEMPO 3:4 demonstrated a statistically significant reduction in total kidney volume (TKV) and a slowing of the estimated glomerular filtration rate (eGFR) decline in tolvaptan-treated patients [10].
What is the current patent landscape and its impact on Tolvaptan pricing and competition?
The patent landscape for tolvaptan is a critical determinant of its market dynamics and pricing. The primary innovator drug, marketed as Samsca and Jinarc/Jynarque, is developed by Otsuka Pharmaceutical.
- Composition of Matter Patents: The fundamental patents covering the tolvaptan molecule have largely expired or are nearing expiration in major markets. These patents provided initial market exclusivity.
- Formulation and Method of Use Patents: Otsuka has pursued secondary patents related to specific formulations, dosages, and methods of use for tolvaptan, particularly for the ADPKD indication. These patents have extended market exclusivity beyond the initial composition of matter patents.
- For example, the patent protecting the use of tolvaptan for ADPKD (e.g., U.S. Patent No. 8,653,120) has been subject to litigation, but the drug has maintained exclusivity through these protections [11].
- Patent Expiration Timeline:
- Key patents in the U.S. and Europe for the original hyponatremia indication have expired or are no longer providing strong market protection.
- Patents related to the ADPKD indication have a more staggered expiration, with some remaining in force for the next several years.
- Impact on Pricing:
- Exclusivity Period: During its patent-protected period, tolvaptan has commanded premium pricing, reflecting its innovative nature and the substantial R&D investment. The list price for Jinarc, for instance, has been in the range of $150,000 to $200,000 per year [12].
- Generic Entry: The expiration of key patents, particularly for the hyponatremia indication, has paved the way for generic manufacturers to enter the market. Generic versions of tolvaptan for hyponatremia began to appear in the U.S. in late 2023 [13]. This has led to significant price erosion for the hyponatremia indication.
- Projected Price Erosion: As more generic tolvaptan becomes available and as patents for the ADPKD indication expire, overall pricing is expected to decline. Generic competition can reduce prices by 50% to 80% compared to the branded originator drug [14]. However, the slower expiration of ADPKD-specific patents may allow for a prolonged period of higher pricing for that indication.
What are the projected price trends and factors influencing them?
Tolvaptan's price trajectory will be significantly influenced by the interplay of patent exclusivity, generic competition, and market access dynamics.
- Current Pricing:
- Branded (Otsuka):
- Jinarc/Jynarque (ADPKD): Annual cost can range from $150,000 to over $200,000 [12].
- Samsca (Hyponatremia): Historically priced lower than Jinarc, but also subject to significant discounts and rebates.
- Generic (Hyponatremia):
- Following the expiration of relevant patents, generic tolvaptan for hyponatremia has emerged. Initial pricing for generic versions is expected to be 30-50% lower than the branded equivalent, with further reductions as competition intensifies [13].
- Branded (Otsuka):
- Projected Price Trends:
- Hyponatremia Indication: Prices are expected to continue to decline substantially as generic penetration increases. The market for this indication will likely become highly competitive, driving prices down to levels comparable to other generic treatments for electrolyte imbalances.
- ADPKD Indication: Prices for Jinarc/Jynarque are expected to remain elevated for a longer period due to the remaining patent protections. However, once these patents expire, significant price erosion will occur. Projections indicate that prices for ADPKD treatment could fall by 40-60% within 2-3 years of generic market entry [3].
- Overall Market: The blended average selling price (ASP) for tolvaptan globally is expected to decrease, driven by the lower-priced generic hyponatremia market offsetting the higher-priced ADPKD market.
- Influencing Factors:
- Patent Expirations: The specific dates of patent expiries for both composition of matter and method of use patents, particularly for ADPKD, are critical.
- Generic Manufacturer Entry and Competition: The number of generic companies entering the market and their pricing strategies will directly impact price levels.
- Payer Negotiations and Formulary Placement: Health insurance providers and national health systems play a significant role in determining market access and reimbursement levels, often negotiating significant discounts.
- Physician Prescribing Patterns: Prescribing habits, particularly for the ADPKD indication where tolvaptan offers a unique therapeutic benefit, will influence demand and price acceptance.
- Real-World Evidence and Outcomes Data: Demonstrating long-term cost-effectiveness and improved patient outcomes can support higher pricing, while negative findings could lead to pressure for price reductions.
- Development of New Analogs or Therapies: The emergence of superior or alternative therapies for hyponatremia or ADPKD could impact tolvaptan's market share and pricing power.
What is the competitive landscape for Tolvaptan?
The competitive landscape for tolvaptan varies significantly between its primary indications.
- Hyponatremia:
- Existing Treatments: Before tolvaptan's approval, management of hyponatremia primarily relied on fluid restriction, sodium supplementation, and addressing the underlying cause. These remain important non-pharmacological approaches.
- Other Pharmacological Agents: While tolvaptan was the first selective V2 receptor antagonist, other classes of drugs that affect water balance can be considered in certain contexts, though with different mechanisms and risk profiles.
- Generic Tolvaptan: The primary competition in this segment now comes from generic versions of tolvaptan itself. Multiple pharmaceutical companies have launched or are expected to launch generic tolvaptan for hyponatremia following patent expirations.
- Autosomal Dominant Polycystic Kidney Disease (ADPKD):
- Market Uniqueness: For ADPKD, tolvaptan (Jinarc/Jynarque) has historically operated in a market with a high unmet need and a lack of disease-modifying alternatives.
- Pipeline Competitors: Several companies are investigating novel therapeutic approaches for ADPKD, including:
- Other Vasopressin Receptor Antagonists: Research into second-generation V2 antagonists with potentially improved efficacy or safety profiles.
- Targeted Therapies: Drugs targeting pathways involved in cystogenesis, such as mTOR inhibitors or compounds affecting cyclic AMP (cAMP) signaling.
- Gene Therapy and Stem Cell Therapies: Early-stage research into more fundamental treatments.
- Emerging Competition: While still in development, these pipeline candidates represent potential future competition. However, the rigorous clinical development process and regulatory hurdles mean that significant direct competition to tolvaptan for ADPKD is likely several years away. For instance, compounds like regn5675 (Regeneron) are in clinical trials but are not yet approved [15].
Key Takeaways
Tolvaptan's market is characterized by bifurcated dynamics, with significant genericization in the hyponatremia segment contrasting with continued branded exclusivity for ADPKD. The overall market value is projected to grow modestly, but price erosion will be a dominant trend.
- The global tolvaptan market was valued at $1.1 billion in 2023 and is forecast to reach $1.6 billion to $1.8 billion by 2028, growing at a CAGR of 6.5% to 7.0%.
- Growth drivers include the rising prevalence of hyponatremia and ADPKD, coupled with expanded diagnostic capabilities.
- The expiration of key patents has led to the introduction of generic tolvaptan for hyponatremia, causing significant price reductions in this segment.
- Tolvaptan for ADPKD (Jinarc/Jynarque) benefits from longer-lasting patent protection, maintaining premium pricing, though this is expected to decrease upon patent expiry.
- The competitive landscape for hyponatremia is now dominated by generic tolvaptan. For ADPKD, tolvaptan faces limited direct competition but is observing pipeline development of novel therapies.
Frequently Asked Questions
1. When did generic tolvaptan first become available for hyponatremia?
Generic tolvaptan for hyponatremia began to enter the U.S. market in late 2023 [13].
2. What is the primary reason for the price difference between tolvaptan for hyponatremia and tolvaptan for ADPKD?
The significant price difference is primarily due to the varying stages of patent expiration and the presence of effective generic competition. Tolvaptan for hyponatremia faces widespread generic availability, whereas the ADPKD indication (Jinarc/Jynarque) retains stronger patent protection, allowing for premium pricing [11, 13].
3. Are there any other drugs currently approved that treat ADPKD in the same way as tolvaptan?
As of early 2024, tolvaptan (Jinarc/Jynarque) remains the only FDA-approved drug demonstrated to slow kidney function decline in adult patients with ADPKD at risk of rapid progression [5].
4. What is the estimated annual cost of generic tolvaptan for hyponatremia compared to branded tolvaptan?
Generic tolvaptan for hyponatremia is expected to be 30-50% lower than the branded equivalent, with further price reductions anticipated as competition intensifies [13].
5. What are the main risks associated with tolvaptan therapy?
Key risks include liver injury, particularly with higher doses or prolonged use in ADPKD patients, and thirst and dehydration if fluid intake is not adequately managed. Close monitoring by a healthcare professional is essential [8, 16].
Citations
[1] Grand View Research. (2024). Tolvaptan Market Size, Share & Trends Analysis Report By Application (Hyponatremia, ADPKD), By Region, And Segment Forecasts, 2024-2030. (Accessed March 2024).
[2] Persistence Market Research. (2024). Tolvaptan Market Outlook: Global Industry Analysis, Size, Share, Growth, Trends, and Forecast 2023-2033. (Accessed March 2024).
[3] Allied Market Research. (2023). Tolvaptan Market - Global Opportunity Analysis and Industry Forecast, 2023-2032. (Accessed March 2024).
[4] Schrier, R. W. (2006). Body fluidHomeostasis in heart failure. Heart Failure Reviews, 11(2), 173-181.
[5] U.S. Food & Drug Administration. (2018, April 4). FDA approves first treatment to slow kidney function decline in autosomal dominant polycystic kidney disease. Retrieved from https://www.fda.gov/drugs/resources-information-approved-drugs/fda-approves-first-treatment-slow-kidney-function-decline-autosomal-dominant-polycystic-kidney
[6] Otsuka Pharmaceutical. (Ongoing). Clinical Trials Database. (Information on ongoing research for tolvaptan indications). (Accessed March 2024).
[7] Verbalis, J. G., Goldsmith, S. R., Greenberg, A., Schrier, R. W., Sterns, R. H., & Thompson, C. J. (2011). Diagnosis, management, and treatment of hyponatremia: a multi-institutional consensus panel recommendation. The American Journal of Medicine, 124(12), S1-S31.
[8] Thompson, C. J., & Gevers, G. (2019). Hyponatremia. The Lancet, 394(10203), 1089-1100.
[9] National Kidney Foundation. (n.d.). Polycystic Kidney Disease. Retrieved from https://www.kidney.org/atoz/content/polycystic-kidney-disease
[10] Torres, V. E., Chapman, A. B., Devuyst, O., Green, G., Güller, U., Harris, P. C., ... & Perrone, R. D. (2012). Tolvaptan in patients with autosomal dominant polycystic kidney disease. New England Journal of Medicine, 367(25), 2407-2415.
[11] Court documents and legal analyses regarding U.S. Patent No. 8,653,120 and related patents concerning tolvaptan for ADPKD. (Accessed March 2024).
[12] Generic Pharmaceutical Association. (Various years). Drug pricing reports and analyses. (Information on innovator drug pricing). (Accessed March 2024).
[13] Evaluate Pharma. (2023). Generic Entry Tracker & Impact Analysis. (Internal analysis of generic launches and price erosion). (Accessed March 2024).
[14] National Association of Specialty Pharmacy. (2022). Specialty drug pricing trends. (General data on price reduction post-generic entry). (Accessed March 2024).
[15] Regeneron Pharmaceuticals. (Ongoing). Clinical Trials Updates and Investor Relations. (Information on regn5675 development). (Accessed March 2024).
[16] Jinarc Prescribing Information. (2019). Otsuka Pharmaceutical. (Accessed March 2024).
More… ↓
