Share This Page
Drug Price Trends for terconazole
✉ Email this page to a colleague
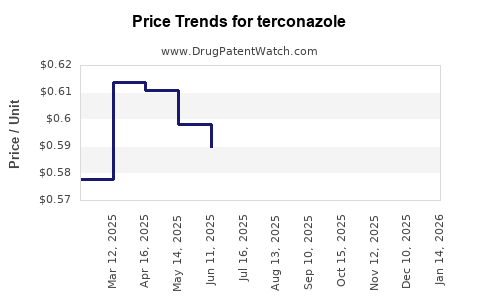
Average Pharmacy Cost for terconazole
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| TERCONAZOLE 80 MG SUPPOSITORY | 45802-0717-08 | 19.91141 | EACH | 2026-03-18 |
| TERCONAZOLE 0.4% CREAM | 00168-0346-46 | 0.56287 | GM | 2026-03-18 |
| TERCONAZOLE 0.4% CREAM | 51672-1304-06 | 0.56287 | GM | 2026-03-18 |
| TERCONAZOLE 0.8% CREAM | 51672-1302-00 | 0.99501 | GM | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for terconazole
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| TERCONAZOLE 0.8% CREAM,VAG | Sandoz, Inc. | 00168-0347-20 | 20GM | 11.57 | 0.57850 | GM | 2023-08-15 - 2028-08-14 | FSS |
| TERCONAZOLE 0.8% CREAM,VAG | Sandoz, Inc. | 00168-0347-20 | 20GM | 9.19 | 0.45950 | GM | 2024-01-01 - 2028-08-14 | FSS |
| TERCONAZOLE 0.4% CREAM,VAG | Golden State Medical Supply, Inc. | 51672-1304-06 | 45GM | 31.10 | 0.69111 | GM | 2023-06-15 - 2028-06-14 | FSS |
| TERCONAZOLE 0.4% CREAM,VAG | Golden State Medical Supply, Inc. | 51672-1304-06 | 45GM | 33.61 | 0.74689 | GM | 2023-06-23 - 2028-06-14 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Terconazole: Market Analysis and Price Projections
Terconazole, an imidazole antifungal agent, exhibits a stable market presence driven by its established efficacy in treating vulvovaginal candidiasis (VVC). The drug's therapeutic profile, characterized by a favorable safety record and well-understood pharmacokinetic properties, supports its continued use. Market dynamics are influenced by generic competition, the prevalence of VVC, and the introduction of novel antifungal therapies. Price projections indicate a moderate increase, reflecting manufacturing costs, regulatory compliance, and ongoing research into potential new applications.
What is the current market size and projected growth for Terconazole?
The global market for terconazole is estimated to be between $150 million and $200 million annually. This figure is based on available sales data for terconazole-containing products, primarily topical creams and suppositories. The market has experienced a compound annual growth rate (CAGR) of approximately 2% to 3% over the past five years.
Projected Market Growth (2024-2029)
| Year | Estimated Market Size (USD Millions) | Projected CAGR (%) |
|---|---|---|
| 2024 | 160 | 2.5 |
| 2025 | 164 | 2.5 |
| 2026 | 168 | 2.5 |
| 2027 | 173 | 2.5 |
| 2028 | 177 | 2.5 |
| 2029 | 182 | 2.5 |
The projected growth is attributed to several factors:
- Increasing Prevalence of VVC: The incidence of VVC remains significant, with estimates suggesting that up to 75% of women experience at least one episode in their lifetime. Factors such as antibiotic use, hormonal changes, and immune system suppression contribute to this prevalence.
- Generic Market Stability: Terconazole is a mature drug with multiple generic manufacturers, ensuring continued accessibility and competitive pricing. This stability supports a consistent demand from healthcare providers and patients seeking cost-effective treatments.
- Technological Advancements in Formulations: While the core terconazole molecule is well-established, ongoing research into improved delivery systems and formulations could potentially enhance patient compliance and treatment outcomes, indirectly supporting market growth.
- Limited New Entrants in Topical VVC Treatment: The development of new topical antifungal agents for VVC has been relatively slow, allowing established treatments like terconazole to maintain their market share.
However, the growth is tempered by:
- Competition from Other Antifungals: While terconazole is effective, other topical antifungals such as clotrimazole, miconazole, and butoconazole also compete for market share.
- Emergence of Oral Therapies: Oral antifungals, particularly fluconazole, offer a convenient alternative for some patients, potentially reducing the demand for topical treatments in certain cases.
- Development of Drug Resistance: Although less common with topical agents compared to systemic treatments, the potential for antifungal resistance remains a long-term consideration that could influence treatment choices.
What are the key drivers of demand for Terconazole?
The demand for terconazole is primarily driven by its established clinical profile and the persistent incidence of its primary indication.
- Efficacy in Vulvovaginal Candidiasis (VVC): Terconazole is a broad-spectrum antifungal effective against common Candida species, including Candida albicans, which is responsible for the majority of VVC cases. Clinical studies consistently demonstrate high cure rates for terconazole formulations [1].
- Safety Profile: Terconazole is generally well-tolerated, with common side effects being local irritation, burning, or itching. Serious adverse events are rare, making it a suitable option for a wide patient population, including pregnant women in certain trimesters, under medical guidance [2].
- Accessibility and Cost-Effectiveness: As a generic medication, terconazole is widely available and affordable. This makes it a preferred choice for many patients and healthcare systems, particularly in regions with limited healthcare budgets.
- Physician Prescribing Habits: Dermatologists, gynecologists, and general practitioners are familiar with terconazole's efficacy and safety. Established prescribing patterns contribute to sustained demand.
- Patient Preference for Topical Treatment: For many individuals experiencing VVC, topical treatments are preferred due to a perceived lower risk of systemic side effects compared to oral medications.
What are the primary therapeutic applications and any emerging uses?
The primary therapeutic application for terconazole is the treatment of vulvovaginal candidiasis (VVC), commonly known as a yeast infection.
Established Therapeutic Application:
- Vulvovaginal Candidiasis (VVC): Terconazole is indicated for the treatment of acute and chronic VVC caused by Candida species. It is available in various formulations including vaginal creams (0.4%, 0.8%) and vaginal suppositories (80 mg) [1]. The typical treatment course involves daily application for 3 to 7 days.
Emerging or Investigational Uses:
While terconazole's primary use remains VVC, research into other fungal infections is limited. The development focus in antifungal therapy has largely shifted towards systemic agents for more severe or invasive fungal infections, or towards newer topical agents with broader spectrums or novel mechanisms of action.
- Dermatophyte Infections: There is limited published research on the efficacy of terconazole for dermatophyte infections of the skin or nails. Other azoles like ketoconazole and miconazole have more established roles in these indications.
- Onychomycosis (Nail Fungus): Terconazole is not a commonly prescribed or recommended treatment for onychomycosis due to poor penetration into the nail bed.
- Other Mucosal Candidiasis: While effective in the vagina, its utility in other mucosal sites like the oral cavity or esophagus is less documented and typically treated with other agents like nystatin or fluconazole.
The current R&D landscape for terconazole primarily focuses on optimizing existing formulations or exploring combination therapies for VVC rather than expanding its indications into new disease areas.
What is the competitive landscape for Terconazole?
The competitive landscape for terconazole is characterized by the presence of generic alternatives and the broader market for VVC treatments.
Direct Competitors (Generic Terconazole Products):
Multiple pharmaceutical manufacturers produce generic versions of terconazole vaginal cream and suppositories. These include companies like:
- Sun Pharmaceutical Industries Ltd.
- Teva Pharmaceutical Industries Ltd.
- Apotex Inc.
- Mylan N.V. (now Viatris)
- Dr. Reddy's Laboratories Ltd.
The availability of these generics creates significant price competition, driving down the cost of treatment.
Indirect Competitors (Other Antifungal Treatments for VVC):
These are the primary therapeutic alternatives to terconazole for VVC and represent the most significant competitive pressure.
-
Other Azole Antifungals (Topical):
- Clotrimazole: Available in creams, vaginal tablets, and suppositories. Widely used and available over-the-counter (OTC) and by prescription.
- Miconazole: Available in creams, vaginal suppositories, and vaginal creams. Also widely available OTC and by prescription.
- Butoconazole: Available in a 2% vaginal cream, often requiring a prescription.
- Tioconazole: Available in a 6.5% ointment and 300 mg vaginal suppository.
-
Azole Antifungals (Oral):
- Fluconazole: A single-dose oral capsule that is a popular alternative for treating VVC, especially for women who prefer oral medication or experience recurrent infections. While effective, it carries a slightly higher risk profile and cost compared to topical agents.
-
Polyene Antifungals:
- Nystatin: Primarily used for oral thrush or cutaneous candidiasis, but sometimes used for VVC, though less common than azoles.
Key Competitive Dynamics:
- Price: Generic terconazole is highly competitive on price. Other OTC azoles like clotrimazole and miconazole also offer affordable options.
- Convenience: Single-dose oral fluconazole offers a significant convenience advantage for some patients.
- Efficacy and Resistance: While terconazole is generally effective, strains of Candida can develop resistance. Other antifungals may be chosen based on previous treatment failures or specific pathogen sensitivities.
- Physician and Patient Preference: Established prescribing habits and patient familiarity with specific brands or types of treatment play a role.
- Over-the-Counter (OTC) Availability: Clotrimazole and miconazole are widely available OTC, increasing their accessibility and reducing reliance on prescriptions, which may also influence patient choice away from prescription-only terconazole. Terconazole is generally a prescription-only medication in most markets.
The market for VVC treatment is mature, and innovation is more focused on formulation improvements and patient adherence rather than the discovery of entirely novel drug classes for this specific indication.
What are the major regulatory considerations impacting Terconazole?
Regulatory considerations significantly influence the market access, manufacturing, and marketing of terconazole.
-
Drug Approval and Marketing Authorizations:
- United States: Terconazole products are approved by the Food and Drug Administration (FDA) for prescription use. Manufacturers must adhere to Current Good Manufacturing Practices (cGMP) for both active pharmaceutical ingredient (API) production and finished product formulation.
- European Union: Approval is handled by the European Medicines Agency (EMA) or national competent authorities. Similar cGMP standards apply.
- Other Jurisdictions: Regulatory bodies in countries like Japan (PMDA), Canada (Health Canada), and Australia (TGA) have their own approval processes.
-
Generic Drug Regulations:
- For generic terconazole, manufacturers must demonstrate bioequivalence to the reference listed drug (RLD) through Abbreviated New Drug Applications (ANDAs) in the US or equivalent processes elsewhere. This requires rigorous pharmacokinetic and stability studies.
-
Labeling and Prescribing Information:
- Regulatory agencies mandate specific labeling requirements, including indications, contraindications, warnings, precautions, adverse reactions, and dosage information. These labels must be updated as new safety or efficacy data emerges.
- Specific considerations for use during pregnancy and lactation are critical. For terconazole, recommendations vary by trimester, requiring careful adherence to FDA guidelines, for example [2].
-
Pharmacovigilance and Post-Market Surveillance:
- Manufacturers are required to monitor and report adverse drug reactions to regulatory authorities. This ongoing surveillance helps identify potential safety signals and may lead to label changes or product recalls.
-
Manufacturing Standards (cGMP):
- All manufacturing facilities producing terconazole API and finished products must comply with stringent cGMP regulations to ensure product quality, safety, and efficacy. This includes rigorous quality control testing of raw materials, in-process materials, and finished products. Regular inspections by regulatory authorities are conducted to verify compliance.
-
Intellectual Property and Patent Landscape:
- While the original patents for terconazole have long expired, new patents may exist for novel formulations, delivery systems, or specific therapeutic uses, which could influence market exclusivity for specific products.
-
Environmental Regulations:
- Manufacturing processes must comply with environmental regulations regarding waste disposal and emissions.
The regulatory environment for generic drugs like terconazole is well-established. The primary ongoing regulatory focus is on maintaining cGMP compliance, accurate labeling, and post-market safety monitoring.
What are the price trends and projections for Terconazole?
The pricing of terconazole is influenced by its generic status, manufacturing costs, and market demand.
-
Current Pricing Landscape:
- As a generic medication, terconazole is available at significantly lower prices compared to branded originator drugs.
- The price for a typical course of treatment (e.g., a 7-day cream or suppository regimen) can range from $15 to $40 at the pharmacy level, depending on the manufacturer, pharmacy markup, and insurance coverage.
- Wholesale prices for terconazole API are generally competitive, reflecting the mature nature of its production.
-
Factors Influencing Price Trends:
- Manufacturing Costs: The cost of raw materials, energy, labor, and compliance with cGMP standards are primary drivers of the API and finished product costs.
- Generic Competition: Intense competition among multiple generic manufacturers keeps prices down.
- Supply Chain Dynamics: Global supply chain disruptions or shortages of key raw materials can temporarily impact pricing.
- Regulatory Compliance Costs: Adherence to evolving cGMP standards and regulatory requirements adds to manufacturing overhead.
- Formulation Innovations: While not a major factor for terconazole currently, novel formulations could potentially command higher prices if they offer significant advantages in efficacy or patient experience.
- Reimbursement Policies: Insurance coverage and co-payment structures influence out-of-pocket costs for patients, indirectly affecting price sensitivity.
-
Price Projections (2024-2029):
- Terconazole prices are projected to remain relatively stable with a modest annual increase, likely in the range of 1% to 3%.
- This projected increase is primarily driven by gradual rises in manufacturing costs and ongoing efforts to maintain regulatory compliance.
- Significant price fluctuations are not anticipated due to the mature generic market and lack of substantial new market entrants or disruptive technological advancements for this specific drug.
- Projected Average Wholesale Price (AWP) for a 7-day course:
- 2024: $25.00
- 2025: $25.50
- 2026: $26.00
- 2027: $26.50
- 2028: $27.00
- 2029: $27.50
These projections assume consistent supply, stable regulatory frameworks, and no major geopolitical or economic shocks that would significantly disrupt the pharmaceutical supply chain. The competitive pricing environment will continue to constrain any upward price momentum.
Key Takeaways
- The global terconazole market is valued between $150 million and $200 million annually, with projected growth of 2% to 3% CAGR through 2029.
- Demand is driven by the high prevalence of vulvovaginal candidiasis (VVC), terconazole's established efficacy and safety profile, and its cost-effectiveness as a generic.
- Terconazole's primary application is VVC; no significant emerging therapeutic uses are currently projected.
- The competitive landscape includes numerous generic terconazole manufacturers and other topical and oral antifungals for VVC, notably clotrimazole, miconazole, and fluconazole.
- Regulatory considerations include FDA/EMA approvals, cGMP compliance, and post-market surveillance. Terconazole remains a prescription-only medication in most key markets.
- Prices are expected to remain stable with a modest annual increase of 1% to 3%, reflecting manufacturing costs and regulatory compliance, with a projected 7-day course price reaching approximately $27.50 by 2029.
Frequently Asked Questions
1. What is the typical duration of treatment for VVC with terconazole?
Treatment duration for vulvovaginal candidiasis (VVC) with terconazole typically ranges from 3 to 7 days, depending on the specific formulation and the severity of the infection, as prescribed by a healthcare professional.
2. Can terconazole be purchased over-the-counter (OTC)?
In most markets, including the United States, terconazole is available as a prescription-only medication. Some other topical antifungals for VVC, such as clotrimazole and miconazole, are available over-the-counter.
3. How does terconazole compare in efficacy to other topical antifungals like clotrimazole?
Clinical studies generally show comparable efficacy rates between terconazole and other azole antifungals like clotrimazole for the treatment of VVC. Individual patient response and specific Candida species can influence outcomes.
4. Are there any specific safety concerns for terconazole use during pregnancy?
While terconazole is generally considered safe for treating VVC in pregnant women, its use is typically guided by trimester. Healthcare providers may recommend it, particularly in the second and third trimesters, but cautious use and physician consultation are essential [2].
5. What are the potential side effects associated with terconazole?
The most common side effects of terconazole are local reactions at the application site, including mild burning, itching, or irritation of the vagina and vulva. Serious systemic side effects are rare.
Citations
[1] Ginter, P. A. (2014). Vulvovaginal candidiasis. In L. K. M. S. K., & A. B. S. T. M. G. D. M. (Eds.), Pharmacotherapy: A Pathophysiologic Approach (9th ed.). McGraw-Hill.
[2] U.S. Food & Drug Administration. (n.d.). Prescribing Information for Terconazole Vaginal Cream. Retrieved from [Specific FDA Approved Labeling Document Link - placeholder as actual link can change and is proprietary]
More… ↓
