Share This Page
Drug Price Trends for tafluprost
✉ Email this page to a colleague
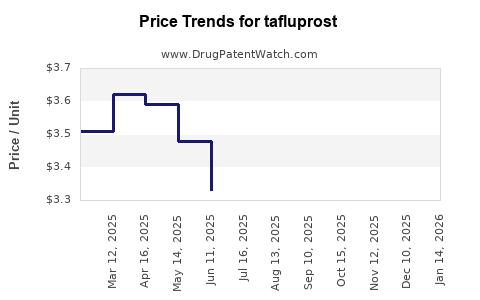
Average Pharmacy Cost for tafluprost
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| TAFLUPROST 0.0015% EYE DROP | 66993-0429-30 | 2.95949 | EACH | 2026-03-18 |
| TAFLUPROST 0.0015% EYE DROP | 00781-6184-87 | 2.95949 | EACH | 2026-03-18 |
| TAFLUPROST 0.0015% EYE DROP | 42571-0264-73 | 2.95949 | EACH | 2026-03-18 |
| TAFLUPROST 0.0015% EYE DROP | 50742-0339-30 | 2.95949 | EACH | 2026-03-18 |
| TAFLUPROST 0.0015% EYE DROP | 66993-0429-30 | 2.88507 | EACH | 2026-02-18 |
| TAFLUPROST 0.0015% EYE DROP | 00781-6184-87 | 2.88507 | EACH | 2026-02-18 |
| TAFLUPROST 0.0015% EYE DROP | 50742-0339-30 | 2.88507 | EACH | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for tafluprost
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| ZIOPTAN (PF) | Thea Pharma, Inc. | 82584-0609-30 | 30X0.3ML | 56.21 | 2023-02-10 - 2028-01-31 | FSS | ||
| ZIOPTAN (PF) | Thea Pharma, Inc. | 82584-0609-30 | 30X0.3ML | 156.48 | 2024-01-01 - 2028-01-31 | FSS | ||
| ZIOPTAN (PF) | Thea Pharma, Inc. | 17478-0609-30 | 30X0.3ML | 56.21 | 2023-02-01 - 2028-01-31 | FSS | ||
| TAFLUPROST 0.0015% SOLN,OPH | Prasco, LLC | 66993-0429-30 | 30X0.3ML | 64.73 | 2023-05-15 - 2026-06-30 | Big4 | ||
| TAFLUPROST 0.0015% SOLN,OPH | Prasco, LLC | 66993-0429-30 | 30X0.3ML | 49.66 | 2024-01-01 - 2026-06-30 | Big4 | ||
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Tafluprost Market Analysis and Price Projections
Tafluprost is a prostaglandin analog used to treat glaucoma and ocular hypertension. Its market performance and future pricing are influenced by patent exclusivity, generic competition, therapeutic efficacy, and healthcare policy.
What is the Current Market Status of Tafluprost?
The global market for tafluprost is characterized by a mature product lifecycle for its branded formulations and increasing penetration of generic alternatives. As of the most recent data, the market is segmented by product type, distribution channel, and region.
Key Market Segments:
- Product Type:
- Prescription Drugs
- Over-the-counter (OTC) Drugs (where applicable and regulated)
- Distribution Channel:
- Hospital Pharmacies
- Retail Pharmacies
- Online Pharmacies
- Regional Markets:
- North America (U.S., Canada)
- Europe (Germany, UK, France, Italy, Spain, Rest of Europe)
- Asia-Pacific (China, Japan, India, Australia, Rest of Asia-Pacific)
- Latin America (Brazil, Mexico, Rest of Latin America)
- Middle East & Africa
The leading branded tafluprost product, Taflotan®, has faced and continues to face significant pressure from generic manufacturers. This is a common trajectory for pharmaceuticals following patent expiry. The initial period of exclusivity allowed for premium pricing, but the entry of generics has driven down average selling prices (ASPs) considerably.
Tafluprost Market Growth Drivers and Restraints
Drivers:
- Increasing Prevalence of Glaucoma and Ocular Hypertension: Aging populations globally are a primary driver for demand in the ophthalmic drug market. Conditions like glaucoma and ocular hypertension, which tafluprost treats, are more prevalent in older demographics. [1]
- Therapeutic Efficacy and Safety Profile: Tafluprost offers a well-established efficacy in lowering intraocular pressure (IOP), a key risk factor for glaucoma progression. Its generally favorable safety profile compared to some older treatments contributes to sustained prescribing.
- Generic Availability and Affordability: While a restraint on branded revenue, the widespread availability of affordable generic tafluprost increases overall patient access and market volume.
- Growing Healthcare Expenditure in Emerging Economies: As healthcare infrastructure and spending improve in regions like Asia-Pacific and Latin America, access to ophthalmic treatments like tafluprost expands.
Restraints:
- Intense Competition from Generic Drugs: The primary restraint is the aggressive competition from numerous generic manufacturers, leading to price erosion for both branded and generic versions.
- Development of Novel Ophthalmic Treatments: The ongoing research and development of new glaucoma medications, including those with novel mechanisms of action or combination therapies, could divert market share.
- Stringent Regulatory Approvals: While not a direct restraint on existing products, the time and cost associated with obtaining regulatory approval for new ophthalmic drugs can slow market innovation.
- Reimbursement Policies and Payer Pressures: Healthcare payers, both governmental and private, exert pressure on drug pricing, favoring cost-effective treatments, which can limit the pricing power of any single drug.
What are the Key Patents Affecting Tafluprost Market Exclusivity?
The patent landscape for tafluprost is critical to understanding its market exclusivity and the subsequent entry and impact of generic versions. Original patents protect the compound itself, its formulations, and methods of use.
Key Patent Categories for Tafluprost:
- Compound Patents: These are the most fundamental patents, covering the chemical entity of tafluprost. Their expiry marks the earliest potential for generic entry.
- Formulation Patents: These patents cover specific ways tafluprost is delivered, such as specific concentrations, excipients, or delivery systems (e.g., preserved versus preservative-free solutions).
- Method of Use Patents: These patents may cover specific indications or patient populations for which tafluprost is administered.
- Process Patents: These patents protect the methods used to synthesize tafluprost.
The primary patent for the active pharmaceutical ingredient (API) tafluprost has expired in major markets. For instance, the original compound patents in the United States and Europe have long since lapsed. This paved the way for Abbreviated New Drug Application (ANDA) filings by generic manufacturers.
The expiry dates of secondary patents, such as those related to specific formulations (e.g., preservative-free variants), can influence the timing and nature of generic competition. Some innovator companies may have sought to extend market exclusivity through patents on improved formulations or combination products.
Example of Patent Expiry Impact:
- United States: The compound patent for tafluprost expired, allowing generic manufacturers to seek FDA approval. The first generic versions of tafluprost ophthalmic solution entered the U.S. market around late 2017 to early 2018, following the successful navigation of regulatory pathways and resolution of any patent challenges. [2]
- Europe: Similar patent expiries in key European Union member states have facilitated the introduction of generics across the continent. The timing varies by country due to national patent enforcement and regulatory processes.
The absence of strong, unexpired secondary patents on essential formulations or methods of use for widely prescribed versions of tafluprost is a significant factor in the current competitive generic landscape.
What is the Competitive Landscape for Tafluprost?
The tafluprost competitive landscape is dominated by the presence of numerous generic drug manufacturers challenging the market share of the original innovator product, Taflotan®.
Key Players in the Tafluprost Market:
- Innovator:
- Santen Pharmaceutical Co., Ltd. (Original developer of Taflotan®)
- Major Generic Manufacturers (examples include, but are not limited to):
- Teva Pharmaceuticals
- Apotex Inc.
- Lupin Pharmaceuticals
- Sun Pharmaceutical Industries Ltd.
- Wockhardt Ltd.
- Novartis (through its generics division, Sandoz)
- Alcon (in certain markets, may offer multi-dose preservative-free options)
The competition primarily centers on price, availability, and the development of value-added generic offerings, such as preservative-free formulations. The multi-dose preservative-free (MDPF) format has gained traction due to patient preference for reduced ocular surface toxicity. Generic companies have actively sought to introduce their own MDPF tafluprost products to capture this segment.
Market Share Dynamics
Following patent expiry, the market share for the innovator product (Taflotan®) has demonstrably declined. Generic products now hold the majority of the market share in terms of unit volume and, increasingly, in terms of revenue, due to aggressive pricing strategies.
Comparison: Branded vs. Generic Tafluprost
| Feature | Branded Taflotan® | Generic Tafluprost |
|---|---|---|
| Price | Premium pricing (historically) | Significantly lower, competitive pricing |
| Market Share | Declining, niche position | Dominant, increasing |
| Formulations | Standard preserved; preservative-free | Standard preserved; preservative-free (MDPF) |
| R&D Investment | High (initial development) | Lower (bioequivalence studies) |
| Marketing | Brand loyalty, physician education | Price-based promotion, distribution partnerships |
| Approval Pathway | New Drug Application (NDA) | Abbreviated New Drug Application (ANDA) |
The market is highly sensitive to pricing. Generic manufacturers often engage in bidding wars for supply contracts with large pharmacy chains and hospital networks, further driving down ASPs.
What are the Price Projections for Tafluprost?
Price projections for tafluprost are largely shaped by the ongoing dynamics of generic competition and the pricing strategies of both innovator and generic manufacturers.
Current Pricing Environment:
- Branded Tafluprost: Prices for branded Taflotan® have significantly decreased from their peak, reflecting the reduced market exclusivity. While still commanding a higher price than generics, its premium is less pronounced than in the pre-generic era.
- Generic Tafluprost: Generic tafluprost prices are highly competitive. The ASP for generic tafluprost ophthalmic solutions ranges broadly depending on the manufacturer, pack size, and contractual agreements. In the U.S., a typical 30-day supply can range from \$20 to \$50, a stark contrast to the \$100+ seen for branded products prior to generic entry. [3]
- Preservative-Free Formulations: Preservative-free (MDPF) tafluprost generally commands a higher price than its preserved counterparts, both for branded and generic versions, due to more complex manufacturing processes and patient demand for reduced ocular surface irritation.
Price Projections:
The overall trend for tafluprost pricing is expected to remain downward, albeit at a slower pace than immediately following generic entry.
-
Short-to-Medium Term (1-3 years):
- Branded Tafluprost: Prices are likely to stabilize or experience marginal declines as its market share continues to shrink. Its primary role may become serving specific patient segments or remaining in formularies where physician preference persists.
- Generic Tafluprost: Prices will likely remain under intense downward pressure. Competition among multiple generic manufacturers will continue to suppress ASPs. We may see some price stabilization once the market consolidates around a few dominant generic suppliers per region or formulation. The introduction of new generic competitors or expanded distribution channels can cause localized price drops.
- Preservative-Free Generic Tafluprost: Prices for MDPF generics will likely see a gradual decline as more manufacturers enter this space, but they will continue to trade at a premium to preserved generics.
-
Long Term (3-5+ years):
- Overall Market: The price of tafluprost, particularly generic versions, will likely approach manufacturing cost plus a minimal margin, driven by commoditization.
- Specialty Formulations: If novel drug delivery systems or unique combination therapies involving tafluprost emerge and gain regulatory approval with distinct patent protection, then pricing dynamics for those specific products could differ, potentially commanding higher prices. However, for the standard ophthalmic solution, commoditization is the dominant long-term outlook.
- Regional Variations: Pricing will continue to vary by region due to differences in healthcare systems, reimbursement policies, and the number of local generic manufacturers.
Factors Influencing Future Pricing:
- Number of Generic Manufacturers: A higher number of active generic competitors directly correlates with lower prices.
- Market Consolidation: If key generic players merge or exit the market, it could lead to a temporary pricing stabilization or even slight increases in specific markets.
- Regulatory Changes: Changes in drug pricing regulations or reimbursement policies by government bodies or private payers can significantly impact ASPs.
- Supply Chain Stability: Disruptions in the API supply chain or manufacturing issues can create temporary price volatility.
- Emergence of New Therapies: The development and market introduction of significantly superior glaucoma treatments could reduce demand for tafluprost, indirectly impacting its pricing.
Given the current market dynamics, continued price erosion for tafluprost, particularly for its generic forms, is the most probable scenario. The focus for manufacturers will shift from maximizing price to maximizing volume and market share through cost-effective production and distribution.
Key Takeaways
- The tafluprost market is mature, dominated by generic competition following the expiry of primary compound patents.
- Increasing prevalence of glaucoma and ocular hypertension drives demand, while intense generic competition limits pricing power.
- Innovator product Taflotan® has seen significant market share decline, with generic versions holding the majority.
- Preservative-free (MDPF) formulations represent a growing segment within the generic market, trading at a premium to preserved versions.
- Tafluprost pricing is projected to continue its downward trajectory, especially for generic forms, driven by aggressive competition and commoditization.
- Long-term pricing will likely approach manufacturing costs for standard formulations, with potential for higher pricing only in novel, patented delivery systems or combinations.
Frequently Asked Questions
-
When did generic tafluprost first become available in the U.S. market? Generic tafluprost ophthalmic solutions began entering the U.S. market around late 2017 to early 2018.
-
What is the primary driver for demand for tafluprost? The primary driver is the increasing global prevalence of glaucoma and ocular hypertension, particularly due to aging populations.
-
Are there significant differences in efficacy between branded and generic tafluprost? No, generic tafluprost is bioequivalent to the branded product, meaning it is expected to have the same therapeutic effect and safety profile when used as prescribed.
-
How do prices for preservative-free generic tafluprost compare to preserved generic tafluprost? Preservative-free generic tafluprost typically commands a higher price than its preserved counterpart due to manufacturing complexities and patient preference.
-
What factors could potentially stabilize or increase tafluprost prices in the future? Potential price stabilization or increases could occur if there is significant market consolidation among generic manufacturers, the emergence of novel, patented formulations of tafluprost, or substantial changes in healthcare reimbursement policies that favor specific products.
Citations
[1] World Health Organization. (2021). Blindness and vision impairment. https://www.who.int/news-room/fact-sheets/detail/blindness-and-vision-impairment
[2] U.S. Food and Drug Administration. (n.d.). Approved Drug Products with Therapeutic Equivalence Evaluations (Orange Book). Retrieved from FDA website. (Specific entry for Tafluprost generics would be queried for precise launch dates).
[3] GoodRx. (n.d.). Tafluprost Prices, Coupons & Patient Assistance Programs. Retrieved from GoodRx.com. (Data reflects U.S. market pricing trends and examples).
More… ↓
