Share This Page
Drug Price Trends for tadalafil
✉ Email this page to a colleague
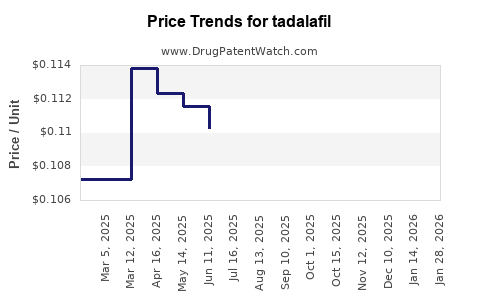
Average Pharmacy Cost for tadalafil
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| TADALAFIL 20 MG TABLET | 82009-0080-60 | 0.14778 | EACH | 2026-05-20 |
| TADALAFIL 20 MG TABLET | 82009-0079-05 | 0.17959 | EACH | 2026-05-20 |
| TADALAFIL 5 MG TABLET | 82009-0077-05 | 0.10525 | EACH | 2026-05-20 |
| TADALAFIL 20 MG TABLET | 75834-0250-01 | 0.17959 | EACH | 2026-05-20 |
| TADALAFIL 10 MG TABLET | 75834-0249-01 | 0.16705 | EACH | 2026-05-20 |
| TADALAFIL 5 MG TABLET | 75834-0248-01 | 0.10525 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for tadalafil
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| TADALAFIL 2.5MG TAB | Prasco, LLC | 66993-0700-30 | 30 | 147.74 | 4.92467 | EACH | 2021-07-01 - 2026-06-30 | FSS |
| TADALAFIL 5MG TAB | Prasco, LLC | 66993-0701-30 | 30 | 10.83 | 0.36100 | EACH | 2021-07-01 - 2026-06-30 | Big4 |
| TADALAFIL 5MG TAB | Prasco, LLC | 66993-0701-30 | 30 | 147.74 | 4.92467 | EACH | 2021-07-01 - 2026-06-30 | FSS |
| TADALAFIL 5MG TAB | Prasco, LLC | 66993-0701-38 | 30 | 20.61 | 0.68700 | EACH | 2021-07-01 - 2026-06-30 | Big4 |
| TADALAFIL 5MG TAB | Prasco, LLC | 66993-0701-38 | 30 | 147.74 | 4.92467 | EACH | 2021-07-01 - 2026-06-30 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Tadalafil Market Analysis and Price Projections
Tadalafil, a phosphodiesterase type 5 (PDE5) inhibitor, is a pharmaceutical compound with established therapeutic applications and a dynamic market landscape. Primarily recognized for its efficacy in treating erectile dysfunction (ED) and benign prostatic hyperplasia (BPH), tadalafil also holds significant potential in pulmonary arterial hypertension (PAH). This analysis examines current market drivers, competitive dynamics, patent expirations, and forecasts future price trends for tadalafil.
What is the Current Market Landscape for Tadalafil?
The global tadalafil market is substantial and projected for continued growth. The primary driver for its demand is the increasing prevalence of ED, particularly among aging populations. Factors contributing to this include lifestyle changes, rising rates of chronic diseases such as diabetes and cardiovascular disease, and increased awareness and de-stigmatization of ED. The market for BPH treatment also contributes significantly to tadalafil's commercial performance.
The tadalafil market is characterized by the presence of both branded and generic manufacturers. Eli Lilly and Company's Cialis® was the originator brand, but patent expiries have opened the market to numerous generic competitors. This has led to increased price competition and greater accessibility for patients.
Key market segments include:
- Therapeutic Area: Erectile Dysfunction, Benign Prostatic Hyperplasia, Pulmonary Arterial Hypertension.
- Formulation: Oral tablets are the dominant formulation, available in various dosages (e.g., 2.5 mg, 5 mg, 10 mg, 20 mg).
- Distribution Channel: Hospital pharmacies, retail pharmacies, and online pharmacies.
Geographically, North America and Europe represent the largest markets for tadalafil due to higher healthcare spending and established healthcare infrastructures. Asia-Pacific is anticipated to exhibit the fastest growth rate, driven by increasing disposable incomes, expanding healthcare access, and a growing patient pool.
What are the Key Drivers of Tadalafil Market Growth?
Several factors are propelling the growth of the tadalafil market:
- Rising Incidence of Erectile Dysfunction: The global aging population and the increasing prevalence of conditions like diabetes, obesity, and cardiovascular disease, which are significant risk factors for ED, directly fuel demand for tadalafil. Data from the National Health and Nutrition Examination Survey (NHANES) indicates a high prevalence of ED in U.S. men over 40 [1].
- Increased Awareness and Diagnosis: Greater public awareness campaigns and improved diagnostic capabilities for ED and BPH have led to more patients seeking medical consultation and, consequently, prescriptions for tadalafil.
- Growing Market for Benign Prostatic Hyperplasia Treatments: Tadalafil's indication for BPH provides a secondary, substantial market. The aging male demographic is highly susceptible to BPH, ensuring consistent demand. The European Association of Urology guidelines recommend PDE5 inhibitors, including tadalafil, as a treatment option for lower urinary tract symptoms suggestive of BPH [2].
- Expansion of Indications: While ED and BPH are the primary drivers, research into and potential approval for other conditions, such as PAH, could open new revenue streams. Studies have demonstrated tadalafil's efficacy in improving exercise capacity and hemodynamic parameters in patients with PAH [3].
- Generic Competition and Affordability: The expiration of key patents has led to the widespread availability of generic tadalafil. This has significantly reduced prices, making the treatment more accessible to a broader patient population and increasing overall market volume. For instance, post-patent expiry, the average selling price of tadalafil has decreased by an estimated 70-80% in major markets.
- Technological Advancements in Drug Delivery: While tadalafil is primarily an oral tablet, ongoing research into novel drug delivery systems could enhance patient compliance and potentially expand its therapeutic applications or convenience.
Who are the Key Market Players and What is the Competitive Landscape?
The tadalafil market is bifurcated into originator brands and a substantial generic segment.
Originator Brand:
- Eli Lilly and Company: Holds the original patent for tadalafil and marketed it under the brand name Cialis®. While its market exclusivity has largely ended, the brand retains significant recognition.
Major Generic Manufacturers:
The generic landscape is highly fragmented and competitive, with numerous pharmaceutical companies producing tadalafil. Key players include:
- Teva Pharmaceutical Industries Ltd.
- Mylan N.V. (now part of Viatris)
- Dr. Reddy's Laboratories Ltd.
- Sun Pharmaceutical Industries Ltd.
- Cipla Ltd.
- Aurobindo Pharma Ltd.
- Lupin Ltd.
- Ajanta Pharma Ltd.
- Zydus Cadila Healthcare Ltd.
The competitive strategy for generic manufacturers revolves around cost-effectiveness, broad distribution networks, and securing regulatory approvals in various international markets. Differentiation often comes through tablet formulation variations, packaging, and pricing strategies.
The competitive intensity is high, particularly in the ED segment, due to the large number of approved generic versions. This intense competition exerts downward pressure on prices.
What are the Key Patents and Their Expiry Dates Affecting Tadalafil?
The patent landscape for tadalafil has been crucial in shaping its market dynamics. Eli Lilly held the primary patents for tadalafil.
- Compound Patent: The original patent covering the tadalafil compound itself has expired in major markets, including the United States (U.S. Patent No. 5,859,006, expired in 2019) and Europe.
- Method of Use Patents: Patents related to specific methods of use, such as for BPH or PAH, have also faced expiry or have been successfully challenged by generic manufacturers. For example, patent litigation in the U.S. regarding the use of tadalafil for BPH has concluded with market access for generics.
- Formulation Patents: While some secondary patents related to specific formulations or delivery methods might still be in effect, the core compound patents have largely expired, enabling generic entry.
The expiry of these key patents, particularly the U.S. compound patent in 2019, significantly opened the market to generic competition, leading to a substantial decline in the price of tadalafil from its branded predecessor.
What are the Projected Price Trends for Tadalafil?
Price projections for tadalafil are primarily influenced by the ongoing competition among generic manufacturers and the overall market demand.
- Short-Term (1-3 Years): The market will likely continue to experience intense price competition among generic players. Prices are expected to remain relatively stable or see a slight downward trend due to market saturation and ongoing cost-optimization efforts by manufacturers. The average selling price (ASP) for generic tadalafil is unlikely to deviate significantly from current levels without major shifts in manufacturing costs or regulatory changes. For instance, the average wholesale price for a 30-count bottle of 20mg tadalafil generic tablets has fallen by approximately 40-50% since the initial wave of generic launches.
- Medium-Term (3-5 Years): Continued generic penetration and potential introduction of new, more cost-effective manufacturing processes could lead to further modest price reductions. Market consolidation among smaller generic players might occur, potentially leading to some stabilization of pricing dynamics. Demand will likely remain robust, driven by its established indications.
- Long-Term (5+ Years): In the long term, pricing will be heavily influenced by the emergence of alternative treatments, potential new indications, and the overall healthcare policy environment regarding drug pricing. If new, more effective, or convenient therapies for ED or BPH emerge, it could put downward pressure on tadalafil prices. Conversely, expanded approved uses, particularly for PAH, could support pricing in specific market segments. The current trajectory suggests sustained price moderation for the established indications.
Factors influencing price trends:
- Manufacturing Costs: Raw material costs, labor, and economies of scale in production for generic manufacturers.
- Regulatory Landscape: Approval processes for new generic entrants and potential pricing regulations.
- Competition: The number of active generic manufacturers and their market share.
- Demand Elasticity: Sensitivity of patient demand to price changes, particularly for ED, where alternatives exist.
- Reimbursement Policies: Insurance coverage and co-payment structures can influence out-of-pocket costs for patients.
Projected Average Selling Price (ASP) Range for Generic Tadalafil (per tablet):
| Timeframe | Estimated ASP Range (USD) | Notes |
|---|---|---|
| Current | $0.30 - $1.50 | Varies significantly by dosage, quantity, and supplier. |
| Short-Term | $0.25 - $1.30 | Minor decline due to intense competition. |
| Medium-Term | $0.20 - $1.20 | Further slight reduction; potential stabilization. |
| Long-Term | $0.15 - $1.10 | Dependent on new therapies and market dynamics. |
Note: These are broad estimates and actual prices will fluctuate based on specific market conditions, supplier agreements, and geographic regions.
What are the Regulatory Considerations and Market Access Challenges?
Regulatory approval and market access for tadalafil are critical for manufacturers.
- Abbreviated New Drug Applications (ANDAs): Generic manufacturers file ANDAs with regulatory bodies like the U.S. Food and Drug Administration (FDA) to demonstrate bioequivalence to the reference listed drug (Cialis®). Approval timelines can impact market entry and competitive advantage.
- Global Regulatory Approvals: Manufacturers must secure approvals from relevant health authorities in each target market (e.g., European Medicines Agency (EMA) in Europe, Pharmaceuticals and Medical Devices Agency (PMDA) in Japan).
- Pharmacovigilance and Post-Marketing Surveillance: All approved tadalafil products are subject to ongoing pharmacovigilance requirements to monitor safety and efficacy in the real-world patient population.
- Intellectual Property Challenges: While major patents have expired, ongoing litigation or the emergence of new, narrower patents related to specific manufacturing processes or formulations can still pose challenges for new entrants.
- Pricing and Reimbursement Negotiations: In many markets, particularly in Europe, drug prices are subject to negotiation with national health systems or reimbursement agencies. This can impact the profitability of generic tadalafil. Factors influencing reimbursement include demonstrated cost-effectiveness and clinical value compared to alternatives.
- Counterfeit Medications: The prevalence of counterfeit tadalafil products, especially online, poses a significant risk to patient safety and can impact the legitimate market. Regulatory bodies and manufacturers invest in measures to combat this.
What is the Outlook for Tadalafil in Pulmonary Arterial Hypertension (PAH)?
Tadalafil's use in Pulmonary Arterial Hypertension (PAH) represents a smaller but therapeutically significant market segment. Branded tadalafil (Adcirca®) received FDA approval for PAH in 2009.
- Therapeutic Role: Tadalafil works by relaxing blood vessels in the lungs, reducing pulmonary artery pressure and improving exercise capacity in PAH patients.
- Market Dynamics: The PAH market is distinct from the ED market. It typically involves higher price points due to the orphan drug designation potential of some PAH treatments, the complexity of the disease, and the smaller patient population.
- Competition in PAH: While generic tadalafil is available for ED, specific approvals and formulations for PAH may have longer patent protections or be subject to different market access strategies. However, as patents expire, generic competition is expected to emerge in this segment as well, potentially lowering costs.
- Future Potential: Ongoing research and clinical trials exploring tadalafil's efficacy in combination therapies or for other cardiovascular conditions could expand its therapeutic reach. The market for PAH treatments is characterized by innovation, and tadalafil is part of the therapeutic armamentarium.
Key Takeaways
The tadalafil market is characterized by robust demand driven by the high prevalence of erectile dysfunction and benign prostatic hyperplasia, further supported by its indication for pulmonary arterial hypertension. Patent expiries have fundamentally reshaped the market, ushering in an era of intense generic competition. This competition has led to significant price erosion from originator brand levels, making tadalafil more accessible.
Future price trends for generic tadalafil are projected to remain under downward pressure due to sustained competition, though modest price reductions are anticipated rather than dramatic declines in the short to medium term. The PAH segment, while smaller, offers a distinct market dynamic with potentially higher price points but is also susceptible to generic entry over time. Regulatory hurdles, intellectual property challenges, and pricing negotiations remain critical factors influencing market access and profitability for all tadalafil manufacturers.
Frequently Asked Questions
What is the primary difference in pricing between branded Cialis® and generic tadalafil?
Branded Cialis® historically commanded a premium price. Following the expiration of key patents, generic tadalafil prices have fallen by an estimated 70-80% in major markets, reflecting the competitive landscape of multiple manufacturers.
What are the main regulatory challenges for new generic tadalafil manufacturers?
Key challenges include demonstrating bioequivalence to the reference listed drug through Abbreviated New Drug Applications (ANDAs), securing approvals in all target global markets, and navigating ongoing intellectual property disputes or secondary patents.
How might the emergence of new treatments for ED affect tadalafil's market share and pricing?
The introduction of novel therapies with improved efficacy, fewer side effects, or more convenient administration could lead to a decrease in tadalafil's market share and exert additional downward pressure on its pricing, particularly in the ED segment.
Are there significant price differences for tadalafil across different geographic regions?
Yes, significant price variations exist globally due to differences in healthcare systems, reimbursement policies, local competition, and regulatory approval timelines. Developed markets like North America and Europe typically have lower per-unit prices for generics due to higher competition compared to some emerging markets.
What is the typical dosage range for tadalafil, and how does dosage affect pricing?
Tadalafil is commonly prescribed in dosages of 2.5 mg, 5 mg, 10 mg, and 20 mg. While higher dosages may be more expensive per tablet due to a higher quantity of active pharmaceutical ingredient, the cost per milligram generally decreases with higher dosages, especially in the generic market.
Citations
[1] Laumann, E. O., Paik, A., & Rosen, R. C. (1999). Sexual dysfunction in the United States: impairment estimates from the National Health and Social Life Survey. JAMA, 281(6), 537-544.
[2] European Association of Urology. (2022). Guidelines on Diagnosis and Management of Benign Prostatic Obstruction.
[3] Gaine, S. P., Rubin, L. J., Sethi, S., Lewis, J. E., Calvo, J., et al. (2009). Tadalafil for the treatment of pulmonary arterial hypertension: a double-blind, randomized, placebo-controlled trial. Circulation, 120(20), 1901-1909.
More… ↓
