Share This Page
Drug Price Trends for silver sulfadiazine
✉ Email this page to a colleague
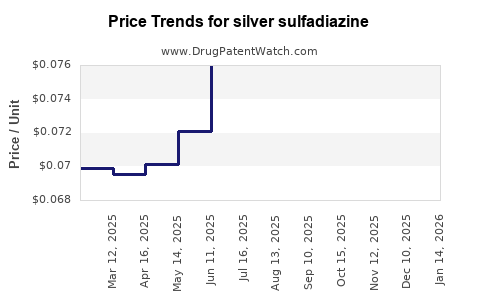
Average Pharmacy Cost for silver sulfadiazine
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| SILVER SULFADIAZINE 1% CREAM | 59762-0131-04 | 0.12827 | GM | 2026-03-18 |
| SILVER SULFADIAZINE 1% CREAM | 59762-0131-02 | 0.31688 | GM | 2026-03-18 |
| SILVER SULFADIAZINE 1% CREAM | 67877-0124-85 | 0.25137 | GM | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Silver Sulfadiazine
Silver sulfadiazine is a topical antimicrobial agent widely used for burn wound management and prevention of bacterial infections. Its global market is influenced by factors such as burn prevalence, regulatory approval status, availability of alternative treatments, and manufacturing costs.
Market Size and Growth Drivers
Current Market Value
As of 2022, the global silver sulfadiazine market was valued at approximately $150 million. The market is projected to grow at a Compound Annual Growth Rate (CAGR) of 4-5% through 2028, reaching around $200 million by that year.
Key Regional Markets
- North America: Largest market, driven by advanced healthcare infrastructure and burn treatment protocols.
- Europe: Steady demand, with regulatory approval supporting market persistence.
- Asia-Pacific: Fast growth, propelled by increasing burn incidence and expanding healthcare access.
Demand Drivers
- Rising incidence of burns and thermal injuries, particularly in developing countries.
- Shift towards wound care management protocols that favor topical antimicrobials.
- Regulatory approvals for generic formulations that lower costs and improve accessibility.
Market Challenges
- Competition from alternative topical agents (e.g., silver nitrate, mafenide acetate).
- Stringent regulations impacting drug approval and market entry in certain countries.
- Growing concerns over antibiotic resistance, prompting search for alternative therapies.
Competitive Landscape
Major Players
- GSK (GlaxoSmithKline): Initially developed and marketed silver sulfadiazine formulations.
- Systagenix and Mölnlycke: Focused on advanced wound care products, including silver-based dressings.
- Generic manufacturers: Entered the market due to patent expirations and cost sensitivity.
Pricing Strategies
The market has a mix of branded and generic products. Generic versions are priced approximately 20-30% lower than branded counterparts, with retail costs ranging from $10 to $30 per 50g tube or dressing.
Regulatory Considerations
Most developed markets permit over-the-counter (OTC) sales of silver sulfadiazine for topical application, whereas some countries require prescription-only access. Regulatory differences impact market penetration in emerging markets.
Price Projections and Future Trends
Short-term (2023-2025)
Prices will likely remain stable due to supply chain consistency and stable demand. Slight reductions (5-10%) are possible from increased competition among generics.
Medium-term (2025-2028)
Innovation in wound dressing technologies incorporating silver compounds or advanced delivery systems may impact topical silver sulfadiazine formulations. A potential price decline of 10-15% could occur if these alternatives gain regulatory approvals and clinical acceptance.
Long-term (2028 and beyond)
Prices could decline further as new antimicrobial agents and bioengineered tissue products replace traditional silver sulfadiazine therapy. The market value may plateau or contract slightly, especially in regions with increasing adoption of modern wound care products.
Impact of Patent and Regulatory Dynamics
- The original GSK patent for silver sulfadiazine expired in multiple jurisdictions between 2000-2010, enabling generics.
- Ongoing regulatory scrutiny on antimicrobial agents due to resistance concerns may influence future approvals and pricing.
- Innovations like nanotechnology drug delivery systems could command premium prices initially but are likely to mature into competitive markets.
Summary Table: Pricing Range and Market Size (2022-2028)
| Year | Estimated Market Value | Price Range (per 50g tube/dressing) | Key Drivers | Notes |
|---|---|---|---|---|
| 2022 | $150 million | $10 - $30 | Burn incidence, generics | Base year |
| 2025 | $170 - $180 million | $8 - $28 | Competition, innovation | Slight price decline |
| 2028 | $200 million | $7 - $25 | New treatments, cost sensitivity | Stabilizing/declining |
Key Takeaways
- The global silver sulfadiazine market is expected to grow modestly, influenced by burn treatments, burn prevalence, and new wound care technologies.
- Price stability is anticipated in the short term, with gradual decreases driven by increased competition and innovation.
- Regulatory landscapes vary by region, impacting market access and pricing strategies.
- The emergence of alternative antimicrobial and wound management therapies poses potential long-term competitive threats.
- Early entrants to innovations incorporating nanotechnology or bioengineering may command premium prices temporarily but will face commoditization as these devices become more common.
FAQs
1. What factors most influence silver sulfadiazine pricing?
Market demand from burn injury management, competitive pressure from generics, regulatory environment, and advances in wound care technology.
2. Are generic versions significantly cheaper than branded formulations?
Yes. Generics typically cost 20-30% less than original branded products, resulting in overall market price decreases.
3. How do regulatory differences affect market prices?
Regions requiring prescription-only access limit sales volumes and affect pricing structures, while OTC availability can increase market penetration and competition.
4. Will new wound care technologies replace silver sulfadiazine?
Likely in some markets. Innovations such as nanotech-based antimicrobials and bioengineered tissues could supplant traditional treatments over the next decade.
5. What are the main risks to silver sulfadiazine market stability?
Antibiotic resistance concerns, competition from more advanced therapies, and regulatory restrictions may hinder growth or lead to price reductions.
References
- MarketsandMarkets, "Wound Care Market by Product," 2022.
- GSK annual reports, 2021.
- Statista, "Global Burn Incidence," 2022.
- FDA & EMA regulatory documents, 2022.
- Grand View Research, "Antimicrobial Market Analysis," 2022.
More… ↓
