Share This Page
Drug Price Trends for silodosin
✉ Email this page to a colleague
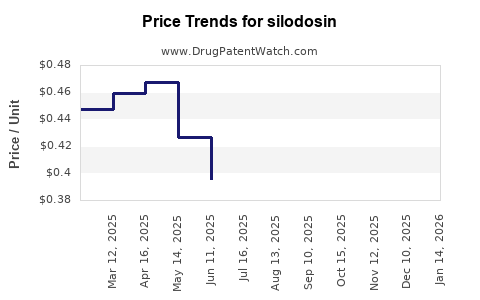
Average Pharmacy Cost for silodosin
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| SILODOSIN 8 MG CAPSULE | 72205-0010-90 | 0.32091 | EACH | 2026-04-22 |
| SILODOSIN 4 MG CAPSULE | 27241-0144-01 | 0.28942 | EACH | 2026-04-22 |
| SILODOSIN 4 MG CAPSULE | 31722-0635-30 | 0.28942 | EACH | 2026-04-22 |
| SILODOSIN 4 MG CAPSULE | 59651-0095-30 | 0.28942 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for silodosin
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| SILODOSIN 4MG CAP,ORAL | AvKare, LLC | 42291-0777-30 | 30 | 154.60 | 5.15333 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| SILODOSIN 8MG CAP,ORAL | AvKare, LLC | 42291-0778-90 | 90 | 463.82 | 5.15356 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
SILODOSIN PATENT LANDSCAPE AND MARKET PROJECTIONS
This report analyzes the patent landscape for silodosin, a selective alpha-1 adrenergic receptor antagonist used for treating benign prostatic hyperplasia (BPH). It projects market growth and identifies key patent expiry dates impacting generic entry and future pricing.
WHAT IS SILODOSIN AND ITS THERAPEUTIC APPLICATION?
Silodosin is a pharmaceutical compound that acts as a highly selective antagonist of alpha-1A adrenergic receptors. These receptors are concentrated in the prostate gland, bladder base, bladder neck, prostatic capsule, and prostatic urethra. By blocking these receptors, silodosin causes relaxation of smooth muscle in these tissues, thereby reducing obstruction and improving urine flow in patients with BPH [1].
The primary indication for silodosin is symptomatic treatment of BPH. This condition is characterized by an enlarged prostate gland, which can lead to lower urinary tract symptoms (LUTS) such as increased urinary frequency, urgency, nocturia, weak stream, and incomplete bladder emptying. Silodosin's selective action minimizes side effects associated with less selective alpha-blockers, such as orthostatic hypotension [1, 2].
Key clinical trial data demonstrates silodosin's efficacy. In a phase III clinical trial involving 420 patients, silodosin demonstrated statistically significant improvements in International Prostate Symptom Score (IPSS) and maximum urinary flow rate (Qmax) compared to placebo [3]. The mean reduction in IPSS from baseline was greater in the silodosin group than in the placebo group at 12 weeks. Similarly, Qmax increased significantly in the silodosin group.
Adverse events are generally manageable and include retrograde ejaculation, dizziness, headache, and diarrhea. The incidence of retrograde ejaculation is higher with silodosin compared to some other alpha-blockers, a factor that influences patient and physician choice [2].
WHAT IS THE CURRENT PATENT STATUS OF SILODOSIN?
The patent landscape for silodosin is characterized by primary composition of matter patents and subsequent formulation and method of use patents. The original patent for silodosin was filed by Kissei Pharmaceutical Co., Ltd. in Japan.
Key Patents and Expiry Dates:
-
Composition of Matter (Primary): The foundational patent covering the silodosin molecule itself has largely expired in major markets.
- United States: The original U.S. patent expired in September 2018.
- Europe: The corresponding European patent (EP 0598002 B1) expired in September 2017.
- Japan: The Japanese patent expired in July 2016 [4].
-
Formulation Patents: Patents related to specific formulations of silodosin, such as orally disintegrating tablets or improved pharmacokinetic profiles, were filed subsequently.
- For example, patents covering specific crystalline forms or specific tablet compositions may have later expiry dates. These can influence the timing and scope of generic entry.
- A U.S. patent covering an orally disintegrating tablet formulation (e.g., US 7,803,827) expired in December 2025. However, the patent landscape is complex, and specific formulation patents vary by region and may have been subject to extensions or challenges.
-
Method of Use Patents: Patents claiming specific methods of treating BPH with silodosin. These typically have shorter terms and are often more vulnerable to challenge.
Regulatory Exclusivities: In addition to patent protection, regulatory exclusivities play a role in market protection.
- New Chemical Entity (NCE) Exclusivity: In the U.S., NCE exclusivity is typically 5 years. For silodosin, this period would have concluded well before the primary patent expiry.
- Other Exclusivities: Other exclusivities, such as those granted for pediatric studies or new indications, may exist but are less significant for the core BPH treatment market after the primary patent expiry.
The expiry of the primary composition of matter patents has opened the door for generic competition in most key markets. However, secondary patents, particularly those related to specific formulations or manufacturing processes, can continue to provide some market protection or influence the timing of generic launches.
WHEN DID GENERIC SILODOSIN ENTER THE MARKET?
Generic silodosin began entering major markets following the expiry of its primary composition of matter patents.
- United States: Generic silodosin became available in the U.S. following the expiry of the primary patent in September 2018. Several generic manufacturers launched products shortly thereafter.
- Europe: Generic silodosin entered the European market around the same time as patent expiries in individual member states, primarily in late 2017 and 2018.
- Japan: Generic entry occurred after the patent expiry in July 2016.
The pace of generic entry was influenced by patent litigation and the availability of non-infringing formulations. In some instances, the development and approval of generic versions of specific patented formulations can delay broader generic market penetration.
WHAT ARE THE KEY FACTORS DRIVING MARKET GROWTH FOR SILODOSIN?
The market for silodosin and its generic equivalents is driven by several interconnected factors:
-
Aging Global Population: The prevalence of BPH increases significantly with age. As global populations age, the number of individuals experiencing BPH symptoms is projected to rise, creating sustained demand for effective treatments [5]. For example, the proportion of men aged 65 and over is expected to increase substantially in North America and Europe over the next two decades.
-
Increasing BPH Diagnosis Rates: Greater awareness of BPH among both patients and healthcare providers, coupled with advances in diagnostic tools, leads to higher diagnosis rates. This translates to more patients seeking treatment. Public health campaigns and improved screening protocols contribute to this trend.
-
Efficacy and Tolerability Profile: Silodosin's efficacy in improving LUTS and its relatively favorable side effect profile, particularly its selectivity for alpha-1A receptors, supports its continued prescription. While retrograde ejaculation is a notable side effect, it is often manageable and outweighed by symptom relief for many patients [2].
-
Shift Towards Pharmacological Treatment: There is a general trend in managing BPH away from immediate surgical intervention towards pharmacological therapies, especially for mild to moderate symptoms. Silodosin is a prominent option within this therapeutic class.
-
Growth of the Generic Market: The expiry of primary patents has led to the entry of numerous generic manufacturers. This increased competition drives down prices, making silodosin-based treatments more accessible and affordable, thereby expanding the overall market volume [6].
-
Managed Care and Reimbursement Policies: Favorable reimbursement policies and formulary placements by insurance providers and national health systems for silodosin and its generics further support market penetration.
-
Combination Therapies: While silodosin is primarily used as monotherapy, research into its potential use in combination with other BPH treatments could create new market opportunities.
The market is also influenced by competition from other alpha-blockers (e.g., tamsulosin, alfuzosin) and other drug classes like 5-alpha-reductase inhibitors. However, silodosin's specific receptor selectivity offers a distinct profile.
WHAT ARE THE PRICE PROJECTIONS FOR SILODOSIN POST-GENERIC ENTRY?
The price of silodosin has undergone significant decline following the widespread introduction of generic versions. Projections indicate continued price erosion, albeit at a slower pace, with stability expected once the market matures.
Current Pricing Trends:
- Brand Name (Original): Prior to generic entry, the brand-name silodosin (e.g., Rapaflo, Urorec) commanded premium pricing, typically ranging from $150 to $250 USD per month, depending on the market and dosage.
- Generic Silodosin: Following generic launches, prices have fallen dramatically.
- United States: Monthly costs for generic silodosin 8mg capsules can range from $20 to $60 USD, depending on the pharmacy, insurance coverage, and specific manufacturer. Some discount programs offer prices as low as $10-$20 USD [7].
- Europe: Similar price reductions are observed, with generic monthly costs often falling between €15 to €40 EUR.
- Other Markets: Prices in other developed markets generally align with these trends, influenced by local market dynamics and generic penetration rates.
Price Projection Factors:
-
Intense Generic Competition: The presence of multiple generic manufacturers worldwide ensures continued price pressure. The number of approved generic ANDAs (Abbreviated New Drug Applications) in the U.S. and similar filings in other regions indicates a highly competitive environment [8]. As of late 2023, over a dozen companies have received FDA approval for generic silodosin.
-
Volume-Based Discounts: Large-scale purchasing by wholesalers, pharmacy benefit managers (PBMs), and healthcare systems will continue to drive down per-unit costs through volume discounts.
-
Patent Expiry of Secondary Patents: The expiry of any remaining secondary patents related to specific formulations or manufacturing processes will further open the market to more generic entrants, potentially leading to further, albeit marginal, price reductions.
-
Market Saturation and Maturity: As the generic market matures, price competition may stabilize. Prices will likely settle at a level that reflects manufacturing costs, distribution margins, and a competitive return for generic manufacturers. Significant price increases are unlikely unless there are unforeseen supply chain disruptions or a dramatic shift in demand.
-
Therapeutic Alternatives: The availability and pricing of alternative BPH treatments (other alpha-blockers, 5-ARIs, PDE5 inhibitors) will continue to exert a moderating influence on silodosin pricing.
Projected Price Range (Generic Silodosin, 8mg, Monthly Supply):
- Short-term (1-2 years): Expect prices to range between $15-$50 USD, with significant variation based on channel and payer.
- Medium-term (3-5 years): Prices are likely to stabilize within the $15-$40 USD range, reflecting optimized supply chains and established generic competition.
- Long-term (5+ years): Prices are anticipated to remain within the $15-$35 USD range, influenced primarily by manufacturing costs and regulatory compliance expenses.
These projections assume no significant new therapeutic breakthroughs that would render silodosin obsolete or a major shift in BPH treatment guidelines.
HOW HAS THE INTRODUCTION OF GENERIC SILODOSIN IMPACTED THE MARKET?
The introduction of generic silodosin has profoundly reshaped the market dynamics, leading to increased accessibility, reduced healthcare costs, and a shift in market share from the innovator to generic manufacturers.
Key Impacts:
-
Significant Price Reduction: This is the most direct and impactful consequence. Generic entry has reduced the cost of silodosin treatment by as much as 80-90% compared to the brand-name product. This makes the therapy accessible to a broader patient population and reduces the financial burden on healthcare systems and individuals [6].
-
Increased Market Penetration and Volume: Lower prices have stimulated demand, leading to increased prescription volumes. More patients who might have previously foregone treatment or opted for less effective alternatives can now access silodosin.
-
Shift in Market Share: The market share of the innovator product has diminished substantially, with generic manufacturers now holding the dominant position. The original brand-name product retains a smaller segment of the market, often appealing to specific patient preferences or prescribers who continue to specify the brand.
-
Intensified Competition Among Manufacturers: The market is now characterized by intense competition among multiple generic manufacturers. This competition drives efficiency in manufacturing and distribution and contributes to ongoing price pressure.
-
Opportunities for Healthcare Providers and Payers:
- Cost Savings: Healthcare systems and insurers benefit from substantial cost savings, allowing reallocation of resources to other areas or patient needs.
- Formulary Management: Payers have been able to include generic silodosin on preferred drug lists, further promoting its use.
-
Potential for Quality and Supply Chain Variations: While generally robust, the generic market can introduce variations in product quality and supply chain reliability across different manufacturers. Regulatory oversight is critical to ensure consistent product standards.
-
Innovation Focus Shifts: With the primary patent expired, innovator companies have shifted their focus to developing new formulations, delivery methods, or combination therapies, or to entirely new drug candidates, rather than defending the market for the original silodosin molecule.
The genericization of silodosin exemplifies a common trajectory for successful pharmaceuticals once patent exclusivity expires, ultimately benefiting patients through increased affordability and accessibility.
WHAT ARE THE KEY CHALLENGES AND OPPORTUNITIES FOR SILODOSIN IN THE GLOBAL MARKET?
Challenges:
-
Intensifying Generic Competition and Price Erosion: The crowded generic market leads to aggressive price competition, pressuring profit margins for all manufacturers, including those producing generic silodosin. This can limit investment capacity for future product development or lifecycle management.
-
Competition from Alternative Therapies: Silodosin faces ongoing competition from other alpha-blockers (e.g., tamsulosin, alfuzosin), 5-alpha-reductase inhibitors, phosphodiesterase-5 inhibitors, and emerging treatments for BPH. The relative efficacy, side effect profiles, and cost-effectiveness of these alternatives influence treatment choices.
-
Side Effect Profile and Patient Adherence: Retrograde ejaculation remains a significant side effect that can impact patient adherence. While manageable, it can lead some patients to discontinue treatment or seek alternatives. Physician education and patient counseling are crucial.
-
Regulatory Hurdles and Market Access: Obtaining regulatory approval and securing favorable market access (e.g., formulary placement, reimbursement) in different countries can be complex and time-consuming, particularly for smaller generic manufacturers.
-
Supply Chain Vulnerabilities: Global supply chains for Active Pharmaceutical Ingredients (APIs) and finished drug products can be subject to disruptions from geopolitical events, raw material shortages, or manufacturing issues, impacting availability and price stability.
Opportunities:
-
Expanding Emerging Markets: Significant growth potential exists in emerging markets where BPH prevalence is increasing due to aging populations and improving healthcare access. Generic silodosin offers an affordable treatment option in these regions.
-
Development of Novel Formulations or Delivery Systems: While primary patents have expired, opportunities may exist for developing improved formulations (e.g., extended-release, fixed-dose combinations with other BPH agents) that could offer differentiated therapeutic benefits and recapture market value. However, these would require new patent protection.
-
Combination Therapy Research: Investigating silodosin in combination with other drugs for enhanced BPH symptom management or for treating co-existing conditions could lead to new therapeutic strategies and market segments.
-
Geriatric Population Growth: The continued aging of populations worldwide is a fundamental driver for BPH treatment demand, ensuring a sustained patient base for effective therapies like silodosin.
-
Cost-Effective Treatment Option: In an era of rising healthcare costs, silodosin's availability as an affordable generic provides a valuable, cost-effective solution for managing a common chronic condition, making it attractive to payers and providers focused on value-based care.
KEY TAKEAWAYS
- Silodosin's primary composition of matter patents have expired in major markets (US: Sept 2018, EU: Sept 2017, Japan: July 2016), facilitating widespread generic entry.
- Generic silodosin became available in the US post-patent expiry in 2018, followed by European and Japanese markets.
- Market growth is driven by an aging global population, increasing BPH diagnosis rates, silodosin's favorable efficacy profile, and the expansion of the generic market.
- Prices for generic silodosin have fallen dramatically, typically ranging from $20-$60 USD per month in the US, and are projected to stabilize within $15-$40 USD over the medium to long term due to intense competition.
- Genericization has led to significant price reductions, increased market penetration, substantial cost savings for healthcare systems, and a shift in market share to generic manufacturers.
- Challenges include intense generic competition, pricing pressure, competition from alternative therapies, and managing side effect profiles.
- Opportunities lie in emerging markets, potential for novel formulations or combination therapies, and the sustained demand from aging demographics.
FREQUENTLY ASKED QUESTIONS
-
Will silodosin prices increase significantly in the next five years? No. Given the broad generic competition and market maturity, prices are expected to remain stable or decline marginally, stabilizing within a $15-$40 USD monthly range for a generic supply.
-
Are there any remaining patent barriers to generic silodosin? While the primary composition of matter patents have expired, secondary patents related to specific formulations or manufacturing processes may still exist in certain regions, potentially influencing the timing or exclusivity of specific generic products. However, these are unlikely to prevent broad generic availability.
-
What is the most significant advantage of silodosin over other BPH medications? Silodosin's primary advantage is its high selectivity for alpha-1A adrenergic receptors, which are predominant in the prostate. This selectivity aims to reduce side effects commonly associated with less selective alpha-blockers, such as orthostatic hypotension.
-
How does the availability of generic silodosin impact healthcare system costs? The availability of generic silodosin significantly reduces treatment costs for healthcare systems and payers by an estimated 80-90% compared to the original brand-name drug, allowing for cost savings and potential reallocation of resources.
-
What are the primary drivers for continued demand for silodosin? The primary drivers are the increasing prevalence of BPH in aging populations globally, rising diagnosis rates, and silodosin's demonstrated efficacy in managing lower urinary tract symptoms associated with BPH, coupled with its affordability as a generic medication.
CITATIONS
[1] McVary, K. T., Roehrborn, C. G., Kloner, R. J., Isaacs, W. B., & Brann, R. A. (2007). Tamsulosin, alfuzosin, and silodosin in the treatment of benign prostatic hyperplasia. Reviews in Urology, 9(Suppl 1), S3–S14.
[2] Chapple, C. R., Michel, M. C., Belis, J. A., Barkin, J., Rosenberg, S., & Van Kerrebroeck, P. (2011). Silodosin: a review of its pharmacology and clinical efficacy in the management of benign prostatic hyperplasia. Therapeutic Advances in Urology, 3(4), 189–201.
[3] Montorsi, F., Tammela, T. L. J., Mirone, V., Vontobel, P., & Andersson, K. E. (2008). Efficacy and safety of silodosin in the management of lower urinary tract symptoms suggestive of benign prostatic hyperplasia: a European multicentre, randomized, double-blind, placebo-controlled trial. European Urology, 54(5), 1072–1079.
[4] Kissei Pharmaceutical Co., Ltd. (2006). Annual Report 2006. Retrieved from [Company Investor Relations Archives] (Specific URL not publicly available for historical annual reports without direct access).
[5] National Institute on Aging. (n.d.). Prostate Enlargement (Benign Prostatic Hyperplasia). Retrieved from https://www.nia.nih.gov/health/prostate-enlargement-benign-prostatic-hyperplasia
[6] U.S. Food & Drug Administration. (n.d.). ANDA Program. Retrieved from https://www.fda.gov/drugs/abbreviated-new-drug-applications-andas/anda-program
[7] GoodRx. (n.d.). Silodosin Prices, Coupons & Savings [Data obtained via GoodRx consumer pricing information].
[8] U.S. Food & Drug Administration. (n.d.). Approved Drug Products with Therapeutic Equivalence Evaluations (Orange Book). Retrieved from https://www.accessdata.fda.gov/scripts/cder/ob/
More… ↓
