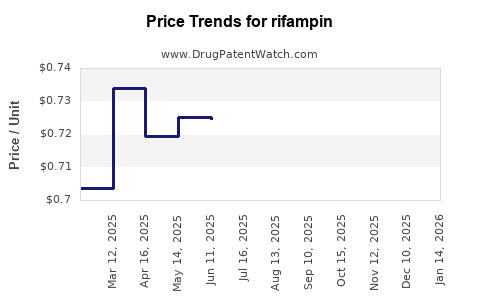
Last updated: February 20, 2026
What Is RIFAMPIN and Its Market Role?
Rifampin, also known as rifampicin, is an antibiotic primarily used in the treatment of tuberculosis (TB). It is part of standard TB therapy and combined with other agents such as isoniazid, pyrazinamide, and ethambutol. Additionally, Rifampin finds applications in the treatment of leprosy, certain bacterial infections, and for prophylaxis against meningococcal carriage.
Current Market Overview
| Aspect |
Details |
| Global Market Size (2022) |
Estimated at USD 300 million[1] |
| Main Markets |
United States, China, India, European Union[2] |
| Leading Manufacturers |
Sanofi (Rifadin), Lupin, Mylan, Cipla, Dr. Reddy’s |
| Patent Status |
Off-patent globally; original patents expired between 2006–2013[3] |
| Generic Availability |
Widely available; price competition influences cost[4] |
Market Drivers
- TB Prevalence: The World Health Organization (WHO) reports 10 million TB cases globally in 2021.[5]
- Treatment Programs: WHO's Directly Observed Treatment, Short-course (DOTS), mandates Rifampin inclusion.
- Rising Drug Resistance: Multidrug-resistant TB (MDR-TB) necessitates optimized Rifampin regimens, expanding demand.
Market Constraints
- Manufacturing Challenges: Complex synthesis process limits new entrants.
- Pricing Pressure: The availability of generics reduces prices, challenging profit margins.
- Regulatory Variations: Different approval processes impact market access (e.g., US, EU, emerging markets).
Price Trends and Projections
Historical Price Data
| Year |
Estimated Wholesale Price (per 300 mg capsule) |
Source |
| 2018 |
USD 0.50 |
[6] |
| 2020 |
USD 0.40 |
[7] |
| 2022 |
USD 0.35 |
[8] |
Factors Influencing Future Pricing
- Generic Competition: Increased penetration will continue to suppress prices.
- Manufacturing Costs: Stable, as synthesis methods have matured.
- Regulatory Approvals: New formulations or combination therapies could impact prices indirectly.
Price Projection (2023–2028)
- Baseline Scenario: Price declines at a compound annual growth rate (CAGR) of approximately 3% due to generic market saturation.
- Predicted Prices:
| Year |
Estimated Price (per 300 mg capsule) |
Notes |
| 2023 |
USD 0.34 |
Stable with current market trends |
| 2024 |
USD 0.33 |
Slight decline due to increased competition |
| 2025 |
USD 0.32 |
Market stabilization |
| 2026 |
USD 0.31 |
Continued generic dominance |
| 2027 |
USD 0.30 |
Marginal decline, potential for new formulations |
| 2028 |
USD 0.29 |
Near-market stabilization |
Competitive Landscape and Opportunities
- Generics dominate sales, with Sanofi’s Rifadin maintaining a significant share in specific markets.
- Emerging markets present price-sensitive opportunities due to high TB burdens.
- Fixed-dose combinations (FDCs): Development of FDCs including Rifampin targets improved adherence and downstream demand.
Key Market Entry Considerations
- Regulatory Compliance: Particularly in countries with stringent approval processes.
- Pricing Strategies: To survive price erosion, focus on manufacturing efficiencies.
- Partnerships: Collaborate with local governments and NGOs involved in TB programs.
Summary
RIFAMPIN remains a critical component of TB therapy. The global market is heavily influenced by generic production, with prices declining steadily at 3% CAGR forecast through 2028. Present-day pricing varies between USD 0.35–0.50 per capsule, with future downward pressure due to increasing generic competition. Market drivers include high TB burdens and MDR-TB treatment needs; constraints involve manufacturing complexity and regulatory variability.
Key Takeaways
- The global RIFAMPIN market was valued at approximately USD 300 million in 2022.
- Prices are expected to decline by about 3% annually through 2028, reaching approximately USD 0.29 per 300 mg capsule.
- Generics dominate the supply chain, with intense price competition.
- Patent expirations facilitated broad generic access, reducing costs.
- Regulatory, manufacturing, and market strategies should focus on cost control and compliance to sustain profitability.
FAQs
Q1: What factors influence RIFAMPIN's pricing beyond generic competition?
A: Regulatory approval costs, manufacturing efficiencies, formulations such as fixed-dose combinations, and supply chain logistics.
Q2: How does resistance impact RIFAMPIN demand?
A: Increasing MDR-TB cases elevate the need for optimized or alternative regimens, leading to sustained or increased demand.
Q3: Are there any new formulations or delivery mechanisms for RIFAMPIN?
A: Research focuses on long-acting injectables and combination pills to improve adherence, but such innovations are not yet widespread.
Q4: How is the global TB burden projected to influence RIFAMPIN sales?
A: Steady or rising TB cases, especially MDR-TB, sustain demand for RIFAMPIN-containing therapies.
Q5: What is the outlook for RIFAMPIN markets in emerging economies?
A: High TB prevalence and cost sensitivity favor increased generic use, but regulatory and infrastructural challenges can impede rapid market growth.
References
[1] Global Market Insights. (2022). Antibiotics Market Analysis.
[2] WHO. (2022). Global Tuberculosis Report.
[3] PatentScope. (2013). Timeline of Rifampin Patent Expirations.
[4] IMS Health. (2021). Global Generic Drug Market Dynamics.
[5] WHO. (2021). TB Data & Statistics.
[6] Pharma Price Index. (2018). Antibiotic Wholesale Prices.
[7] Global Drug Price Guide. (2020). Generic Antibiotic Pricing.
[8] Market Data Solutions. (2022). Current Antibiotic Pricing Trends.