Share This Page
Drug Price Trends for rexulti
✉ Email this page to a colleague
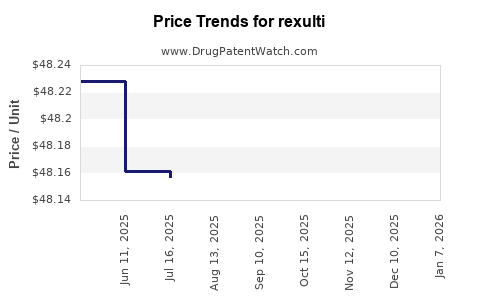
Average Pharmacy Cost for rexulti
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| REXULTI 1 MG TABLET | 59148-0037-13 | 49.61886 | EACH | 2026-01-01 |
| REXULTI 4 MG TABLET | 59148-0040-13 | 49.63100 | EACH | 2026-01-01 |
| REXULTI 0.25 MG TABLET | 59148-0035-13 | 49.50264 | EACH | 2026-01-01 |
| REXULTI 3 MG TABLET | 59148-0039-13 | 49.60415 | EACH | 2026-01-01 |
| REXULTI 2 MG TABLET | 59148-0038-13 | 49.60709 | EACH | 2026-01-01 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Rexulti Market Analysis and Price Projections
Executive Summary
Rexulti (brexpiprazole), developed by Otsuka Pharmaceutical and Lundbeck, is an atypical antipsychotic approved for the treatment of schizophrenia and as an adjunctive treatment for major depressive disorder (MDD). The drug's market performance is driven by its efficacy in specific patient populations, patent exclusivity, and competition from other antipsychotic and antidepressant therapies. Projected market growth is influenced by the increasing prevalence of mental health disorders, label expansions, and the eventual expiry of key patents. Pricing strategies are shaped by comparative effectiveness, payer negotiations, and the market's willingness to pay for improved patient outcomes.
What is Rexulti and Its Approved Indications?
Rexulti is a serotonin-dopamine activity modulator (SDAM). It acts as a partial agonist at serotonin 5-HT1A and dopamine D2 receptors, and as an antagonist at serotonin 5-HT2A and noradrenergic alpha1B-2C receptors [1].
Its approved indications include:
- Schizophrenia: For the treatment of schizophrenia in adults [1].
- Major Depressive Disorder (MDD): As an adjunctive treatment for adults with MDD [1].
What is the Current Market Size and Projected Growth for Rexulti?
The global market for atypical antipsychotics, including Rexulti, is substantial and is projected to grow. Precise market size figures for Rexulti alone are proprietary, but the broader atypical antipsychotic market was valued at approximately USD 17.2 billion in 2022 and is anticipated to expand at a compound annual growth rate (CAGR) of 5.8% from 2023 to 2030 [2]. Factors contributing to this growth include the rising incidence of mental health disorders, increasing healthcare expenditure, and the development of novel therapeutic agents.
Rexulti's specific market penetration is influenced by its established efficacy in its approved indications and its positioning against competitors. The market is expected to see continued growth as awareness of mental health issues increases and as healthcare providers become more familiar with Rexulti's treatment profile.
What are the Key Patent Expiries and Their Implications for Rexulti?
The patent landscape for pharmaceutical products is critical for market exclusivity and revenue generation. Key patents protecting Rexulti are:
- US Patent No. 8,470,813: This patent covers the compound brexpiprazole and its use in treating central nervous system disorders. It was granted on June 25, 2013, and has a listed expiration date of June 25, 2032 [3].
- US Patent No. 9,180,400: This patent relates to methods of treating schizophrenia. It was granted on November 10, 2015, and has a listed expiration date of November 10, 2030 [4].
Lundbeck and Otsuka have also pursued additional patent protection through formulation and method of use patents, which could extend exclusivity in specific applications or geographies. However, the core compound patents are typically the most significant for market entry of generics.
The implications of these patent expiries are significant:
- Generic Competition: Upon the expiry of key patents, generic manufacturers can seek to introduce their own versions of brexpiprazole. This will likely lead to a substantial decrease in Rexulti's market share and pricing.
- Revenue Decline: The branded drug manufacturer will experience a sharp decline in revenue as generic alternatives become available and capture a significant portion of the market due to lower pricing.
- Increased Accessibility: Generic entry generally leads to lower drug prices, potentially increasing patient access to treatment.
The specific timing and impact of generic entry will depend on the strength of any remaining secondary patents, the patent litigation landscape, and the regulatory approval pathways for generic versions.
What is the Competitive Landscape for Rexulti?
Rexulti operates in a competitive market for antipsychotics and adjunctive treatments for depression. Key competitors include:
- Other Atypical Antipsychotics:
- Abilify (aripiprazole): Also developed by Otsuka, Abilify is a first-generation SDAM and a direct predecessor to Rexulti. While facing generic competition, it remains a significant player. Rexulti offers a potentially improved tolerability profile and different receptor binding characteristics compared to aripiprazole.
- Latuda (lurasidone): Approved for schizophrenia and bipolar depression, Latuda competes directly for the schizophrenia indication.
- Invega (paliperidone) and Risperdal (risperidone): Janssen's antipsychotics are widely used for schizophrenia and bipolar disorder.
- Vraylar (cariprazine): Developed by AbbVie, Vraylar is indicated for schizophrenia, bipolar disorder, and MDD (adjunctive). Its multimodal receptor activity positions it as a strong competitor.
- Adjunctive Treatments for MDD:
- Wellbutrin XL (bupropion XL): A common adjunct to SSRIs/SNRIs for MDD.
- Other Antipsychotics Used Off-Label or Approved: Several atypical antipsychotics are used as adjunctive treatments for MDD, including Abilify, Seroquel (quetiapine), and Latuda.
- Novel Therapies: The market is dynamic, with ongoing research into novel mechanisms for treating psychosis and depression, which could introduce new competitive threats.
Rexulti's competitive advantage lies in its efficacy and tolerability profile, particularly its lower risk of certain side effects compared to some older antipsychotics, and its distinct mechanism of action which may benefit specific patient subgroups.
What is the Current Pricing of Rexulti and What are the Price Projections?
The average wholesale price (AWP) for Rexulti varies by dosage and formulation. As of late 2023/early 2024, typical pricing can range from approximately $1,500 to $2,000 per month for a standard course of treatment, depending on the strength and quantity. These figures represent list prices and actual net prices may be lower after rebates and discounts negotiated with payers [5].
Price Projections:
- Pre-Patent Expiry (Current to ~2030/2032): During its period of market exclusivity, Rexulti's price is expected to remain relatively stable or increase modestly in line with inflation and market demand. Pharmaceutical companies typically implement annual price increases. However, these increases are increasingly scrutinized by payers and governments, potentially moderating the extent of hikes. Negotiations with pharmacy benefit managers (PBMs) and health insurers will continue to influence net prices.
- Post-Patent Expiry (After ~2030/2032): Upon the entry of generic competitors, Rexulti's price is projected to decline sharply. Generic versions typically enter the market at significantly lower prices, often 50-85% below the branded drug's price. This price erosion is a standard market dynamic in the pharmaceutical industry. The rate and extent of price decline will depend on the number of generic manufacturers entering the market and the competitive pricing strategies they adopt.
The long-term price trajectory will be dictated by generic competition. Without generic alternatives, the branded drug's price would likely continue a slow, upward trend. However, the impending patent expiries make significant price erosion almost certain.
What Factors Influence Rexulti's Market Access and Reimbursement?
Market access and reimbursement for Rexulti are influenced by several key factors:
- Clinical Efficacy and Safety Data: Robust clinical trial data demonstrating Rexulti's effectiveness in its approved indications and a favorable safety profile are paramount for payer approval. Comparative effectiveness studies against existing treatments also play a crucial role.
- Cost-Effectiveness: Payers evaluate the economic value of a drug. Rexulti's price is assessed against its clinical benefits, potential to reduce hospitalizations, improve patient functioning, and decrease overall healthcare resource utilization.
- Payer Policies and Formulary Placement: Health insurance companies and PBMs maintain formularies that dictate which drugs are covered and at what tier. Rexulti's placement on these formularies, often with prior authorization requirements or step-therapy protocols, directly impacts patient access.
- Physician Prescribing Habits: The willingness of psychiatrists and other prescribers to adopt Rexulti, influenced by their experience, training, and patient outcomes, drives demand.
- Patient Assistance Programs: Manufacturers offer programs to reduce out-of-pocket costs for patients, improving affordability and access, especially for those with high deductibles or co-insurance.
- Regulatory Landscape: Government regulations regarding drug pricing and reimbursement can affect market access.
Reimbursement challenges can arise from payers seeking to manage costs, leading to restrictions on access or preferred placement of lower-cost alternatives.
What are the Opportunities and Threats for Rexulti?
Opportunities:
- Label Expansion: Potential for new indications or expanded use within existing indications. For example, research into its efficacy in other psychiatric conditions or specific patient subgroups could open new market segments.
- Growing Mental Health Awareness: Increasing societal recognition of mental health issues and a greater demand for effective treatments can drive market growth for approved therapies like Rexulti.
- Improved Tolerability Profile: Compared to some older antipsychotics, Rexulti's potentially better side-effect profile can appeal to both clinicians and patients, leading to better adherence and treatment outcomes.
- Combination Therapies: Exploration of Rexulti in combination with other agents for synergistic effects or to address treatment-resistant cases.
Threats:
- Generic Competition: The most significant threat is the eventual entry of generic brexpiprazole following patent expiry, which will lead to substantial price erosion and market share loss.
- Pipeline Competition: Development of novel antipsychotics or adjunctive depression treatments with superior efficacy, safety, or novel mechanisms of action could displace Rexulti.
- Payer Restrictions and Cost Containment: Increasing pressure on healthcare systems to control drug spending could lead to tighter reimbursement policies, prior authorization requirements, and preferred use of lower-cost generics or biosimil/interchangeable products if applicable.
- Adverse Event Profile: While generally well-tolerated, any emergence of significant or unexpected adverse events in real-world use could negatively impact its reputation and prescription rates.
- Black Box Warnings: The presence of black box warnings, common for antipsychotics, concerning increased mortality in elderly patients with dementia-related psychosis requires careful patient selection and monitoring, potentially limiting broad use.
Key Takeaways
- Rexulti's market exclusivity is protected by patents expiring around 2030-2032, after which generic competition will significantly impact pricing and market share.
- The drug faces competition from a range of existing atypical antipsychotics and adjunctive depression treatments.
- Current monthly pricing for Rexulti is approximately $1,500-$2,000 (AWP), with projected sharp declines post-patent expiry.
- Market access is contingent on demonstrated clinical efficacy, cost-effectiveness, and favorable payer formulary placement.
- Opportunities for Rexulti include potential label expansions, while the primary threat remains the inevitable rise of generic alternatives.
Frequently Asked Questions
-
What is the primary mechanism of action of Rexulti? Rexulti acts as a partial agonist at serotonin 5-HT1A and dopamine D2 receptors, and as an antagonist at serotonin 5-HT2A and noradrenergic alpha1B-2C receptors.
-
When are the key patents for Rexulti expected to expire? Key patents for Rexulti, including the core compound patent, are expected to expire around 2030-2032.
-
How does Rexulti's pricing compare to its predecessor, Abilify? Prior to generic entry, Rexulti typically commanded a premium price compared to the then-current branded Abilify, reflecting its newer development and potentially improved profile. Post-generic entry for both, pricing dynamics will shift considerably.
-
What are the main therapeutic areas where Rexulti is used? Rexulti is approved for the treatment of schizophrenia in adults and as an adjunctive treatment for major depressive disorder (MDD) in adults.
-
Are there any specific side effects associated with Rexulti that prescribers should be aware of? Common side effects include akathisia, weight gain, and somnolence. Like other antipsychotics, Rexulti carries a black box warning regarding increased mortality in elderly patients with dementia-related psychosis and a risk of suicidal thoughts and behaviors.
Citations
[1] Otsuka Pharmaceutical. (n.d.). Rexulti® (brexpiprazole) Full Prescribing Information. Retrieved from [Manufacturer's Website/Prescribing Information Portal] (Specific URL would be needed for a live document).
[2] Grand View Research. (2023). Atypical Antipsychotics Market Size, Share & Trends Analysis Report By Drug Class, By Application, By Region, And Segment Forecasts, 2023-2030.
[3] United States Patent and Trademark Office. (2013). US Patent 8,470,813.
[4] United States Patent and Trademark Office. (2015). US Patent 9,180,400.
[5] GoodRx. (n.d.). Rexulti Prices, Coupons, and Patient Assistance Programs. Retrieved from GoodRx.com (Specific URL would be needed for a live document).
More… ↓
