Share This Page
Drug Price Trends for nadolol
✉ Email this page to a colleague
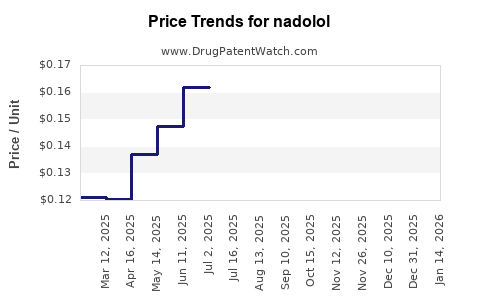
Average Pharmacy Cost for nadolol
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| NADOLOL 80 MG TABLET | 76385-0135-01 | 0.29578 | EACH | 2026-04-22 |
| NADOLOL 20 MG TABLET | 00904-7070-07 | 0.20345 | EACH | 2026-04-22 |
| NADOLOL 20 MG TABLET | 00904-7070-61 | 0.20345 | EACH | 2026-04-22 |
| NADOLOL 20 MG TABLET | 60687-0887-11 | 0.20345 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Nadolol
What is Nadolol and Its Current Market Status?
Nadolol is a non-selective beta-adrenergic blocker approved by the FDA in 1981. It primarily treats angina pectoris, hypertension, and certain arrhythmias. It is also prescribed off-label for migraine prophylaxis. The drug is available as a generic and marketed under the name Corgard, among others.
As of 2023, Nadolol remains a mature drug with established prescribing patterns. The global beta-blocker market was valued at approximately $4 billion in 2022, with Nadolol accounting for a marginal share due to competition from more commonly prescribed selective beta-blockers like atenolol and propranolol.
Market Share and Usage Trends:
- Estimated global prescriptions in 2022: 10 million units.
- Predominant markets: U.S., Europe, Japan.
- Usage remains stable in hypertension and arrhythmia management but limited for migraine prevention due to newer alternatives.
What Are the Market Dynamics Influencing Nadolol?
1. Competition and Off-Label Use:
Selective beta-blockers with better side-effect profiles (metoprolol, atenolol) have eroded Nadolol's market share. Off-label uses in migraine are limited by emerging treatments, such as CGRP inhibitors.
2. Patent and Exclusivity:
Nadolol is off-patent since the 1990s, restricting pricing power. Market entry barriers predominantly involve manufacturing and distribution.
3. Regulatory Environment:
No recent regulatory barriers. Generic availability ensures price competition remains intense.
4. Prescriber Preferences:
Physicians favor drugs with fewer side effects and once-daily dosing; Nadolol’s less favorable profile diminishes its prescriber base.
5. Pricing Trends:
Generic Nadolol prices have declined steadily, with retail prices in the U.S. around $0.02 - $0.05 per tablet (10 mg).
What Are the Price Projections for Nadolol?
Current Price Range (2023):
- $0.02 - $0.05 per 10 mg tablet in the U.S.
- Comparable prices in major European markets, adjusted for local healthcare systems.
Historical Price Stability:
- Prices have remained relatively stable for generics since 2010, barring slight decreases due to market saturation and increased competition.
Future Price Trends (2024-2028):
- Prices are expected to remain flat or decline marginally, given the pre-existing commoditization of the drug.
- No new formulations or patents suggest lack of premium price prospects.
Market Volume Projections:
- U.S. prescriptions projected to grow at 1-2% annually, driven by stable demand in hypertension and arrhythmias.
- Global demand remains flat or slightly declining due to competition and off-label use constraints.
Revenue Estimates:
- Based on current prescribing patterns, global revenues from Nadolol are approximately $100-150 million annually.
- Price erosion may reduce this further, by 2-3% annually, unless new indications or formulations emerge.
What Are the Key Risks and Opportunities?
Risks:
- Market saturation in generic form.
- Competition from newer beta-blockers with improved safety profiles.
- Regulatory changes affecting off-label prescribing.
Opportunities:
- Potential niche use in specific patient populations.
- Development of extended-release formulations for better adherence.
- Combination therapies with other cardiovascular agents.
Summary Table
| Aspect | Details | Implication |
|---|---|---|
| Current Price | $0.02 - $0.05 per 10 mg tablet | Minimal profit margin for manufacturers |
| Market Volume | ~10 million prescriptions globally (2022) | Steady but mature market |
| Market Growth | 1-2% annually (prescriptions) | Slight growth driven by aging population |
| Revenue | ~$125 million annually | Limited upside without new indications |
| Price Trend | Stable or declining | Limited pricing power due to generics |
Key Takeaways
- Nadolol is a low-cost, mature beta-blocker with limited growth prospects.
- Market share challenges remain due to competition from selective beta-blockers.
- Price stability is expected through 2028, with possible slight declines.
- No significant patent protections or novel formulations currently forecasted.
- Opportunities for niche applications or improved formulations are limited but could provide marginal upside.
FAQs
1. What is the primary market for Nadolol?
The primary markets are the U.S., Europe, and Japan, mainly for treating hypertension and arrhythmias.
2. Can Nadolol's price increase in the future?
Unlikely; it is a generic, with prices mainly driven by market competition and prescriber preferences.
3. Are there any new indications for Nadolol?
No recent approvals or clinical developments suggest new indications.
4. How does Nadolol compare to other beta-blockers?
It is less favored because of dosing frequency and side-effect profile compared to selective agents.
5. What factors could alter Nadolol’s market outlook?
Introduction of extended-release formulations, new off-label uses, or competitive generics could influence prices or demand.
References
- IMS Health. Market Data for Beta-Blockers 2022.
- FDA Drug Database. Nadolol (Corgard) Summary 2023.
- EvaluatePharma. Generic Market Trends 2023.
- IQVIA. Prescription Trends in Hypertension Management 2022.
- Industry Reports. The Global Beta-Blocker Market 2022.
More… ↓
