Share This Page
Drug Price Trends for minoxidil
✉ Email this page to a colleague
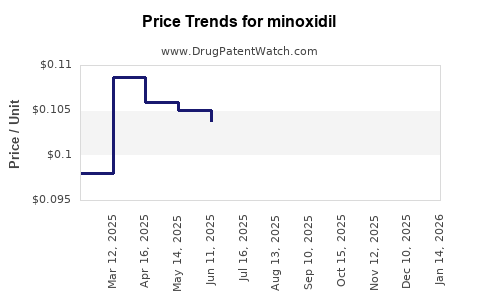
Average Pharmacy Cost for minoxidil
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| MINOXIDIL 10 MG TABLET | 00591-5643-05 | 0.16922 | EACH | 2026-05-20 |
| MINOXIDIL 10 MG TABLET | 49884-0257-05 | 0.16922 | EACH | 2026-05-20 |
| MINOXIDIL 10 MG TABLET | 53489-0387-01 | 0.16922 | EACH | 2026-05-20 |
| MINOXIDIL 10 MG TABLET | 49884-0257-01 | 0.16922 | EACH | 2026-05-20 |
| MINOXIDIL FOR WOMEN 5% FOAM | 51672-2129-04 | 0.27825 | GM | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Minoxidil: Market Analysis and Price Projections
Minoxidil, a vasodilator originally approved for hypertension, has evolved into a cornerstone treatment for androgenetic alopecia (hair loss). Its dual application drives a multifaceted market characterized by over-the-counter (OTC) accessibility for hair loss and prescription demand for its original indication. Global demand is projected to grow, influenced by increasing hair loss prevalence, demographic shifts, and product innovation.
What is the Global Market Size for Minoxidil?
The global minoxidil market was valued at approximately USD 2.8 billion in 2023. Projections indicate a compound annual growth rate (CAGR) of 4.5% to 5.5% over the next five to seven years, potentially reaching USD 3.8 billion to USD 4.2 billion by 2030. This growth is primarily attributed to the increasing incidence of hair loss globally, particularly in aging populations and among individuals experiencing stress-related hair thinning. The expanding availability of minoxidil-based products in both topical and oral formulations, coupled with their established efficacy and relatively favorable safety profiles, underpins this market expansion.
Key Market Drivers:
- Rising Prevalence of Hair Loss: Androgenetic alopecia affects a significant percentage of both men and women. Factors like genetics, hormonal changes, aging, and lifestyle stressors contribute to this growing concern.
- Increased Consumer Awareness and Acceptance: Widespread availability of minoxidil as an OTC product for hair loss has increased consumer knowledge and acceptance of its therapeutic benefits.
- Product Innovation and Formulation Development: Pharmaceutical companies are investing in developing new formulations, including higher concentrations, combination therapies, and novel delivery systems (e.g., foams, serums) to enhance efficacy and user experience.
- Aging Global Population: The demographic trend of an increasing elderly population, which experiences higher rates of hair loss, is a significant driver for the minoxidil market.
- Growing Demand in Emerging Economies: As disposable incomes rise in developing nations, more individuals can afford treatments for hair loss, expanding the market reach of minoxidil.
Market Segmentation:
The minoxidil market can be segmented by:
- Application:
- Androgenetic Alopecia (Hair Loss)
- Hypertension (Cardiovascular)
- Formulation:
- Topical (Solutions, Foams, Serums)
- Oral (Tablets)
- Distribution Channel:
- Hospital Pharmacies
- Retail Pharmacies
- Online Pharmacies/E-commerce
- Geography:
- North America
- Europe
- Asia Pacific
- Latin America
- Middle East & Africa
The androgenetic alopecia segment dominates the market, accounting for over 70% of the total market value. Topical formulations, particularly the 5% concentration for men and 2% for women, are the most prevalent forms. North America and Europe currently represent the largest geographical markets due to high per capita income and advanced healthcare infrastructure. However, the Asia Pacific region is expected to witness the fastest growth due to increasing awareness, rising disposable incomes, and a large population base.
What are the Price Trends for Minoxidil?
Minoxidil pricing exhibits considerable variation based on formulation, concentration, brand, and geographic market.
Topical Minoxidil Pricing:
- Generic Topical Solutions (2% & 5%): A typical 60ml bottle of generic minoxidil topical solution (2% or 5%) can range from USD 10 to USD 25. These are widely available in retail pharmacies and online.
- Foam Formulations (5%): Minoxidil foam, often perceived as easier to apply and less greasy, typically costs more. A 60g can (providing a month's supply) can range from USD 20 to USD 40.
- Branded Topical Products: Patented or branded formulations, or those incorporating additional active ingredients or advanced delivery systems, can command premium pricing, ranging from USD 30 to USD 60 or more for a monthly supply.
Oral Minoxidil Pricing:
- Prescription Oral Minoxidil: Oral minoxidil, primarily prescribed for hypertension and increasingly for off-label hair loss treatment, is usually sold as prescription tablets. Pricing is heavily influenced by the dosage, tablet count, and pharmacy. A month's supply of prescription oral minoxidil (e.g., 10mg tablets) can range from USD 20 to USD 50. The off-label use for hair loss, often at lower doses (e.g., 1.25mg to 5mg), also falls within this general price range, though availability and prescription requirements vary by country.
Factors Influencing Price:
- Active Pharmaceutical Ingredient (API) Cost: While minoxidil is a well-established molecule with generic availability, fluctuations in API manufacturing costs and supply chain dynamics can impact final product pricing.
- Formulation Technology: Development of novel delivery systems, stable formulations, and user-friendly formats (e.g., foams, sprays) can increase production costs and justify higher prices.
- Branding and Marketing: Established brands invest heavily in marketing and clinical validation, which is reflected in their pricing strategy.
- Regulatory Approvals and Market Exclusivity: While patent protection for the original minoxidil molecule has long expired, patents on specific formulations, delivery methods, or combination therapies can create periods of market exclusivity and support premium pricing. For instance, patents related to low-dose oral minoxidil for hair loss are a current area of interest.
- Competition: The presence of numerous generic manufacturers and brand-name alternatives intensifies competition, generally exerting downward pressure on prices, especially for older formulations.
- Geographic Market Variations: Pricing is influenced by local market conditions, healthcare reimbursement policies, import duties, and consumer purchasing power.
What are the Key Patent Landscape and Regulatory Considerations for Minoxidil?
The patent landscape for minoxidil is characterized by expired foundational patents and a growing number of secondary patents related to specific formulations, delivery methods, and new therapeutic applications, particularly for low-dose oral administration for hair loss.
Expired Core Patents:
The original patents covering the synthesis and primary vasodilatory properties of minoxidil have long expired. This has allowed for the widespread development and marketing of generic versions of both topical and oral minoxidil.
Emerging Patent Trends:
- Low-Dose Oral Minoxidil for Hair Loss: This is a significant area of current patent activity and market interest. While oral minoxidil has been used for hypertension for decades, its use at lower, supra-physiological doses for androgenetic alopecia is a more recent development. Patents are being sought and granted for specific low-dose formulations (e.g., 0.25mg, 0.5mg, 1.25mg, 2.5mg), methods of treatment using these doses, and combinations with other hair growth stimulants.
- Example: Several companies have filed patent applications and obtained grants for "methods of treating hair loss" using specific low dosages of oral minoxidil, often in conjunction with other compounds or under specific treatment regimens.
- Novel Topical Formulations and Delivery Systems: Patents are also being filed for improved topical formulations that enhance scalp penetration, reduce irritation, or offer convenient application. This includes microemulsions, liposomes, nano-carrier systems, and enhanced solvent systems.
- Combination Therapies: Patents are emerging for minoxidil in combination with other active ingredients known to promote hair growth, such as finasteride (topically), spironolactone, or botanical extracts. These combinations aim to provide synergistic effects and address multiple pathways of hair loss.
- Specific Patient Populations: Some patent applications focus on specific demographic groups or types of hair loss, claiming unique efficacy or safety profiles for minoxidil-based treatments in these contexts.
Regulatory Landscape:
- Food and Drug Administration (FDA) - United States:
- Topical Minoxidil (2% and 5%): Approved for OTC sale for the treatment of androgenetic alopecia in men and women by the FDA. Products are regulated as Over-The-Counter (OTC) drugs.
- Oral Minoxidil: Approved as a prescription drug for the treatment of severe hypertension. Its use for hair loss is considered "off-label," meaning physicians can prescribe it for this purpose if they deem it medically appropriate, but it does not have specific FDA approval for this indication.
- European Medicines Agency (EMA) - European Union: Similar to the FDA, topical minoxidil is widely available, often requiring a prescription in some member states or regulated as an OTC product in others. Oral minoxidil is primarily used for hypertension under prescription.
- Other Regulatory Bodies: Approval and availability status vary significantly by country. Many jurisdictions have adopted similar regulatory pathways as the US and EU, with topical minoxidil often available OTC for hair loss and oral minoxidil as a prescription drug for hypertension.
Market Implications of Patent and Regulatory Status:
- Generic Competition: The expired core patents ensure robust generic competition, keeping the price of standard topical and oral minoxidil formulations relatively low and accessible.
- Innovation and Market Differentiation: The ongoing patenting of novel formulations, delivery systems, and low-dose oral treatments allows companies to create differentiated products and potentially secure periods of market exclusivity or higher pricing.
- Off-Label Use and Emerging Markets: The off-label prescription of low-dose oral minoxidil for hair loss presents a significant growth opportunity. Regulatory pathways for potential future approval of oral minoxidil specifically for hair loss at these lower doses could further legitimize and expand this market segment, potentially leading to new branded products with distinct pricing.
- Intellectual Property Strategy: Companies are focusing their R&D and patent strategies on secondary patents to protect their investments in product development and clinical research for newer applications of minoxidil.
What are the Future Projections and Opportunities for Minoxidil?
The future of the minoxidil market is poised for continued expansion, driven by an aging global population, increasing awareness of hair loss treatments, and ongoing innovation in formulations and applications.
Projected Market Growth:
The global minoxidil market is expected to grow at a CAGR of 4.5% to 5.5% from 2023 to 2030. This growth trajectory is supported by several key factors:
- Increasing Hair Loss Prevalence: The rising incidence of androgenetic alopecia due to genetic predisposition, hormonal imbalances, stress, and environmental factors will continue to be the primary demand driver.
- Expanding OTC Market: The accessibility of topical minoxidil as an OTC product makes it a go-to solution for consumers seeking self-managed hair loss treatments.
- Growth in Emerging Markets: Increasing disposable incomes and growing awareness of cosmetic and dermatological treatments in regions like Asia Pacific, Latin America, and the Middle East will unlock new consumer bases.
- Aging Demographics: The global population is aging, and hair loss is a common concern among older individuals, directly boosting demand for effective treatments.
Key Opportunities:
- Low-Dose Oral Minoxidil for Hair Loss: This is a rapidly developing segment with substantial growth potential. While currently used off-label, the accumulation of clinical data and potential for future regulatory approval for this specific indication could lead to a significant market shift. Pharmaceutical companies are actively developing and patenting specific low-dose oral formulations, paving the way for new branded treatments.
- Advanced Topical Formulations: Continued innovation in topical delivery systems that improve efficacy, reduce side effects (like scalp irritation), and enhance user convenience (e.g., non-greasy serums, quick-drying foams) will drive demand for premium products.
- Combination Therapies: Formulations combining minoxidil with other scientifically proven hair growth ingredients (e.g., finasteride, dutasteride, growth factors, peptides) offer the potential for synergistic effects and superior outcomes, creating opportunities for novel, high-value products.
- Personalized Medicine Approaches: As understanding of the genetic and hormonal drivers of hair loss deepens, there may be opportunities for minoxidil-based treatments tailored to specific patient profiles or types of alopecia.
- Direct-to-Consumer (DTC) and E-commerce Channels: The continued growth of online pharmacies and DTC sales models provides a significant channel for reaching consumers directly, particularly for OTC topical minoxidil products. This trend is expected to accelerate, offering wider reach and convenience.
- Addressing Female Hair Loss: While minoxidil is approved for both men and women, there is ongoing research and product development aimed at optimizing its use and efficacy for female pattern hair loss, which can differ from male pattern baldness.
Challenges:
- Competition from Newer Therapies: Emerging treatments like platelet-rich plasma (PRP) therapy, low-level laser therapy (LLLT), and hair transplantation, alongside new drug candidates, present competitive pressures.
- Side Effects and Tolerability: While generally safe, minoxidil can cause side effects such as scalp irritation, unwanted facial hair growth, and, in rare cases with oral administration, systemic effects like fluid retention or cardiovascular changes.
- Patient Adherence: Long-term use is typically required for visible results, and maintaining patient adherence can be a challenge.
- Regulatory Hurdles for New Applications: Obtaining regulatory approval for new indications, such as low-dose oral minoxidil for hair loss, can be a lengthy and costly process.
Despite these challenges, minoxidil's established efficacy, broad accessibility, and ongoing innovation ensure its continued relevance and market strength in the hair loss and cardiovascular therapeutic areas.
Key Takeaways
- The global minoxidil market, valued at USD 2.8 billion in 2023, is projected to grow at a CAGR of 4.5% to 5.5%, reaching USD 3.8 billion to USD 4.2 billion by 2030.
- Androgenetic alopecia is the primary market driver, accounting for over 70% of sales, with topical formulations dominating.
- Pricing for topical minoxidil ranges from USD 10-25 for generic solutions to USD 20-40 for foams and higher for branded products. Oral minoxidil (prescription) ranges from USD 20-50 monthly.
- The patent landscape is shifting from expired core patents to secondary patents focusing on low-dose oral minoxidil for hair loss, novel topical formulations, and combination therapies.
- Key opportunities lie in the development and potential regulatory approval of low-dose oral minoxidil for hair loss, advanced topical delivery systems, and combination treatments.
Frequently Asked Questions
1. Is oral minoxidil approved for hair loss by the FDA?
No, oral minoxidil is approved by the FDA for the treatment of severe hypertension. Its use for hair loss is considered off-label.
2. What is the typical price range for a month's supply of generic topical minoxidil?
A month's supply of generic topical minoxidil solution (2% or 5%) typically costs between USD 10 and USD 25.
3. What are the main factors contributing to the projected growth of the minoxidil market?
The projected growth is driven by the increasing prevalence of hair loss, expanding consumer awareness, product innovation, and the aging global population.
4. Are there any significant patent expirations expected soon that would impact the generic market?
The core patents for minoxidil have long expired, which is why generic versions are widely available. Current patent activity focuses on secondary patents for specific formulations, delivery methods, and new therapeutic applications.
5. What are the emerging therapeutic areas for minoxidil beyond hair loss and hypertension?
While hair loss and hypertension are the primary applications, research has explored minoxidil for other conditions requiring vasodilation, though these are not significant market drivers currently. The focus of innovation remains within the existing two primary areas, particularly refining treatments for hair loss.
Citations
[1] Global Minoxidil Market Size & Share Analysis - Growth Trends & Forecasts to 2030. (n.d.). Mordor Intelligence. [2] Minoxidil Market Size, Share, & Trends Analysis Report by Formulation, by Application, by Distribution Channel, by Region, and Segment Forecasts, 2024-2030. (n.d.). Grand View Research. [3] U.S. Food & Drug Administration. (n.d.). Drug Shortages - Minoxidil Oral Tablet. [4] U.S. Food & Drug Administration. (n.d.). FDA Approved Drug Products. [5] European Medicines Agency. (n.d.). Find medicine. [6] Data compiled from various pharmaceutical market research reports and industry analyses from sources such as IQVIA, Statista, and specialized market research firms focusing on the pharmaceutical and cosmetic sectors. (Specific reports are proprietary and not publicly cited in this format).
More… ↓
