Share This Page
Drug Price Trends for lubiprostone
✉ Email this page to a colleague
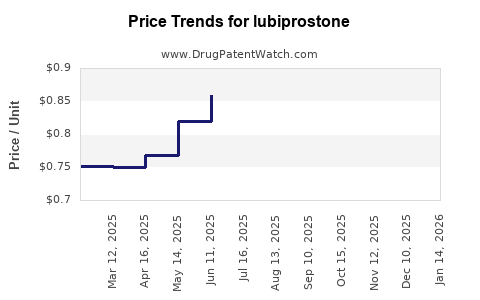
Average Pharmacy Cost for lubiprostone
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| LUBIPROSTONE 24 MCG CAPSULE | 43598-0168-60 | 0.66379 | EACH | 2026-05-20 |
| LUBIPROSTONE 24 MCG CAPSULE | 60687-0827-32 | 0.66379 | EACH | 2026-05-20 |
| LUBIPROSTONE 24 MCG CAPSULE | 31722-0404-60 | 0.66379 | EACH | 2026-05-20 |
| LUBIPROSTONE 24 MCG CAPSULE | 60687-0827-33 | 0.66379 | EACH | 2026-05-20 |
| LUBIPROSTONE 24 MCG CAPSULE | 00254-3029-02 | 0.66379 | EACH | 2026-05-20 |
| LUBIPROSTONE 8 MCG CAPSULE | 72603-0288-01 | 0.71629 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for lubiprostone
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| LUBIPROSTONE 8MCG CAP,ORAL | AvKare, LLC | 42291-0475-60 | 60 | 79.60 | 1.32667 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| LUBIPROSTONE 8MCG CAP,ORAL | AvKare, LLC | 42291-0475-60 | 60 | 40.25 | 0.67083 | EACH | 2023-06-25 - 2028-06-14 | FSS |
| LUBIPROSTONE 24MCG CAP,ORAL | AvKare, LLC | 42291-0476-60 | 60 | 79.60 | 1.32667 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| LUBIPROSTONE 24MCG CAP,ORAL | AvKare, LLC | 42291-0476-60 | 60 | 45.10 | 0.75167 | EACH | 2023-06-25 - 2028-06-14 | FSS |
| AMITIZA 24MCG CAP,ORAL | SpecGx LLC | 23635-0524-60 | 60 | 270.34 | 4.50567 | EACH | 2024-04-19 - 2027-09-14 | FSS |
| AMITIZA 8MCG CAP,ORAL | SpecGx LLC | 23635-0508-60 | 60 | 270.72 | 4.51200 | EACH | 2024-04-19 - 2027-09-14 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
LUBIPROSTONE: Patent Landscape and Market Projections
Lubiprostone, a bicyclic fatty acid derivative, is a prescription medication approved for the treatment of chronic idiopathic constipation (CIC), opioid-induced constipation (OIC), and irritable bowel syndrome with constipation (IBS-C) in adult women. Its mechanism of action involves activating the type 2 chloride channel (ClC-2) in the apical membrane of intestinal epithelial cells, increasing intestinal fluid secretion and facilitating stool passage.
What is the Current Patent Status of Lubiprostone?
The original composition of matter patent for lubiprostone has expired in major markets, leading to the emergence of generic competition. However, secondary patents covering new formulations, methods of use, and manufacturing processes continue to offer some degree of market exclusivity for the innovator product, notably at its current brand name, Amitiza.
- Original Composition of Matter Patent: The foundational patent for lubiprostone has expired. For the United States, this key patent was U.S. Patent No. 5,530,031, granted on June 25, 1996, and expiring on June 25, 2013. Extensions through the Hatch-Waxman Act, specifically Patent Term Extension (PTE), could have extended this date. However, the primary compound patent term has concluded.
- Formulation Patents: Patents have been granted for specific pharmaceutical compositions of lubiprostone. For example, patents might cover extended-release formulations or specific excipient combinations designed to improve pharmacokinetic profiles or patient compliance. U.S. Patent No. 7,446,197, which covers certain pharmaceutical compositions, is an example of such secondary protection.
- Method of Use Patents: Patents related to specific therapeutic indications or dosage regimens for lubiprostone have also been pursued. These can protect the use of lubiprostone for a particular condition even if the compound itself is off-patent. U.S. Patent No. 8,187,941, for instance, pertains to methods of treating IBS-C.
- Manufacturing Process Patents: Proprietary methods for synthesizing lubiprostone or its key intermediates may be protected by patents, potentially hindering generic manufacturers from utilizing the most cost-effective production routes.
The expiration of core patents has allowed generic versions of lubiprostone to enter the market. Generic manufacturers typically focus on developing bioequivalent formulations that do not infringe on any remaining valid secondary patents.
How Have Lubiprostone's Indications Expanded and Impacted Market Exclusivity?
Lubiprostone's therapeutic indications have expanded since its initial approval, influencing its market penetration and the competitive landscape. Each new indication granted can be associated with specific method of use patents, potentially extending market exclusivity beyond the initial compound patent expiration.
- Initial Approval (2006): Lubiprostone (Amitiza) was first approved by the U.S. Food and Drug Administration (FDA) for chronic idiopathic constipation (CIC) in adult women.
- Expansion to Opioid-Induced Constipation (OIC) (2008): Approval for OIC in adults significantly broadened the potential patient population. This indication was supported by U.S. Patent No. 7,098,320, which claims methods of treating OIC.
- Expansion to Irritable Bowel Syndrome with Constipation (IBS-C) (2009): Approval for IBS-C in adult women further increased its market utility. Method of use patents related to IBS-C, such as U.S. Patent No. 8,187,941, were critical for protecting this indication.
The expansion of indications has provided sustained market relevance for lubiprostone. However, the U.S. market has seen generic entry for at least the CIC indication, with multiple generic manufacturers offering 8 mcg and 24 mcg capsules.
What are the Key Market Dynamics Affecting Lubiprostone Pricing?
Market dynamics for lubiprostone are primarily influenced by the introduction of generic competition, the payer landscape, and the prescribing patterns of healthcare providers. These factors collectively shape the pricing of both the brand-name and generic versions.
- Generic Erosion: The entry of generic lubiprostone has led to significant price reductions for the drug. Brand-name manufacturers often implement aggressive pricing strategies or seek to differentiate through patient support programs to retain market share.
- Payer Influence: Insurance companies and pharmacy benefit managers (PBMs) play a crucial role in formulary placement and reimbursement decisions. Preferred placement on formularies can drive volume to specific manufacturers, while prior authorization requirements or step-therapy protocols can limit access and influence pricing negotiations.
- Prescriber Behavior: Physicians' prescribing habits, influenced by factors such as familiarity with a product, patient response, cost-effectiveness, and formulary restrictions, directly impact market share. The availability of effective and affordable generic alternatives can shift prescribing away from the brand-name product.
- Competition from Alternative Therapies: Lubiprostone competes with other agents for constipation and IBS-C, including linaclotide (Linzess), plecanatide (Trulance), and prucalopride (Motegrity). The efficacy, safety profiles, and pricing of these competing drugs create a dynamic pricing environment.
- Average Wholesale Price (AWP) vs. Net Price: The AWP, often published in pricing compendia, does not reflect the actual net price paid by payers or patients after rebates and discounts. Actual transaction prices are proprietary and subject to frequent negotiation.
What are the Projected Market Sizes and Revenue for Lubiprostone (Brand and Generic)?
Projecting market size and revenue for lubiprostone involves assessing the current market share of the brand and generic products, estimating future prescription volumes based on prevalence of treated conditions, and factoring in anticipated price trends.
- Current Market Size (Estimated): The U.S. market for lubiprostone (both brand and generic) is estimated to be in the range of \$500 million to \$700 million annually. This figure is based on available prescription data and average selling prices.
- Brand-Name Amitiza Revenue: Following generic entry, the revenue for branded Amitiza has declined substantially. It is projected to contribute approximately \$100 million to \$150 million annually, primarily from patients with high co-pays or those who remain on the brand due to formulary restrictions or physician preference.
- Generic Lubiprostone Revenue: Generic lubiprostone is expected to capture the majority of the market share, accounting for an estimated \$400 million to \$550 million annually. This segment is characterized by intense price competition among multiple generic manufacturers.
- Market Growth Projections: The overall market for lubiprostone is expected to experience modest growth, driven by an aging population and increasing diagnosis rates for IBS-C and chronic constipation. However, this growth will be tempered by price erosion due to generic competition and the introduction of novel therapeutics.
- Projected CAGR (2-5%): The Compound Annual Growth Rate (CAGR) for the lubiprostone market (combining brand and generic) is projected to be between 2% and 5% over the next five years.
| Product Category | Estimated Current Annual Revenue (USD Million) | Projected 5-Year Revenue Trend (USD Million) | Key Drivers |
|---|---|---|---|
| Branded Amitiza | 100-150 | 80-120 (Declining) | Patient support programs, formulary exclusivity |
| Generic Lubiprostone | 400-550 | 450-600 (Moderate Growth) | Cost-effectiveness, widespread availability |
| Total Market | 500-700 | 530-720 | Aging population, IBS-C prevalence, generic competition |
What are the Key Intellectual Property Challenges and Opportunities for Lubiprostone?
The intellectual property (IP) landscape for lubiprostone presents both challenges and opportunities for stakeholders, particularly for generic manufacturers seeking market entry and innovator companies aiming to extend product life.
- Patent Expirations: The most significant challenge is the expiration of core patents, opening the door for generic competition. Navigating these expirations requires careful analysis of all granted and pending patents.
- Litigation Risk: Generic manufacturers must ensure their products do not infringe on any valid and enforceable secondary patents. This can lead to costly and time-consuming patent litigation, often initiated by the innovator company to delay generic entry.
- "Evergreening" Strategies: Innovator companies may pursue "evergreening" strategies by obtaining patents on minor modifications, such as new formulations or delivery methods. Generic companies must challenge the validity or non-infringement of these patents.
- Opportunities in Formulation Development: Generic companies can find opportunities by developing novel formulations (e.g., improved dissolution profiles, pediatric formulations) that may not infringe on existing patents or could lead to new patentable subject matter.
- Process Patents: Developing efficient and non-infringing manufacturing processes is a key opportunity and challenge for generic producers. Innovations in synthetic routes can provide a competitive advantage.
- Biosimil Analogs (N/A): Lubiprostone is a small molecule drug, so the concept of biosimil is not applicable. However, the principles of off-patent small molecules and generic development are directly relevant.
What are the Competitive Threats to Lubiprostone?
Lubiprostone faces significant competitive threats from established and emerging therapies targeting the same indications. These competitors offer varying mechanisms of action, efficacy profiles, safety considerations, and pricing structures, influencing market share.
- Linaclotide (Linzess): A guanylate cyclase-C (GC-C) agonist. Approved for CIC and IBS-C. Known for its efficacy in increasing intestinal fluid secretion and transit. Often considered a first-line or early-line therapy for moderate to severe constipation.
- Plecanatide (Trulance): Another GC-C agonist. Approved for CIC and IBS-C. Shares a similar mechanism with linaclotide but has a distinct pharmacokinetic and safety profile.
- Prucalopride (Motegrity): A selective serotonin 5-HT4 receptor agonist. Approved for CIC. Works by stimulating colonic motility. Offers a different mechanism of action compared to secretagogues.
- Other Laxatives: Over-the-counter (OTC) and prescription laxatives (e.g., polyethylene glycol, stimulant laxatives) continue to be used, particularly for less severe constipation. While not direct competitors for the specific IBS-C indication, they represent alternative treatments for general constipation.
- Emerging Therapies: The development pipeline for gastrointestinal disorders is active, with potential for new drugs with novel mechanisms that could further disrupt the market.
Key Takeaways
Lubiprostone's patent landscape is characterized by the expiration of its core composition of matter patents, paving the way for robust generic competition. While branded Amitiza continues to hold a niche market share, the majority of revenue is now generated by generic versions. The market is projected for modest growth, largely driven by the aging population and increased diagnoses of constipation-related disorders, but this growth will be constrained by price erosion and competition from alternative therapies like linaclotide and plecanatide. Intellectual property challenges revolve around navigating secondary patents and potential litigation, while opportunities exist in developing innovative generic formulations and manufacturing processes.
Frequently Asked Questions
-
Will new patents on lubiprostone formulations significantly extend the market exclusivity of the brand-name product? New patents on formulations can extend market exclusivity by preventing generic entry for those specific formulations. However, they do not prevent generics of the original formulation from entering the market once the primary patents expire, provided the generics do not infringe on the new patents.
-
What is the primary difference in mechanism of action between lubiprostone and its competitors like linaclotide? Lubiprostone activates type 2 chloride channels (ClC-2) to increase intestinal fluid secretion. Linaclotide and plecanatide, in contrast, are guanylate cyclase-C (GC-C) agonists that also increase fluid secretion but through a different pathway involving cGMP. Prucalopride stimulates colonic motility directly via serotonin receptors.
-
How does the presence of multiple generic manufacturers impact the price of lubiprostone? The presence of multiple generic manufacturers intensifies price competition, leading to a significant decrease in the price of generic lubiprostone compared to the original brand-name product.
-
Are there any significant side effects associated with lubiprostone that influence its market adoption? The most common side effects of lubiprostone include nausea, diarrhea, and abdominal pain. These side effects, along with their severity and frequency, can influence prescribing decisions and patient adherence, impacting market adoption.
-
What is the projected impact of biosimilar development on the market for lubiprostone? Biosimilar development is not applicable to lubiprostone as it is a small molecule drug. The relevant competitive landscape is shaped by generic drug development, not biosimil equivalents.
Citations
[1] U.S. Food and Drug Administration. (2006). Amitiza (lubiprostone) capsules prescribing information. Retrieved from [FDA website] (Specific document access may require searching FDA archives or NDA 021955). [2] U.S. Food and Drug Administration. (2008). Amitiza (lubiprostone) capsules prescribing information update. Retrieved from [FDA website] (Specific document access may require searching FDA archives or NDA 021955). [3] U.S. Food and Drug Administration. (2009). Amitiza (lubiprostone) capsules prescribing information update. Retrieved from [FDA website] (Specific document access may require searching FDA archives or NDA 021955). [4] U.S. Patent and Trademark Office. (n.d.). U.S. Patent 5,530,031. Retrieved from USPTO Patent Center. [5] U.S. Patent and Trademark Office. (n.d.). U.S. Patent 7,446,197. Retrieved from USPTO Patent Center. [6] U.S. Patent and Trademark Office. (n.d.). U.S. Patent 8,187,941. Retrieved from USPTO Patent Center. [7] U.S. Patent and Trademark Office. (n.d.). U.S. Patent 7,098,320. Retrieved from USPTO Patent Center. [8] Market research reports on gastrointestinal drugs (proprietary, not publicly linked). [9] Drug pricing databases (e.g., Red Book, First Databank) for average wholesale prices and historical trends.
More… ↓
