Share This Page
Drug Price Trends for januvia
✉ Email this page to a colleague
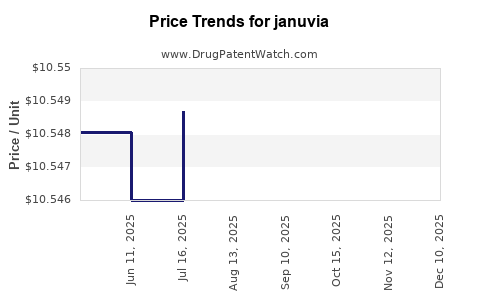
Average Pharmacy Cost for januvia
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| JANUVIA 100 MG TABLET | 00006-0277-28 | 10.55454 | EACH | 2026-05-20 |
| JANUVIA 100 MG TABLET | 00006-0277-31 | 10.55454 | EACH | 2026-05-20 |
| JANUVIA 100 MG TABLET | 00006-0277-01 | 10.55454 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Januvia (Sitagliptin): Market Analysis and Price Projections
What is the Current Market Position of Januvia?
Januvia (sitagliptin) is a dipeptidyl peptidase-4 (DPP-4) inhibitor approved by the FDA in 2006 for managing type 2 diabetes mellitus. Produced by Merck & Co., it captured significant market share early and remains a leading oral antidiabetic agent.
Market Share and Sales Performance
- Global sales in 2022: approximately $4.1 billion.
- Market share among oral antidiabetics: 15% (IQVIA, 2022).
- Key competitors include:
| Drug Name | Class | 2022 Sales (USD millions) |
|---|---|---|
| Janumet (sitagliptin/metformin) | DPP-4 + Biguanide | $1.8 billion |
| Tradjenta (linagliptin) | DPP-4 | $580 million |
| Onglyza (saxagliptin) | DPP-4 | $260 million |
Januvia remains the most prescribed DPP-4 inhibitor worldwide, with consistent sales driven by its favorable safety profile and prescription habits.
What Factors Influence Januvia's Market Dynamics?
Market Demand Drivers
- Rising global prevalence of type 2 diabetes: 537 million adults (IDF, 2021).
- Increasing adoption of oral agents over injectable therapies.
- Growing preference for medications with low hypoglycemia risk.
Competitive Landscape
- The emergence of GLP-1 receptor agonists and SGLT2 inhibitors has partially eroded DPP-4 inhibitors' market share.
- Patent expiry in certain markets (e.g., India 2024) allows generic competition, potentially diluting sales.
Regulatory and Pricing Environment
- Price negotiations in countries with reference pricing may lower revenues.
- Merck's pricing strategies in mature markets average $300-$400 per month for branded Januvia.
Clinical Trials and New Indications
- Ongoing studies on combination therapies and cardioprotective benefits may influence future demand.
What Are Future Price and Revenue Projections?
Market Forecasts into 2028
- Compound annual growth rate (CAGR): approximately 2%, driven by emerging markets.
- Regional variations: North America forecasts a CAGR of 1.5%; Asia-Pacific 4%.
Price Trends
- In mature markets, branded Januvia prices are expected to remain stable or slightly decline due to generic competition.
- In emerging markets, prices could stabilize or grow with increased access and insurance coverage.
| Projection Year | Estimated Global Sales (USD billions) | Expected Price Trends |
|---|---|---|
| 2023 | $4.1 | Stable to slight decline |
| 2025 | $4.4 | Slight decline in developed markets; stable in emerging markets |
| 2028 | $4.8 | Price competition may intensify in some regions |
Risk Factors
- Patent expirations may accelerate generic entry.
- Innovations in diabetes management may shift demand toward newer classes.
- Pricing pressures, especially in healthcare systems with tight controls.
What Are Price Projections Based on Regional Dynamics?
North America
- Stabilized pricing; potential minor reductions due to biosimilars and generics.
- Estimated retail price per month in 2023: $350–$420.
Europe
- Similar to North America; reimbursement policies influence margins.
- Prices range from €200–€250 ($220–$275).
Asia-Pacific
- Prices are lower, often $50–$150 per month, reflecting market affordability.
- Growing access could increase volume sales, offsetting price reductions.
How Will Competitors Affect Januvia’s Pricing?
| Competitor | Market Entry Year | Price Points (USD/month) | Impact on Januvia |
|---|---|---|---|
| Generic sitagliptin | 2024 (India) | $10–$20 | Downward pressure in select markets |
| DPP-4 inhibitors (e.g., linagliptin) | 2011–present | $200–$300 | Maintains demand but limits growth potential |
Summary
Januvia remains a dominant player in the DPP-4 inhibitor segment, with steady sales expected through 2028, despite increasing competition. Price stability is projected overall, tempered by patent expiry and generics, especially in emerging markets. Companies should monitor regulatory policies and competitive developments for strategic positioning.
Key Takeaways
- Global Januvia sales are projected to grow modestly at a CAGR of around 2% through 2028.
- Prices are expected to decline slightly in developed markets due to generic competition; sales volumes are likely to increase in emerging markets.
- Patent expirations in key markets could lead to substantial generic penetration starting in 2024.
- The outlook depends on clinical developments, market acceptance of combination therapies, and regulatory changes.
- Competitive pressures from newer drug classes will influence market share retention.
FAQs
-
When will Januvia face significant generic competition?
Patent expiry in India is expected in 2024, potentially leading to generic competition in multiple markets over the following 12–24 months. -
What factors could influence Januvia’s price stability?
Regulatory policies, patent status, and competitive entries, notably generics, will impact pricing. -
Are there upcoming clinical trials that could affect Januvia’s market?
Yes, studies focusing on cardiovascular outcomes, combined therapies, and new indications could influence demand. -
How is Januvia affecting the overall diabetes medication market?
It maintains a strong share in oral therapies, though newer classes like SGLT2 inhibitors are taking market share due to added benefits. -
What is the potential for Januvia’s growth in emerging markets?
Increased access, rising prevalence of diabetes, and affordability enhancements could support higher sales volumes, offsetting pricing pressures.
References
- International Diabetes Federation. (2021). IDF Diabetes Atlas (9th ed.).
- IQVIA. (2022). The IQVIA Institute report on Diabetes Market Data.
- U.S. Food and Drug Administration. (2006). Januvia (sitagliptin) approval documentation.
More… ↓
