Share This Page
Drug Price Trends for itraconazole
✉ Email this page to a colleague
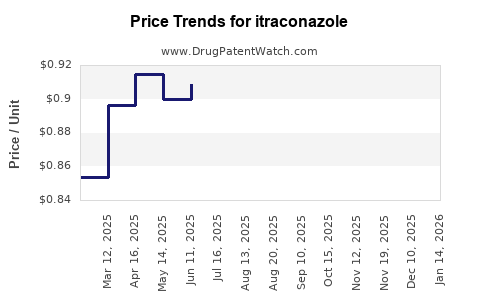
Average Pharmacy Cost for itraconazole
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ITRACONAZOLE 10 MG/ML SOLUTION | 65162-0087-74 | 1.18034 | ML | 2026-03-18 |
| ITRACONAZOLE 10 MG/ML SOLUTION | 31722-0006-31 | 1.18034 | ML | 2026-03-18 |
| ITRACONAZOLE 100 MG CAPSULE | 67877-0454-30 | 0.83842 | EACH | 2026-03-18 |
| ITRACONAZOLE 100 MG CAPSULE | 10147-1700-03 | 0.83842 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for itraconazole
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| ITRACONAZOLE 50MG/5ML SOLN,ORAL | AvKare, LLC | 65162-0087-74 | 150 | 211.40 | 1.40933 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| ITRACONAZOLE 100MG CAP | Johnson & Johnson Health Care Systems, Inc. obo Patriot Pharm | 10147-1700-03 | 30 | 59.71 | 1.99033 | EACH | 2024-01-01 - 2027-12-31 | FSS |
| ITRACONAZOLE 50MG/5ML SOLN,ORAL | Johnson & Johnson Health Care Systems, Inc. obo Patriot Pharm | 10147-0150-01 | 150ML | 88.54 | 0.59027 | ML | 2023-01-01 - 2027-12-31 | FSS |
| ITRACONAZOLE 50MG/5ML SOLN,ORAL | Johnson & Johnson Health Care Systems, Inc. obo Patriot Pharm | 10147-0150-01 | 150ML | 83.17 | 0.55447 | ML | 2024-01-01 - 2027-12-31 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
ITRACONAZOLE: PATENT LANDSCAPE AND MARKET PROJECTIONS
Itraconazole, an azole antifungal agent, faces a complex patent expiration trajectory. Its primary market indications, including onychomycosis and invasive aspergillosis, represent a substantial but maturing segment of the antifungal market. Generic competition has intensified post-2012, impacting branded product pricing and market share. Future market value will be influenced by the emergence of novel antifungal agents and the development of new indications or delivery methods for itraconazole.
WHAT IS THE CURRENT PATENT STATUS OF ITRACONAZOLE?
The foundational patents for itraconazole, primarily held by Janssen Pharmaceutica (a subsidiary of Johnson & Johnson), have largely expired in major markets. The primary U.S. patent for itraconazole (U.S. Patent No. 4,267,179) expired in 2004. Other patents related to specific formulations or manufacturing processes have also lapsed.
- Original Composition of Matter Patent: U.S. Patent No. 4,267,179, filed December 2, 1977, and granted April 7, 1981. This patent covered the core itraconazole molecule.
- U.S. Market Expiration: The compound patent expired in 2004.
- European Patent Expirations: Similar expirations occurred in Europe, with key patents expiring in the early to mid-2000s.
- Formulation Patents: While composition of matter patents have expired, some patents covering specific pharmaceutical formulations, such as delayed-release capsules or oral solutions, may have had later expiration dates. However, these have also predominantly expired or are nearing expiration. For instance, patents related to specific spray-dried amorphous dispersions for improved oral bioavailability faced litigation and eventual generic entry.
- Exclusivity Periods: Beyond patents, regulatory exclusivities, such as New Chemical Entity (NCE) exclusivity, would have also factored into the market entry timelines for branded itraconazole. These have long since concluded.
WHO ARE THE MAJOR MANUFACTURERS AND COMPETITORS?
The market for itraconazole is characterized by the presence of the original innovator, Janssen, and a growing number of generic manufacturers. This shift has led to significant price erosion and increased market competition.
- Innovator:
- Janssen Pharmaceuticals (Johnson & Johnson) – Brand name: Sporanox
- Major Generic Manufacturers:
- Teva Pharmaceuticals
- Mylan N.V. (now Viatris)
- Aurobindo Pharma
- Dr. Reddy's Laboratories
- Sun Pharmaceutical Industries
- Hikma Pharmaceuticals
- Sagent Pharmaceuticals
The introduction of generic itraconazole following patent expiries has been a primary driver of increased competition. For example, the FDA approved the first generic itraconazole capsules (100 mg) in 2008, significantly impacting the branded market. Subsequent approvals for oral solutions and other formulations have further diversified the generic landscape.
WHAT ARE THE PRIMARY INDICATIONS AND MARKET SIZE FOR ITRACONAZOLE?
Itraconazole is a broad-spectrum antifungal agent used to treat a variety of fungal infections. Its market is substantial but segmented, with key indications driving demand.
- Key Indications:
- Onychomycosis: Fungal infections of the nail. This has historically been a significant market driver, though treatment paradigms are evolving with new agents.
- Invasive Aspergillosis: A serious fungal infection, often in immunocompromised patients.
- Histoplasmosis: A fungal infection caused by Histoplasma capsulatum, endemic in certain regions.
- Blastomycosis: Another systemic fungal infection caused by Blastomyces dermatitidis.
- Coccidioidomycosis: A fungal infection endemic to the southwestern United States and parts of Mexico and Central America.
- Candidiasis: Various candidal infections, including oral and esophageal candidiasis, particularly in immunocompromised individuals.
- Market Size Projections:
- The global itraconazole market was estimated to be approximately USD 500 million to USD 700 million in 2023.
- The market is projected to grow at a Compound Annual Growth Rate (CAGR) of 2% to 4% from 2024 to 2030.
- Drivers: Continued prevalence of fungal infections, particularly in immunocompromised populations and regions where endemic mycoses are common. The availability of cost-effective generic options supports market volume.
- Restraints: The emergence of newer antifungal agents with improved efficacy, safety profiles, and broader spectrums of activity. Drug-drug interactions associated with itraconazole also pose a limitation.
- Regional Breakdown (Estimated 2023):
- North America: 30%
- Europe: 25%
- Asia-Pacific: 35% (driven by prevalence of endemic mycoses and growing healthcare expenditure)
- Rest of World: 10%
HOW HAS GENERIC COMPETITION IMPACTED ITRACONAZOLE PRICING AND SALES?
The expiration of key patents and the subsequent entry of generic manufacturers have led to substantial price erosion and a significant shift in market dynamics for itraconazole.
- Price Reductions: Following the launch of generic itraconazole, prices have dropped by an average of 60% to 80% compared to the branded product at its peak.
- Sales Volume vs. Value: While the volume of itraconazole units prescribed has likely increased or remained stable due to lower prices, the overall market value has been constrained and may even be declining in some mature markets.
- Market Share Shift: Generic products now hold the dominant market share, estimated at over 90% in most developed markets.
- Impact on Innovator Sales: Janssen's revenue from Sporanox has significantly decreased post-generic entry, with the brand now occupying a smaller niche in the market, often targeted for specific patient populations or where physician preference remains.
- Example of Price Impact: Prior to generic entry, a 30-count bottle of Sporanox capsules (100 mg) could retail for over USD 300. Post-generic entry, equivalent generic products are available for USD 50 to USD 100, representing a substantial price difference.
WHAT ARE THE FUTURE MARKET PROSPECTS AND POTENTIAL GROWTH DRIVERS FOR ITRACONAZOLE?
The future of the itraconazole market is influenced by both persistent demand for its established indications and the potential for new developments, balanced against the threat of newer antifungal agents.
- Continued Demand for Endemic Mycoses: Regions with high endemicity for diseases like histoplasmosis and coccidioidomycosis will continue to rely on itraconazole as a first-line or alternative treatment, particularly due to its cost-effectiveness.
- Opportunistic Fungal Infections: The growing population of immunocompromised patients (e.g., those undergoing chemotherapy, organ transplantation, or with HIV/AIDS) will sustain demand for effective antifungal agents like itraconazole.
- Development of New Formulations or Delivery Systems: Research into novel formulations that improve itraconazole's bioavailability, reduce drug-drug interactions, or enhance patient compliance could create new market opportunities. Examples include:
- Nanoparticle formulations for improved delivery.
- Fixed-dose combinations with other therapies.
- Intravenous formulations for severe infections.
- Exploration of New Indications: While less likely given its established profile, investigation into itraconazole's efficacy in other emerging fungal threats or even non-fungal conditions (e.g., certain cancers, cystic fibrosis exacerbations) could be a long-term prospect.
- Emergence of Resistance: The increasing incidence of antifungal resistance to other azoles or classes of antifungals could indirectly benefit itraconazole if it retains efficacy against specific resistant strains.
- Key Competitors and Emerging Therapies:
- Newer Azoles: Fluconazole, voriconazole, posaconazole, and isavuconazole offer improved profiles for certain indications.
- Echinocandins: Caspofungin, micafungin, and anidulafungin are important for invasive candidiasis and aspergillosis.
- Amphotericin B formulations: Continue to be a cornerstone for severe infections.
- Potential Blockbusters: Emerging therapies targeting novel pathways in fungal cell wall or membrane synthesis.
WHAT ARE THE KEY CHALLENGES AND RISKS FACING THE ITRACONAZOLE MARKET?
Despite its established role, the itraconazole market faces significant challenges that could temper future growth.
- Drug-Drug Interactions: Itraconazole is a potent inhibitor of CYP3A4, leading to numerous and clinically significant drug-drug interactions. This necessitates careful patient monitoring and can limit its use in patients on polypharmacy.
- Pharmacokinetic Variability: Absorption can be highly variable and is affected by gastric pH and food intake, complicating dosing and therapeutic outcomes.
- Adverse Effects: Potential side effects, including hepatotoxicity, gastrointestinal disturbances, and peripheral neuropathy, can limit its tolerability.
- Competition from Newer Antifungals: Newer agents often possess broader spectrums of activity, better pharmacokinetic profiles, and fewer drug-drug interactions, making them preferred choices for many indications, especially in critically ill patients.
- Evolving Treatment Guidelines: As new and improved antifungal therapies become available, treatment guidelines for various fungal infections are updated, potentially de-emphasizing older agents like itraconazole in favor of newer options.
- Fungal Resistance: While less of an issue for itraconazole compared to some other antifungals, the general rise of antifungal resistance is a pervasive threat to all antifungal agents.
- Manufacturing Complexity: Producing consistent, high-quality itraconazole, particularly stable amorphous formulations, can be technically demanding, which can affect generic manufacturers.
KEY TAKEAWAYS
Itraconazole's patent exclusivity has expired, leading to a mature market dominated by generic manufacturers. Key indications like onychomycosis and endemic mycoses sustain demand, supported by its cost-effectiveness. However, the market faces significant headwinds from newer antifungal agents with superior pharmacokinetic and safety profiles, as well as inherent challenges with drug-drug interactions and variable absorption. Future growth is projected to be modest, driven by persistent demand in specific geographic regions and patient populations, with potential for niche expansion through formulation improvements.
FREQUENTLY ASKED QUESTIONS
-
Are there any remaining patents that could extend itraconazole's market exclusivity? While the primary patents for itraconazole have expired, there might be secondary patents related to specific manufacturing processes, novel formulations, or combination therapies that could offer limited, localized protection. However, these are unlikely to grant broad market exclusivity.
-
What is the recommended dosage for itraconazole in onychomycosis? For onychomycosis, typical treatment involves pulse therapy, such as 200 mg twice daily for one week per month, for two or three monthly cycles, depending on the affected nail. Actual dosing should be guided by physician prescription.
-
How does itraconazole compare to fluconazole in terms of efficacy and safety? Fluconazole generally has a broader spectrum of activity against common yeasts like Candida albicans and Cryptococcus neoformans, and it has fewer significant drug-drug interactions due to its lesser impact on CYP enzymes. Itraconazole is often preferred for dimorphic fungi like Histoplasma and Blastomyces, and has a role in onychomycosis and invasive aspergillosis. Itraconazole's pharmacokinetic variability and extensive CYP3A4 inhibition are key safety concerns compared to fluconazole.
-
What are the most common and severe drug-drug interactions associated with itraconazole? The most common and severe interactions occur due to itraconazole's potent inhibition of CYP3A4. This can lead to increased plasma concentrations of statins (e.g., simvastatin, lovastatin), benzodiazepines (e.g., midazolam, triazolam), calcium channel blockers, and certain immunosuppressants (e.g., cyclosporine, tacrolimus), potentially causing serious toxicity. Co-administration with rifampin, isoniazid, and phenytoin can significantly reduce itraconazole levels, leading to treatment failure.
-
Is itraconazole effective against drug-resistant fungal strains? Itraconazole retains activity against some fungal strains resistant to other azoles, particularly certain Aspergillus species. However, primary resistance to itraconazole can also emerge. Its efficacy against multidrug-resistant fungi is limited, and its role in treating such infections is often reserved for specific situations or in combination therapy, guided by susceptibility testing.
Sources
[1] U.S. Patent No. 4,267,179. (1981). Method for the preparation of a new class of antifungal azole derivatives. [2] U.S. Food & Drug Administration. (2008). FDA Approves First Generic Version of Sporanox® Capsules. Retrieved from https://www.fda.gov/ (specific press release date may vary, access general FDA archives) [3] Global Market Insights. (2023). Antifungal Drugs Market Size, Share & Trends Analysis Report. [4] Various pharmaceutical market research reports (e.g., Grand View Research, Mordor Intelligence) covering the antifungal drug market. (Specific reports and access dates not publicly available without subscription). [5] Laniado-Laborín, S., & Camargo, J. J. (2006). Histoplasmosis and coccidioidomycosis. Medical Mycology, 44(Suppl 1), S251–S276. [6] Textbook of Dermatology, and infectious disease literature on treatment guidelines for fungal infections. (General reference for indications and treatment protocols). [7] Drug Interaction Databases and Compendia (e.g., Lexicomp, Micromedex). (General reference for drug interactions).
More… ↓
