Last updated: February 13, 2026
Current Market Position
Zidovudine (AZT) remains a historical antiretroviral drug primarily used for HIV treatment. Though its market share has declined with newer therapies, AZT continues to be included in combination regimens, especially in resource-limited settings and as part of fixed-dose combinations (FDCs). Its global annual sales were approximately $150 million in 2022, according to IQVIA data. The drug is marketed mainly by Mylan (now part of Viatris), Sun Pharmaceutical, and generic manufacturers.
Regulatory Status
- Approved by the US FDA in 1987.
- Recognized by the World Health Organization (WHO) as an essential medicine.
- Patent expired in 2005 in the US, with generics dominating thereafter.
Market Drivers
- HIV prevalence: Over 38 million people worldwide with HIV, with the majority located in Sub-Saharan Africa.
- WHO guidelines: Recommends AZT as part of first-line therapy, especially in developing countries, boosting demand.
- Cost containment: Low-cost generics are key for public health programs, maintaining market viability.
Market Challenges
- Competition from newer drugs such as Tenofovir, Emtricitabine, and integrase inhibitors.
- Reduced usage in high-income markets due to better efficacy and tolerability profiles of alternatives.
- Limited innovation due to patent expiration; little to no recent product reformulation or new indications.
Market Forecast: 2023-2030
| Year |
Estimated Total Market Revenue |
Key Factors influencing market |
| 2023 |
$140 million |
Continued use in fixed-dose combinations, especially in low-income countries; stable demand in HIV programs. |
| 2024 |
$138 million |
Slight decline due to substitution by newer drugs in developed nations; sustained demand in emerging markets. |
| 2025 |
$135 million |
Further erosion as integrase inhibitors gain prominence; global health policies favor newer regimens. |
| 2026-2030 |
$125 - $130 million |
Gradual decline; ongoing reliance in resource-poor settings and for combination therapies. |
Price Projections
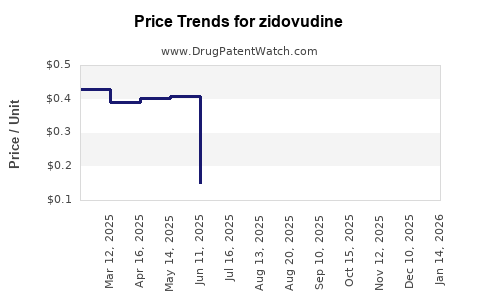
- Average Wholesale Price (AWP): Approximately $0.80 per 300 mg tablet in 2023.
- Generic Price Trends: Prices have declined 50% since patent expiry.
- Forecast: Prices are expected to remain stable or decline slightly, averaging around $0.70 per tablet by 2030, primarily driven by manufacturing efficiencies and increased competition.
Pricing Dynamics & Market Segments
- Low-cost Generics: Dominant in sub-Saharan Africa, India, and Southeast Asia; price range $0.50–$0.75 per tablet.
- Branded or Patent-Associated Products: Limited in presence due to patent expiry; prices are comparable to generics.
Regional Variations
| Region |
2023 Market Share |
Pricing Range |
| Sub-Saharan Africa |
60% |
$0.50–$0.75 per tablet |
| North America |
10% |
$0.70–$1.00 per tablet |
| Europe |
12% |
$0.70–$1.00 per tablet |
| Asia |
15% |
$0.50–$0.80 per tablet |
Competitive Landscape
- Generics dominate due to low barriers post-patent expiry.
- Fixed-dose combinations incorporating AZT are expected to maintain demand.
- Potential for biosimilar development is low; AZT is a small molecule with limited scope for biosimilar competition.
Implications for Stakeholders
- Manufacturers: Focus on optimizing production in emerging markets to sustain margins.
- Investors: Anticipate slow, steady decline; diversification into newer antiretrovirals may be prudent.
- Public Health Agencies: Secure affordable generics for lifelong treatment programs.
Key Takeaways
- Zidovudine's market is stable but declining, driven by the shift toward more effective or better-tolerated therapies.
- Prices are expected to remain low, with slight declines possible as competition increases.
- Demand will persist mainly in resource-limited settings and as part of fixed-dose combinations.
- No significant innovation or patent considerations are expected to alter the market landscape.
- Stakeholders should consider generics' stability and the lifecycle stage of AZT for strategic planning.
Frequently Asked Questions
-
Will AZT regain market share against newer antiretrovirals? No. The trend favors drugs with better efficacy and tolerability profiles, reducing AZT's role.
-
Are there any new formulations or indications for AZT? No significant new formulations or indications are currently in development.
-
How does patent expiry influence pricing? Patent expiry led to the proliferation of generics, significantly reducing AZT's price.
-
What is the outlook for AZT in emerging markets? Demand remains stable in these regions due to affordability and inclusion in HIV treatment protocols.
-
Could biosimilars develop for AZT? No, because AZT is a small-molecule drug, and biosimilars are not applicable.
References
[1] IQVIA, "Market Trends and Sales Data," 2022.
[2] World Health Organization, "Essential Medicines List," 2022.
[3] US FDA, "Zidovudine Approval History," 1987.
[4] EvaluatePharma, "Global Antiretroviral Market Projections," 2023.