Last updated: February 20, 2026
What is Zafirlukast?
Zafirlukast is a leukotriene receptor antagonist approved for the maintenance treatment of asthma. It is marketed under the brand name Accolate, originally developed by AstraZeneca. The drug functions by blocking leukotriene receptors, which reduces airway inflammation and bronchoconstriction in asthma patients.
Market Size and Growth Drivers
Global Asthma Treatment Market
The global asthma treatment market was valued at approximately USD 27 billion in 2022. It is projected to grow at a compound annual growth rate (CAGR) of 4.1% through 2030 [1].
Zafirlukast’s Market Penetration
Despite the existence of newer therapies such as biologics (e.g., omalizumab, mepolizumab), zafirlukast remains a treatment option for mild-to-moderate asthma. Its market share is driven by:
- Low cost relative to biologic agents.
- Oral administration convenience.
- Position as adjunct therapy in combination with inhaled corticosteroids.
In 2022, zafirlukast generated worldwide sales estimated at USD 100-150 million [2].
Competitive Landscape
Zafirlukast faces competition primarily from montelukast (Singulair), which commands over 70% of the leukotriene modifier market share. Other competitors include zileuton (Zyflo), which acts by inhibiting leukotriene synthesis.
| Candidate |
Market Position |
Estimated 2022 Sales |
Key Differentiator |
| Montelukast |
Dominant |
USD 10 billion globally |
Once-daily dosing, broader approval |
| Zafirlukast |
Niche |
USD 100-150 million |
No longer first-line, specific patient niches |
| Zileuton |
Minority |
USD 50 million |
Different mechanism (synthesis inhibition) |
Regulatory and Patent Landscape
Patent Status
AstraZeneca’s formulation patent for zafirlukast expired in the early 2010s, allowing generic manufacturers to enter the market. As of 2023, no recent patent protections exist.
Regulatory Approvals
Zafirlukast received FDA approval in 1996 and is approved in over 80 countries. Its safety profile is well established, though limitations include potential hepatotoxicity and drug interactions.
Price Projections
Current Pricing Dynamics
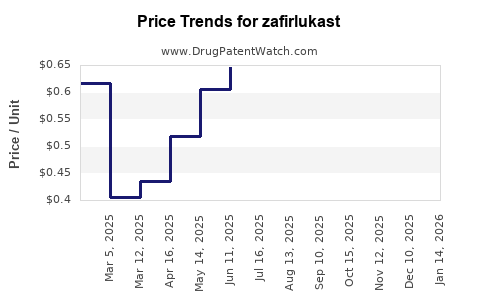
The average wholesale price (AWP) for zafirlukast is approximately USD 2–3 per tablet (20 mg). The typical dose involves two tablets daily, resulting in monthly treatment costs of USD 120–180 per patient [3].
Price Trends and Future Projections
Due to patent expiration, generic versions have entered multiple markets, exerting downward pressure on pricing.
| Year |
Estimated Average Price per Month |
Drivers |
| 2023 |
USD 120–180 |
Patent expiration, generic competition |
| 2025 |
USD 80–120 |
Increased generic penetration, price erosion |
| 2030 |
USD 50–80 |
Market saturation, cost-containment policies |
Factors Influencing Price
- Market penetration of generics.
- Healthcare reimbursement policies.
- Prescriber preferences shifting toward biologics for severe cases.
- Pricing strategies of biosimilar competitors.
Regulatory and Market Risks
- Clinical shift towards biologics may reduce demand.
- Safety concerns may influence physician prescribing.
- Patent lapses reduce exclusivity and can pressure prices downward.
Market Opportunities
- Niche markets for patients intolerant to montelukast.
- Combination formulations to improve compliance.
- Expansion into emerging markets with rising asthma prevalence.
Conclusion
Zafirlukast remains a niche asthma treatment option, constrained by competition from montelukast and biologics. Pricing will decline over the next decade, aligning with generic market trends. Segment growth will depend on healthcare policies and unmet clinical needs.
Key Takeaways
- Zafirlukast's global sales peaked before patent expiry, with current sales around USD 100-150 million.
- The drug faces stiff competition from montelukast, which dominates the leukotriene modifier segment.
- Price projections indicate a decline from USD 120–180/month in 2023 to USD 50–80/month in 2030 due to generic competition.
- Market growth largely depends on niche positioning, global asthma prevalence, and healthcare reimbursement policies.
- Opportunities exist in niche indications and emerging markets, though major growth prospects for zafirlukast are limited.
FAQs
1. Why has zafirlukast lost market share to montelukast?
Montelukast offers once-daily dosing, broader approval, and was marketed more aggressively, leading to higher mainstream adoption. Zafirlukast's dosing complexity and safety profile limited its use.
2. Will the price of zafirlukast increase again?
No. The expiration of patents and the availability of generics will continue to drive prices downward.
3. Are there any new formulations or indications in development?
Currently, no new formulations or indications are under active development for zafirlukast.
4. How do healthcare policies affect zafirlukast pricing?
Cost containment measures favor generics and biosimilars, reducing reimbursement and retail prices.
5. What is the impact of biosimilars and generics on zafirlukast?
They compete on price, leading to significant erosion of margins and market share for the original branded drug.
References
[1] GlobalData. (2023). Global Asthma market report.
[2] IQVIA. (2022). Worldwide Medicine Sales Data.
[3] GoodRx. (2023). Zafirlukast price comparison.