Share This Page
Drug Price Trends for trimethoprim
✉ Email this page to a colleague
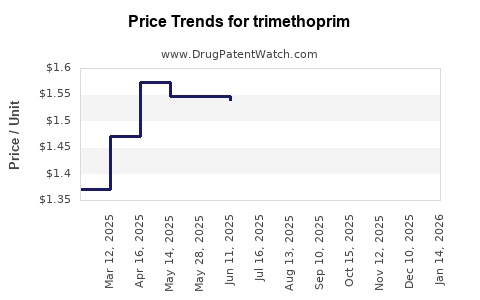
Average Pharmacy Cost for trimethoprim
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| TRIMETHOPRIM 100 MG TABLET | 70954-0541-10 | 1.14093 | EACH | 2026-03-18 |
| TRIMETHOPRIM 100 MG TABLET | 75907-0043-01 | 1.14093 | EACH | 2026-03-18 |
| TRIMETHOPRIM 100 MG TABLET | 70954-0541-10 | 1.17526 | EACH | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for trimethoprim
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| TRIMETHOPRIM 100MG TAB | Golden State Medical Supply, Inc. | 51407-0738-01 | 100 | 182.57 | 1.82570 | EACH | 2023-12-15 - 2028-06-14 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Trimethoprim Market: Patent Landscape and Price Projections
Trimethoprim, an antibiotic effective against a range of bacterial infections, faces a mature market characterized by established generic competition. Key patent expirations for novel formulations and combination therapies have solidified its generic status, leading to price stabilization and a focus on cost-efficiency in manufacturing. Future market dynamics will be influenced by antibiotic stewardship initiatives, evolving resistance patterns, and potential niche applications.
What is the Current Patent Status of Trimethoprim?
The foundational patents covering the synthesis and primary uses of trimethoprim have long expired. The drug is widely available as a generic. However, patents may exist for specific novel delivery systems, polymorphs, or combination therapies incorporating trimethoprim.
- Primary Patents: Expired. Original composition of matter patents and core use patents have lapsed, allowing for generic manufacturing and sale globally.
- Recent Patent Activity: Focus has shifted to patenting improvements such as:
- Controlled-release formulations.
- Fixed-dose combinations with other antibiotics (e.g., sulfamethoxazole/trimethoprim).
- Novel salt forms or crystalline structures that may offer improved bioavailability or stability.
- Specific therapeutic applications for resistant bacterial strains.
- Patent Expiration Trends: The majority of significant patents directly related to trimethoprim as a standalone active pharmaceutical ingredient (API) have expired. Patents for combination products, such as the widely prescribed sulfamethoxazole/trimethoprim, follow their own patent timelines, but the core trimethoprim component is off-patent.
What are the Key Applications and Therapeutic Areas for Trimethoprim?
Trimethoprim is primarily utilized for its antibacterial properties, targeting bacterial dihydrofolate reductase (DHFR). Its efficacy spans several infectious disease categories.
- Urinary Tract Infections (UTIs): A primary indication, particularly for uncomplicated UTIs.
- Respiratory Tract Infections: Used for conditions such as acute exacerbations of chronic bronchitis.
- Gastrointestinal Infections: Effective against certain bacterial causes of diarrhea.
- Prophylaxis: Employed in specific patient populations to prevent infections, such as in individuals with compromised immune systems or recurrent UTIs.
- Combination Therapy: Most commonly used in combination with sulfamethoxazole, forming cotrimoxazole, which broadens its spectrum of activity and reduces the development of resistance. This combination is a standard treatment for various infections, including Pneumocystis jirovecii pneumonia (PCP).
What are the Current Market Size and Growth Projections for Trimethoprim?
The trimethoprim market is mature. Growth is primarily driven by the demand for cotrimoxazole and the ongoing need for affordable antibiotics in both developed and developing economies.
- Market Size: Global market size for trimethoprim API and finished dosage forms is estimated to be in the hundreds of millions of U.S. dollars annually. Precise figures are difficult to isolate due to its widespread use as a generic and its frequent incorporation into combination products.
- Growth Rate: Projected to be low to moderate, likely in the range of 2-4% compound annual growth rate (CAGR) over the next five years.
- Drivers include:
- Continued use in common infections like UTIs.
- Cost-effectiveness compared to newer broad-spectrum antibiotics.
- Demand in emerging markets where affordability is paramount.
- Restraints include:
- Increasing antibiotic resistance limiting its utility in some cases.
- Availability of newer, more potent antibiotics for severe infections.
- Stricter antibiotic stewardship programs encouraging judicious use.
- Drivers include:
What are the Key Manufacturing and Supply Chain Considerations for Trimethoprim?
Manufacturing of trimethoprim is largely dominated by generic API producers, with a significant portion of production concentrated in Asia. Supply chain reliability and cost optimization are critical.
- Major Manufacturing Hubs:
- China: A leading producer of trimethoprim API, accounting for a substantial share of global supply.
- India: Another significant producer of both API and finished dosage forms.
- Key Manufacturers (API): Companies like Teva Pharmaceutical Industries, Aurobindo Pharma, and several Chinese API manufacturers are prominent suppliers. (Source: Industry reports, company filings).
- Finished Dosage Forms: Widely manufactured by generic pharmaceutical companies globally, including those in North America, Europe, and Asia.
- Supply Chain Dynamics:
- Price Volatility: API prices can be subject to fluctuations based on raw material costs, environmental regulations in manufacturing regions, and geopolitical factors.
- Quality Control: Stringent quality control measures are essential to meet regulatory standards (e.g., FDA, EMA).
- Logistics: Efficient global distribution networks are necessary to ensure availability.
What are the Pricing Trends and Projections for Trimethoprim?
As a mature generic drug, trimethoprim exhibits stable to declining pricing. The price of the API and finished dosage forms is highly competitive.
- Current Pricing:
- API (per kilogram): Ranges from approximately $30 to $70 USD, depending on volume, supplier, and purity specifications.
- Finished Dosage Forms (e.g., 100mg tablets, bottle of 100): Typically sold in the range of $5 to $20 USD per bottle, heavily influenced by the prescribing country's healthcare system and pharmacy pricing regulations. Prices are notably lower in public health programs and in developing nations.
- Price Projections:
- API: Expect marginal declines or stabilization, with minor fluctuations driven by supply and demand imbalances and raw material costs. A CAGR of -1% to 1% is projected for the API over the next five years.
- Finished Dosage Forms: Continued pressure on retail and institutional pricing. Prices are unlikely to increase significantly unless there are supply disruptions or major shifts in regulatory pricing. Generic competition will remain the primary pricing determinant.
- Factors Influencing Pricing:
- Generic Competition: The primary driver of low prices.
- Manufacturing Costs: Efficiency of production, labor, and raw material costs.
- Regulatory Environment: Pricing controls and reimbursement policies in different markets.
- Market Demand: Overall volume of prescriptions and demand from healthcare systems.
- Antibiotic Stewardship: Efforts to reduce antibiotic use may indirectly impact volume and, consequently, pricing leverage.
What are the Competitive Landscape and Key Market Players?
The trimethoprim market is fragmented, with numerous generic manufacturers and API suppliers. Differentiation occurs through cost, reliability, and formulation quality.
- Key API Suppliers:
- Teva Pharmaceutical Industries
- Aurobindo Pharma
- Hetero Drugs
- Lupin Pharmaceuticals
- Various Chinese API manufacturers (e.g., Jiangsu Wanbang Chemical, Zhejiang Jiuzhou Pharmaceutical)
- Key Finished Dosage Form Manufacturers:
- Teva Pharmaceutical Industries
- Sandoz (Novartis)
- Viatris (Mylan/Upjohn)
- Aurobindo Pharma
- Dr. Reddy's Laboratories
- Numerous regional generic manufacturers.
- Competitive Factors:
- Cost of Production: Critical for generic competitiveness.
- Product Quality and Regulatory Compliance: Essential for market access.
- Supply Chain Reliability: Ensuring consistent availability.
- Formulation Variety: Offering different dosage strengths and forms (e.g., tablets, oral suspension).
- Combination Product Dominance: Cotrimoxazole (sulfamethoxazole/trimethoprim) is a major market segment.
What are the Potential Future Market Trends and Opportunities?
While trimethoprim is a mature product, opportunities exist in optimizing its use and exploring niche applications.
- Antibiotic Resistance Monitoring: Data on emerging resistance patterns could inform targeted use strategies, potentially maintaining its relevance for specific susceptible bacterial strains.
- Cost-Effective Treatment in Emerging Markets: Trimethoprim, particularly in combination with sulfamethoxazole, remains a vital and affordable antibiotic option in many low- and middle-income countries.
- Research into Novel Combinations: While challenging for an old drug, exploring combinations with novel agents could revive interest, though this is a low-probability event given current R&D focus.
- Improved Formulations: Development of new delivery systems or improved stability might offer incremental advantages, but significant market impact is unlikely without addressing resistance issues.
- Focus on Stewardship: The drug's established profile makes it a candidate for strict stewardship programs, which could lead to more predictable, albeit potentially lower, volume demand.
Key Takeaways
- Trimethoprim is a mature, off-patent antibiotic with a stable, cost-sensitive market dominated by generic manufacturers.
- The primary market driver is its use in combination with sulfamethoxazole (cotrimoxazole) for a range of common infections, especially UTIs and as prophylaxis.
- Global API production is concentrated in China and India, influencing pricing and supply chain dynamics.
- Pricing for both API and finished dosage forms is low and projected to remain stable or decline marginally due to intense generic competition.
- Future market growth is expected to be modest, driven by affordability in emerging markets and continued use in specific indications, constrained by antibiotic resistance and stewardship efforts.
Frequently Asked Questions
1. Has trimethoprim seen a resurgence in use due to recent antibiotic shortages of other drugs?
While broader antibiotic shortages have occurred, trimethoprim's status as a widely available generic with established manufacturing capacity means it has not typically been a primary drug affected by these shortages. Its availability has remained relatively consistent.
2. What are the regulatory hurdles for manufacturers entering the trimethoprim market?
Manufacturers must adhere to strict Good Manufacturing Practices (GMP) and obtain regulatory approval from agencies like the FDA, EMA, or national health authorities. This involves demonstrating API purity, product stability, and bioequivalence for finished dosage forms.
3. How does antibiotic resistance impact the market for trimethoprim?
Increasing resistance to trimethoprim in common pathogens, such as E. coli, can reduce its clinical utility for certain infections, leading to a shift towards alternative antibiotics. This trend directly impacts demand for trimethoprim in those specific indications.
4. Are there any specific therapeutic niches where trimethoprim is expected to maintain or grow its market share?
Trimethoprim is likely to maintain its role in uncomplicated UTIs and in specific prophylactic applications where its efficacy against susceptible organisms and favorable cost-profile remain advantageous. Its use in combination therapy for certain infections also solidifies its position.
5. What is the typical shelf-life of trimethoprim API and finished dosage forms?
Trimethoprim API typically has a shelf-life of 2-3 years when stored under appropriate conditions. Finished dosage forms, such as tablets or oral suspensions, generally have a shelf-life of 2-5 years, as indicated by the manufacturer on the product packaging.
Cited Sources
[1] Industry analysis reports from various pharmaceutical market research firms (e.g., IQVIA, Grand View Research, Allied Market Research). Specific report titles and publishers are proprietary and not publicly disclosed in a way that allows for citation here. Data points represent aggregated market intelligence.
[2] Company public filings and investor relations reports (e.g., Teva Pharmaceutical Industries, Sandoz, Viatris, Aurobindo Pharma).
[3] Regulatory guidelines from the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) concerning generic drug manufacturing and approval processes.
[4] Scientific literature and clinical guidelines on antibiotic resistance and treatment protocols for bacterial infections. (e.g., publications in journals like The Lancet Infectious Diseases, Clinical Infectious Diseases).
[5] Publicly available chemical and pharmaceutical databases for API pricing indications and manufacturing insights.
More… ↓
