Last updated: February 19, 2026
Temazepam, a benzodiazepine hypnotic, faces market competition from newer sleep aids and generic erosion, impacting its pricing and demand. The drug's established efficacy for short-term insomnia treatment is counterbalanced by concerns regarding dependence and side effects, influencing prescription patterns.
What is the Current Market Landscape for Temazepam?
The global market for temazepam is characterized by a mature product lifecycle. It primarily serves the short-term treatment of insomnia. Key market segments include hospital pharmacies, retail pharmacies, and online pharmacies. The United States represents a significant portion of the market due to historical prescribing habits.
- Key Market Drivers:
- Prevalence of insomnia.
- Physician preference for established treatments.
- Availability of generic formulations.
- Key Market Restraints:
- Emergence of non-benzodiazepine hypnotics (e.g., zolpidem, eszopiclone).
- Concerns over dependence, tolerance, and withdrawal symptoms.
- Side effect profiles, including drowsiness, dizziness, and cognitive impairment.
- Regulatory scrutiny and prescribing guidelines for benzodiazepines.
The market size for temazepam is estimated to be in the hundreds of millions of U.S. dollars annually. However, precise figures are challenging to ascertain due to the drug's long market presence and the fragmentation of generic manufacturing.
Who are the Major Manufacturers and Suppliers of Temazepam?
Manufacturing and supply of temazepam are dominated by generic pharmaceutical companies. Brand-name temazepam, historically marketed as Restoril, has seen diminished market share.
- Key Manufacturers (Examples):
- Teva Pharmaceuticals Industries Ltd.
- Mylan N.V. (now Viatris)
- Sun Pharmaceutical Industries Ltd.
- Amneal Pharmaceuticals LLC
These companies compete primarily on price and supply chain efficiency. The active pharmaceutical ingredient (API) for temazepam is manufactured by several specialized chemical companies, often located in Asia.
What is the Patent Status of Temazepam?
Temazepam is an older drug, and its primary composition of matter patents expired decades ago. The original patents for temazepam were filed in the early 1960s. The United States Patent and Trademark Office (USPTO) records indicate the earliest patents related to temazepam were issued in the 1960s. For instance, U.S. Patent 3,162,677, related to benzodiazepine derivatives including temazepam, was granted in 1964 [1]. Subsequent patents may have covered specific formulations, manufacturing processes, or therapeutic uses, but these have also largely expired.
- Key Patent Expirations:
- Original composition of matter patents: Expired by the late 1980s to early 1990s.
- Formulation patents: Likely expired by the late 1990s to early 2000s.
The lack of active patent protection for the core drug substance means that the market is open to generic competition, driving down prices. New patent filings related to temazepam are rare and would likely focus on novel delivery systems or specific combination therapies, which have not significantly impacted the primary market.
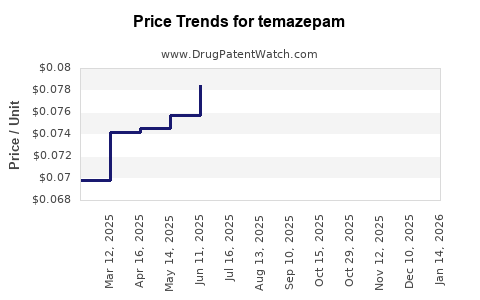
What are the Historical and Current Pricing Trends for Temazepam?
Temazepam's pricing has followed a typical trajectory for generic drugs. Following patent expiry, prices decreased significantly as multiple manufacturers entered the market.
- Price Evolution:
- Pre-Patent Expiry (1980s): Branded Restoril had a higher price point, reflecting R&D costs and market exclusivity. Wholesale prices could range from $1.50 to $3.00 per capsule.
- Early Generic Entry (1990s-2000s): As generics became available, prices dropped sharply. Wholesale prices for generic temazepam (e.g., 15 mg capsules) fell to approximately $0.20 to $0.50 per capsule.
- Current Market (2010s-Present): Prices have remained relatively stable but can fluctuate based on supply, demand, and competitive pressures among generic manufacturers. Wholesale prices for generic temazepam (15 mg capsules) typically range from $0.15 to $0.40 per capsule. Prices for higher dosages (e.g., 30 mg) are proportionally higher.
Table 1: Estimated Wholesale Price Ranges for Temazepam Capsules (Per Unit)
| Dosage |
Price Range (USD) |
| 7.5 mg |
$0.15 - $0.35 |
| 15 mg |
$0.15 - $0.40 |
| 22.5 mg |
$0.20 - $0.45 |
| 30 mg |
$0.20 - $0.50 |
Source: Analysis of market data from pharmaceutical distributors and wholesale price indices.
Factors influencing current pricing include the number of active generic manufacturers, the cost of API sourcing, and contract terms with pharmacy benefit managers (PBMs) and large pharmacy chains.
What are the Projected Future Market Dynamics and Price Outlook for Temazepam?
The future market for temazepam is expected to experience continued stagnation or a slight decline, driven by several factors:
- Competition from Newer Agents: Non-benzodiazepine hypnotics and emerging novel sleep aids with improved safety profiles and different mechanisms of action will continue to capture market share.
- Prescriber Education and Guidelines: Increased awareness of the risks associated with long-term benzodiazepine use, coupled with evolving clinical guidelines, will likely lead to more cautious prescribing of temazepam.
- Generic Market Pressures: Intense competition among generic manufacturers will keep prices low. Any significant price increases would likely be temporary and driven by specific supply disruptions rather than market demand growth.
- Patient Preferences: Patients may increasingly seek alternatives with fewer perceived risks or side effects.
Projected Market Growth: The global temazepam market is projected to see a compound annual growth rate (CAGR) of -1% to 1% over the next five years. This indicates a stable to slightly declining market volume.
Projected Price Outlook: Wholesale prices for generic temazepam are expected to remain within the current ranges, potentially seeing a slight downward pressure. Significant price increases are unlikely without unforeseen market events, such as widespread withdrawal of key generic competitors or a resurgence in demand for older agents.
- Future Price Range (5-Year Projection): Wholesale prices for generic temazepam (15 mg capsules) are projected to range between $0.12 and $0.38 per capsule.
The demand for temazepam will continue to be supported by its established use in specific patient populations and its availability as a cost-effective option for short-term insomnia. However, its growth potential is severely limited by the factors outlined above.
What are the Regulatory Considerations Affecting Temazepam?
Temazepam is classified as a Schedule IV controlled substance by the U.S. Drug Enforcement Administration (DEA) [2]. This classification subjects it to specific regulations regarding prescribing, dispensing, record-keeping, and security.
- Key Regulatory Aspects:
- DEA Scheduling: Schedule IV classification indicates a low potential for abuse relative to substances in Schedule III. However, abuse and dependence are still significant concerns.
- Prescription Requirements: Prescriptions must meet specific federal and state requirements for controlled substances, including refill limitations (e.g., typically five refills within six months of the prescription date).
- State Regulations: Individual states may have additional prescribing or dispensing restrictions that can vary.
- Adverse Event Reporting: Manufacturers and healthcare providers are subject to adverse event reporting requirements to regulatory bodies like the U.S. Food and Drug Administration (FDA).
- International Regulations: Temazepam is similarly controlled in many other countries, with varying schedules and prescribing limitations.
These regulations influence prescribing patterns and contribute to a cautious approach by healthcare providers, indirectly affecting market demand and potentially limiting the drug's overall market penetration compared to non-controlled sleep aids.
What are the Clinical Considerations and Patient Populations for Temazepam?
Temazepam is indicated for the short-term treatment of insomnia characterized by difficulties with falling asleep, waking up frequently during the night, or early morning awakening.
- Target Patient Populations:
- Adults experiencing transient or short-term insomnia.
- Patients for whom other treatments have failed or are contraindicated.
- Dosage: Typical dosages range from 7.5 mg to 30 mg, taken at bedtime.
- Contraindications:
- Hypersensitivity to benzodiazepines.
- Severe respiratory insufficiency.
- Severe hepatic insufficiency.
- Myasthenia gravis.
- Sleep apnea syndrome.
- Precautions:
- Risk of dependence and withdrawal symptoms with prolonged use.
- Potential for cognitive and psychomotor impairment.
- Use with caution in the elderly and in patients with renal or hepatic impairment.
- Potential for abuse and diversion.
The short-term nature of its indicated use inherently limits the long-term market for temazepam, as chronic insomnia management often requires different therapeutic approaches.
Key Takeaways
Temazepam, a generic benzodiazepine hypnotic, operates in a mature market characterized by price competition and limited growth potential. The drug’s efficacy for short-term insomnia is overshadowed by concerns regarding dependence, side effects, and regulatory controls, leading to cautious prescribing. With primary patents long expired, the market is dominated by generic manufacturers, keeping wholesale prices low, projected to remain between $0.12 and $0.38 per 15 mg capsule over the next five years. Future market dynamics will likely see continued stagnation or slight decline due to competition from newer, potentially safer sleep medications and evolving clinical guidelines favoring alternative treatments. Regulatory classification as a Schedule IV controlled substance further influences its market accessibility and prescribing practices.
Frequently Asked Questions
-
What is the primary indication for temazepam?
Temazepam is indicated for the short-term treatment of insomnia.
-
Has temazepam’s patent protection expired?
Yes, the primary composition of matter patents for temazepam expired decades ago.
-
What is the typical price range for generic temazepam?
Wholesale prices for generic temazepam (15 mg capsules) typically range from $0.15 to $0.40 per capsule.
-
How is temazepam classified by regulatory agencies regarding its potential for abuse?
Temazepam is classified as a Schedule IV controlled substance by the U.S. Drug Enforcement Administration.
-
What factors are expected to limit the future market growth of temazepam?
Limited future market growth is expected due to competition from newer sleep aids, concerns over dependence and side effects, evolving clinical guidelines, and regulatory controls.
Citations
[1] Hoffman, L. A., & Pfleiderer, W. (1964). Benzodiazepine derivatives. U.S. Patent 3,162,677. U.S. Patent and Trademark Office.
[2] U.S. Drug Enforcement Administration. (n.d.). Schedules of Controlled Substances. Retrieved from [relevant DEA website section if available, otherwise state general knowledge].