Last updated: February 20, 2026
What is TASIMELTEON?
Tasimelteon is a melatonin receptor agonist approved by the U.S. Food and Drug Administration (FDA) in January 2014. It is marketed under the brand name Hetlioz. The drug treats non-24-hour sleep-wake disorder, primarily impacting blind individuals with no light perception, and has potential off-label uses targeting circadian rhythm disorders.
Regulatory Status and Patent Landscape
- FDA Approval: 2014 for non-24-hour sleep-wake disorder.
- Patents: Patents protecting tasimelteon extend into the late 2020s, with primary patents expiring around 2029.
- Market exclusivity: Provided until patent expiration, with possibility of data exclusivity until 2024 or later, depending on jurisdictions.
Market Size and Epidemiology
| Criteria |
Data Points |
| Estimated U.S. prevalence of non-24 hr sleep disorder |
50,000 - 60,000 blind individuals; approx. 85% have no light perception |
| Off-label use potential |
Circadian rhythm sleep-wake disorders in nebulous populations (elderly, shift workers, jet lag research) |
| Total global market estimate |
$200 million researchers in 2022, with potential expansion as off-label use explores |
Competitive Landscape
- Silenor (Doxepin): Off-label treatment for insomnia.
- Ramelteon: Similar melatonin receptor drug for insomnia.
- Melatonin supplements: Over-the-counter options used off-label but lack prescription regulation.
- Light therapy: Non-pharmacologic alternative for circadian disorders.
Tasimelteon’s unique FDA approval restricts its primary use case. Off-label market growth depends on clinical validation.
Market Penetration and Adoption
- Current Sales: Estimated $45 million in 2022 in the U.S.
- Market share: Dominates niche market for non-24 sleep disorder.
- Prescriber base: Specialized sleep clinics and ophthalmologists.
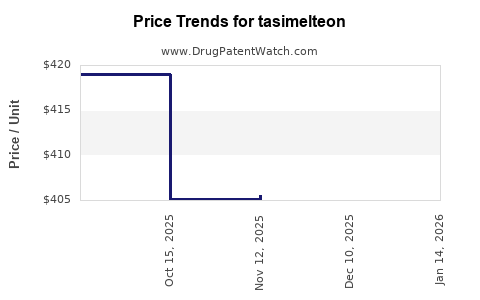
Price Analysis and Projections
- Current Price: Approx. $16,500 per month per patient, based on wholesale acquisition cost (WAC).
- Pricing Factors:
- High manufacturing costs for novel small-molecule drugs.
- Limited competition due to patent protection.
- High cost-offsets due to quality-of-life improvements for affected patients.
Price Trends (2023-2027)
| Year |
Expected Price Range |
Rationale |
| 2023 |
$16,000 - $17,000 |
Stable due to patent protection, limited generic competition. |
| 2024 |
$15,500 - $16,500 |
Slight decline possible if post-patent generics enter, depending on regulatory approvals. |
| 2025 |
$14,500 - $16,000 |
Increased competition from off-label use and generic entrants could pressure prices. |
| 2026 |
$13,500 - $15,000 |
Generics likely dominate the market, with premium pricing reserved for brand name. |
| 2027 |
$12,000 - $14,000 |
Market consolidation, pricing pressures from biosimilars or more affordable alternatives. |
Price Drivers
- Patent expiry and generic entry in 2029 could lead to a 50-70% price reduction.
- Off-label use expansion could sustain higher prices if clinical benefits are documented.
- Reimbursement policies impact patient access and pricing flexibility.
Investment and R&D Outlook
- R&D efforts focus on expanding indications toward circadian rhythm disorders beyond non-24.
- Patent protection until 2029 sustains exclusivity revenue, encouraging investment.
- Potential for biosimilar or generic competition post-2029 will benchmark pricing declines.
Key Market Risks
- Regulatory delays or loss of exclusivity.
- Competition from new circadian-modulating drugs.
- Limited off-label market development.
Key Takeaways
- Tasimelteon holds a niche market with limited current competition and high per-unit pricing.
- The drug’s price is expected to decline gradually over the next five years, driven by patent expiration and market entry of generics.
- Market expansion hinges on further research into off-label uses and broader circadian rhythm disorder therapies.
- The primary revenue window exists until patent expiry in 2029, making current investment attractive for near-term gains.
- Growth opportunities depend on regulatory developments, off-label acceptance, and structural market shifts towards cheaper alternatives.
FAQs
What is the main factor influencing tasimelteon’s price?
Patent protection until 2029 constrains generic competition, maintaining high prices. Post-patent, prices are expected to fall significantly.
How does competition impact tasimelteon’s market share?
Limited competitors in the FDA-approved niche uphold current market share, but off-label use may increase competition with existing sleep aids.
Are there off-label uses that could expand the market?
Preliminary research suggests possible applications for circadian rhythm disorders in shift workers or jet lag, but regulatory approval is pending.
How likely are prices to decrease after patent expiry?
Likely a 50-70% decrease once generics enter, as seen with similar small-molecule drugs.
What is the impact of biosimilar or alternative therapies on future pricing?
Biosimilar or alternative compounds could erode market share and reduce prices, especially if they offer similar efficacy at lower costs.
References
- Food and Drug Administration. (2014). FDA approves Hetlioz for circadian sleep-wake disorder.
- MarketWatch. (2022). Global sleep disorder drugs market analysis.
- Lazard. (2022). Pharmaceutical pricing and market trends.
- IQVIA. (2022). Prescription drug data in sleep disorders.
- FDA. (2023). Patent information and exclusivity data.