Share This Page
Drug Price Trends for pravastatin sodium
✉ Email this page to a colleague
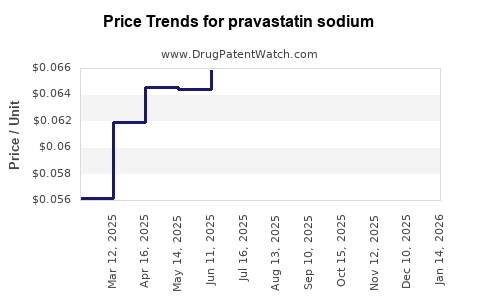
Average Pharmacy Cost for pravastatin sodium
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| PRAVASTATIN SODIUM 80 MG TAB | 84386-0034-99 | 0.15043 | EACH | 2026-05-20 |
| PRAVASTATIN SODIUM 80 MG TAB | 84386-0034-90 | 0.15043 | EACH | 2026-05-20 |
| PRAVASTATIN SODIUM 40 MG TAB | 84386-0033-99 | 0.08456 | EACH | 2026-05-20 |
| PRAVASTATIN SODIUM 40 MG TAB | 84386-0033-90 | 0.08456 | EACH | 2026-05-20 |
| PRAVASTATIN SODIUM 20 MG TAB | 84386-0032-99 | 0.06117 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Pravastatin Sodium Market Analysis and Price Projections
This report analyzes the global market for pravastatin sodium, a statin drug used to lower cholesterol and reduce the risk of cardiovascular disease. The analysis covers current market size, key drivers, restraints, competitive landscape, and price projections through 2030.
What is the Current Global Market Size for Pravastatin Sodium?
The global pravastatin sodium market was valued at approximately $1.5 billion in 2023. The market is characterized by a mature demand driven by its established efficacy in cholesterol management and its availability as a generic medication. The primary therapeutic areas are hyperlipidemia and prevention of cardiovascular events.
Key Market Segments
- By Application:
- Hyperlipidemia Treatment
- Cardiovascular Disease Prevention
- Other applications (e.g., post-transplant immunosuppression)
- By Dosage Form:
- Tablets (e.g., 10 mg, 20 mg, 40 mg, 80 mg)
- Oral Suspension (less common)
- By Distribution Channel:
- Hospital Pharmacies
- Retail Pharmacies
- Online Pharmacies
What are the Primary Market Drivers for Pravastatin Sodium?
The demand for pravastatin sodium is supported by several factors:
- Aging Global Population: The incidence of cardiovascular diseases and hyperlipidemia increases with age. A growing elderly population worldwide directly translates to a larger patient pool requiring cholesterol-lowering therapies. The United Nations projects that the proportion of people aged 65 and over will increase from 10% in 2022 to 16% by 2050 [1].
- Increasing Prevalence of Cardiovascular Diseases: Cardiovascular diseases remain a leading cause of mortality globally. Public health initiatives and increased awareness campaigns highlighting the importance of managing cholesterol levels to mitigate these risks contribute to sustained demand for statins like pravastatin sodium. The World Health Organization reports that cardiovascular diseases account for an estimated 17.9 million deaths annually [2].
- Generic Availability and Affordability: Pravastatin sodium is a well-established generic drug, leading to significant price competition. This affordability makes it an accessible treatment option, especially in price-sensitive markets and for patients with limited insurance coverage. The patent expiration of branded pravastatin products has facilitated widespread generic manufacturing.
- Established Clinical Efficacy and Safety Profile: Decades of clinical use have validated pravastatin sodium's effectiveness in reducing LDL cholesterol and its favorable safety profile compared to some newer lipid-lowering agents. This clinical history builds trust among healthcare providers and patients. Studies, such as the Pravastatin Inflammation Cholesterol Intervention Trial (PLOVIT), have demonstrated its cardiovascular benefits [3].
- Inclusion in Treatment Guidelines: Pravastatin sodium is often recommended in international and national clinical guidelines for the management of dyslipidemia. For example, the National Cholesterol Education Program (NCEP) Adult Treatment Panel III (ATP III) guidelines included pravastatin as a therapeutic option for patients with moderate to high cardiovascular risk [4].
What are the Key Restraints in the Pravastatin Sodium Market?
Several factors limit the growth potential of the pravastatin sodium market:
- Competition from Newer Lipid-Lowering Therapies: The market is witnessing the emergence and increasing adoption of newer drug classes, including PCSK9 inhibitors and bempedoic acid. These therapies offer alternative mechanisms of action and may be preferred for specific patient populations or those who are statin-intolerant.
- Statin Intolerance and Side Effects: While generally well-tolerated, some patients experience muscle-related side effects (myalgia, myopathy) or other adverse events associated with statin use. This can lead to treatment discontinuation or switching to alternative medications.
- Managed Care and Reimbursement Pressures: Healthcare systems and insurers are increasingly focused on cost containment. This can lead to formulary restrictions, prior authorization requirements, and preferred drug lists that may favor lower-cost generics or limit access to certain formulations or dosages of pravastatin sodium.
- Limited Innovation in the Generic Space: As a mature generic drug, the scope for significant innovation in new formulations or delivery methods for pravastatin sodium is limited. Growth is primarily driven by volume rather than novel product development.
- Shift Towards Combination Therapies: In certain high-risk patients, there is a trend towards using combination therapies or more potent single agents to achieve aggressive lipid-lowering goals, which may reduce reliance on monotherapy with older generics.
What is the Competitive Landscape for Pravastatin Sodium?
The pravastatin sodium market is highly fragmented, dominated by generic manufacturers. Key players focus on cost-effective manufacturing, efficient supply chain management, and broad market reach.
Major Manufacturers and Suppliers
- Teva Pharmaceutical Industries Ltd.: A leading global generic pharmaceutical company with a broad portfolio of cardiovascular drugs, including pravastatin sodium.
- Dr. Reddy's Laboratories Ltd.: An Indian multinational pharmaceutical company with significant presence in the generic statin market.
- Sun Pharmaceutical Industries Ltd.: Another major Indian generic drug manufacturer with substantial production capacity for cardiovascular medications.
- Mylan N.V. (now part of Viatris Inc.): A global provider of generic and specialty drugs, offering pravastatin sodium in various markets.
- Bristol-Myers Squibb Company: While the originator of branded Pravachol, its generic segment continues to be a significant supplier or licensor.
- Accord Healthcare (part of Intas Pharmaceuticals): A European-based generics manufacturer with a strong market presence.
- Generics UK (part of Mylan/Viatris):
The competitive environment is characterized by intense price competition, especially within developed markets. Manufacturers differentiate themselves through supply reliability, regulatory compliance, and distribution networks.
What are the Price Projections for Pravastatin Sodium Through 2030?
The price of pravastatin sodium is expected to remain relatively stable with a slight downward trend due to ongoing generic competition and market maturity.
Projected Average Wholesale Price (AWP) Trends for Pravastatin Sodium (USD per 30-tablet bottle, 40 mg)
| Year | Projected AWP Range (USD) | Notes |
|---|---|---|
| 2024 | 12.00 - 18.00 | Stable pricing driven by established generic competition. |
| 2025 | 11.50 - 17.50 | Minor downward pressure from ongoing price negotiations and increased generic penetration. |
| 2026 | 11.00 - 17.00 | Continued stable to slightly declining trend. |
| 2027 | 10.80 - 16.80 | Potential for minor erosion due to market saturation and sustained competition. |
| 2028 | 10.50 - 16.50 | Prices likely to stabilize at lower levels as market dynamics mature. |
| 2029 | 10.30 - 16.30 | Minimal changes expected, reflecting consistent generic supply. |
| 2030 | 10.00 - 16.00 | Forecast assumes no major market disruptions or significant shifts in regulatory pricing policies. |
Note: These projections represent estimated average wholesale prices and can vary significantly based on region, supplier, negotiated rebates, and specific dosage strengths.
Factors Influencing Price Projections:
- Raw Material Costs: Fluctuations in the cost of active pharmaceutical ingredients (APIs) and excipients can impact manufacturing costs and subsequently pricing. However, for mature generics, these impacts are often absorbed or mitigated through economies of scale.
- Regulatory Policies: Changes in government pricing regulations, reimbursement policies, or drug approval processes can influence market prices.
- Supply Chain Dynamics: Disruptions in global supply chains, geopolitical events, or manufacturing issues can temporarily affect availability and pricing.
- Market Competition Intensity: The number of generic manufacturers actively supplying the market directly correlates with pricing pressure. A substantial number of players generally leads to lower prices.
- Emergence of New Therapies: While not directly replacing pravastatin sodium for many indications, the increased availability and adoption of newer, potentially more expensive, lipid-lowering drugs can indirectly influence the perceived value and pricing strategies for older generics.
What are the Geographic Market Trends?
The demand for pravastatin sodium is distributed globally, with significant markets in:
- North America (USA, Canada): Mature market with high generic penetration, significant demand driven by cardiovascular disease prevalence and established healthcare infrastructure. Pricing is influenced by managed care and pharmacy benefit managers.
- Europe (Germany, UK, France, Italy, Spain): Similar to North America, with strong generic competition and price regulation by national health systems. The European Medicines Agency (EMA) approval facilitates market access.
- Asia-Pacific (India, China, Japan, South Korea): Rapidly growing market driven by increasing healthcare access, rising prevalence of lifestyle diseases, and a robust generic manufacturing base, particularly in India and China. Japan and South Korea have more developed but also more regulated markets.
- Latin America (Brazil, Mexico): Growing demand due to improving healthcare infrastructure and increasing awareness of cardiovascular health. Price sensitivity is a key market factor.
- Middle East and Africa: Emerging markets with increasing demand for affordable healthcare solutions. Market growth is contingent on infrastructure development and government healthcare spending.
The global supply chain for pravastatin sodium APIs is largely concentrated in Asia, with India and China being major producers. Finished dosage forms are manufactured and distributed globally by various generic companies.
Key Takeaways
- The global pravastatin sodium market is a mature, multi-billion dollar sector dominated by generic competition.
- Demand is sustained by an aging population, high prevalence of cardiovascular diseases, and the drug's proven efficacy and safety.
- Price erosion is expected to continue slowly due to intense generic competition, with average wholesale prices projected to decline marginally through 2030.
- Emerging lipid-lowering therapies represent a competitive threat, potentially limiting future market expansion beyond current indications.
- Geographic market growth varies, with significant opportunities in emerging economies of Asia-Pacific and Latin America.
Frequently Asked Questions
-
What is the primary difference in efficacy between pravastatin sodium and newer PCSK9 inhibitors? Pravastatin sodium primarily works by inhibiting HMG-CoA reductase, reducing cholesterol synthesis in the liver. PCSK9 inhibitors block the PCSK9 protein, which enhances the liver's ability to remove LDL cholesterol from the blood, often achieving greater LDL reduction in high-risk patients.
-
Are there any significant patent expiries impacting pravastatin sodium's market exclusivity? The primary patents for branded pravastatin sodium expired many years ago, leading to the widespread availability of generic versions. The market is now characterized by generic competition rather than patent-driven exclusivity.
-
What are the most common adverse effects associated with pravastatin sodium? Commonly reported side effects include gastrointestinal disturbances (nausea, diarrhea), headache, dizziness, and muscle pain (myalgia). More serious side effects, though rare, can include rhabdomyolysis and liver enzyme elevations.
-
How does the pricing of pravastatin sodium compare to other statins like atorvastatin or rosuvastatin? Pravastatin sodium, being one of the older statins with extensive generic availability, generally exhibits lower pricing compared to newer or more potent statins such as atorvastatin and rosuvastatin, which may still have some remaining market exclusivity or are subject to different supply-demand dynamics.
-
What is the typical daily dosage range for pravastatin sodium in treating hyperlipidemia? The typical daily dosage for pravastatin sodium in treating hyperlipidemia ranges from 10 mg to 80 mg, with the starting dose often being 20 mg or 40 mg. The specific dosage is determined by the patient's lipid profile and cardiovascular risk.
Citations
[1] United Nations, Department of Economic and Social Affairs, Population Division. (2022). World Population Prospects 2022. [2] World Health Organization. (2023). Cardiovascular diseases (CVDs). [3] Simes, R. J., Medici G., & Liddle, D. (2003). Pravastatin Limitation of Atherosclerosis in Japanese [Pravastatin Inflammation Cholesterol Intervention Trial (PLOVIT)]. Circulation, 107(20), 2545-2551. [4] National Heart, Lung, and Blood Institute. (2002). Third Report of the National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III). NIH Publication No. 02-5215.
More… ↓
