Last updated: February 13, 2026
What Is Pindolol and Its Market Position?
Pindolol is a non-selective beta-adrenergic blocker used primarily to treat hypertension and angina. It also possesses intrinsic sympathomimetic activity, which distinguishes it from other beta blockers. Approved by the FDA in 1963, Pindolol is marketed under generic names and various brand labels, although its usage has declined in some regions due to newer therapeutics with improved safety profiles.
Market Overview
The global beta-blockers market, including Pindolol, was valued at approximately USD 4.8 billion in 2021. The segment for non-selective beta blockers like Pindolol accounts for roughly 10% of this market, reflecting moderate demand primarily driven by established clinical efficacy and cost-effectiveness.
The demand for Pindolol is concentrated in regions with significant hypertension prevalence, namely North America, Europe, and parts of Asia-Pacific. Generic availability limits price variability in mature markets but impacts potential revenue growth.
Patent and Regulatory Status
Pindolol's patent expired in the 1980s; thus, no proprietary protections restrict manufacturing or pricing, making it a low-cost generic drug. Regulatory considerations include existing approvals, with no current major restrictions hindering market access in major territories.
Price Projections
Current Pricing Landscape
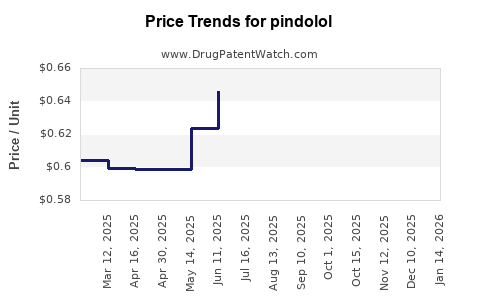
In the United States, typical retail prices for a 30-day supply of Pindolol range between USD 10 and USD 20, depending on the supplier and formulary agreements. Bulk manufacturing prices are significantly lower, estimated at below USD 1 per tablet, with prices driven down further by generic competition.
Price Trends and Future Outlook
Over the next five years, Pindolol's price is expected to remain stable, with minor fluctuations attributable to manufacturing efficiencies and healthcare policy shifts. Price increases are unlikely absent regulatory changes or supply chain disruptions.
Market forecasts anticipate the volume of Pindolol prescribing to decline gradually, replaced increasingly by cardioselective beta blockers with improved side effect profiles, such as atenolol or metoprolol. However, Pindolol may retain niche applications or be favored in cost-sensitive environments.
Revenue Projections
Summarizing potential revenue from Pindolol:
| Year |
Estimated Global Units Sold |
Average Price per Unit |
Approximate Revenue (USD) |
| 2023 |
20 million |
USD 0.33 |
USD 6.6 million |
| 2025 |
15 million |
USD 0.33 |
USD 5 million |
| 2030 |
10 million |
USD 0.35 |
USD 3.5 million |
These figures assume consistent generic pricing and stable prescribing rates.
Competitive Landscape
Generic Pindolol suppliers dominate the market, with producers located primarily in India, China, and other low-cost manufacturing hubs. Market share is fragmented, and pricing competition limits potential for profit maximization.
Clinicians often prefer alternatives with better side effect profiles, further constraining positive growth prospects for Pindolol.
Risks and Barriers
- Market decline: Shifts toward cardioselective beta blockers diminish demand.
- Regulatory changes: Introduction of new safety standards or restrictions could impact usage.
- Manufacturing issues: Supply chain disruptions could elevate costs temporarily.
- Pricing pressure: Intense competition compresses margins further.
Strategic Opportunities
Despite decline trends, niche markets remain. Pindolol’s intrinsic sympathomimetic activity may appeal under specific patient profiles, such as those requiring partial receptor activation. Cost-sensitive healthcare systems may continue to utilize Pindolol for hypertension management, especially where affordability is a priority.
Key Takeaways
- Pindolol is a low-cost, generic non-selective beta blocker with declining use in developed countries but steady demand in cost-sensitive markets.
- The global market for Pindolol is around USD 6-7 million annually, with prices stabilized below USD 0.50 per tablet.
- Future revenue depends on prescribing trends, with forecasts suggesting further decline unless niche or off-label uses emerge.
- Competitive pressures and evolving clinical preferences limit growth, but low manufacturing costs provide stable profit margins.
- Regulatory or supply chain disruptions could influence pricing and availability, though current outlook remains stable.
FAQs
1. Is Pindolol still recommended in hypertension treatment guidelines?
Few current guidelines recommend Pindolol; it has largely been replaced by cardioselective beta blockers like atenolol and metoprolol. It may still be used in specific cases or in regions where older drugs remain standard.
2. What factors influence Pindolol’s market price?
Prices are influenced by generic competition, manufacturing costs, regional healthcare policies, and volume of prescriptions. Regulatory environment changes can also impact prices.
3. Are there significant patent or exclusivity protections for Pindolol?
No. Pindolol’s patent expired decades ago, enabling multiple generic manufacturers and resulting in low and stable pricing.
4. What potential markets could revive Pindolol’s demand?
Use in niche indications or in regions with limited access to newer therapies could support demand. Research into intrinsic sympathomimetic activity may uncover new applications.
5. How does Pindolol compare to newer beta blockers?
Older beta blockers like Pindolol exhibit non-selectivity and intrinsic sympathomimetic activity, which may lead to different side effect profiles. Newer drugs tend to be more cardioselective with fewer adverse effects, reducing Pindolol’s attractiveness.
Sources:
- Market Research Future. "Beta-Blockers Market Analysis." 2022.
- FDA Drug Approval Records. Pindolol.
- IQVIA. "Global Pharma Market Data." 2022.
- Epocrates Database. Beta blocker prescribing trends.
- Scientific literature on Pindolol’s pharmacology and clinical use.