Share This Page
Drug Price Trends for pimozide
✉ Email this page to a colleague
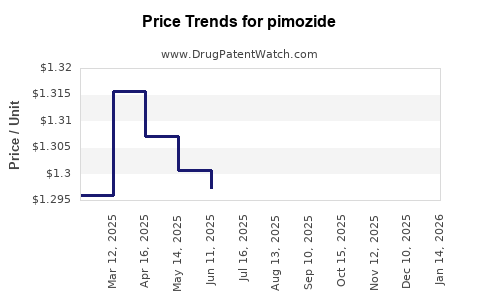
Average Pharmacy Cost for pimozide
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| PIMOZIDE 2 MG TABLET | 49884-0348-01 | 1.71340 | EACH | 2026-03-18 |
| PIMOZIDE 1 MG TABLET | 49884-0347-01 | 1.24604 | EACH | 2026-03-18 |
| PIMOZIDE 2 MG TABLET | 49884-0348-01 | 1.71353 | EACH | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Pimozide Market Analysis and Financial Projection
What is Pimozide and its Current Market Position?
Pimozide is an antipsychotic medication primarily prescribed for Tourette syndrome and certain psychotic disorders. It is a first-generation, dopamine receptor antagonist, marketed under brands such as Orap. Its patent protections have expired, shifting its market role to generics, with limited recent new developments.
What is the Market Size and Demand for Pimozide?
The global neuropsychiatric drug market was valued at approximately $12 billion in 2022, with antipsychotics accounting for around 65% of this figure. Pimozide's specific market segment remains niche due to preferred alternatives, but demand persists for conditions like Tourette syndrome and off-label uses.
Regional Market Dynamics
- United States: The largest market, driven by high prevalence of Tourette syndrome (~0.6% in children), with an estimated annual prescription volume of approximately 30,000–50,000 units.
- European Union: Similar prevalence rates, with moderate prescribing frequency.
- Asia-Pacific: Growing acceptance and increasing diagnosis rates, but lower current prescription volumes.
Competitive Landscape
Main competitors include second-generation antipsychotics like risperidone and aripiprazole, which possess more favorable side effect profiles. As a result, Pimozide's market share diminishes in favor of newer agents, but it remains in use where patients do not tolerate alternative treatments.
What Are Current Price Levels and Trends for Pimozide?
Pimozide is available mainly as a generic, with minimal branded options remaining. Pricing varies by region and procurement channel.
| Region | Approximate Price per 10 mg Tablet | Notes |
|---|---|---|
| United States | $0.05 – $0.10 | Generic, procurement through wholesalers and pharmacies |
| European Union | €0.04 – €0.08 | Generic, varies by country and procurement |
| Asia-Pacific | $0.03 – $0.07 | Lower due to manufacturing scale and market maturity |
Prices have experienced moderate declines over the past five years, driven by increased generic competition and low patent enforcement. No recent significant price spikes or patent disputes have influenced the market.
What Are Future Price Projections and Market Trends?
Given the current landscape, future pricing for Pimozide remains stable with slight downward pressure due to market saturation of generics. The main factors influencing pricing include:
- Generic Competition: Continued proliferation of generic versions will keep prices low.
- Regulatory Changes: Potential indications for off-label use may impact demand slightly but are unlikely to affect pricing significantly.
- Manufacturing Costs: Stable, with no major shifts expected over the next five years.
Price Projection (Next 5 Years)
| Year | Estimated Price per 10 mg Tablet | Assumptions |
|---|---|---|
| 2023 | $0.05 | Current market conditions |
| 2024 | $0.045 | Slight competition-driven reduction |
| 2025 | $0.045–$0.04 | Continued generic proliferation |
| 2026 | $0.04 | Market stabilization |
What Are Key Market Risks and Opportunities?
Risks:
- Shift to second-generation antipsychotics reduces demand.
- Prescribing practices may favor newer agents, impacting volume.
- Regulatory restrictions on off-label uses could limit market expansion.
Opportunities:
- Expansion into new indications, such as specific tic disorders.
- Development of combination formulations or injectable forms, potentially commanding higher prices.
- Geographic expansion into emerging markets with increasing neuropsychiatric diagnoses.
Conclusion and Strategic Recommendations
Pimozide markets remain niche, dominated by generics with stable but declining prices. Manufacturers and investors should monitor shifting prescriber preferences toward newer antipsychotics and regulatory articulations impacting off-label use. Entry opportunities are limited unless innovation or label expansion occurs. Cost reductions and process efficiencies will be critical for maintaining margins.
Key Takeaways
- Pimozide is a niche antipsychotic with a decreasing market share due to competition from second-generation drugs.
- Its market value remains anchored by generic availability, with prices averaging $0.04–$0.10 per 10 mg tablet.
- Market volume is stable but declining, with limited outlook for significant growth.
- Price trends project gradual reductions over the next five years, remaining near current levels.
- Opportunities depend on clinical expansion, formulation innovation, or geographic penetration.
FAQs
1. Why has Pimozide lost market share to newer antipsychotics?
Because second-generation agents have fewer side effects like extrapyramidal symptoms and QT prolongation, making them preferable for long-term use.
2. Is there potential for Pimozide to regain market share?
Only through new indications or clinical trials demonstrating superior efficacy or safety, which currently is limited.
3. How accessible is Pimozide in emerging markets?
Availability is increasing due to generics but limited by local regulatory approvals and prescriber familiarity.
4. Could patent issues affect Pimozide?
As it is off patent, patent expiration has already occurred, leading to widespread generic competition.
5. Are there any ongoing developments or formulations for Pimozide?
No significant new formulations or delivery mechanisms are in advanced development; the focus remains on existing generics.
Sources:
[1] IQVIA. "Global Prescriptions Data 2022."
[2] Pharmapoint. "Antipsychotic Market Analysis 2022."
[3] FDA. "Drug Approvals and Labeling."
[4] IMS Health. "Neuropsychiatric Drugs Market Trends."
[5] National Institute of Mental Health. "Prevalence of Tourette Syndrome."
More… ↓
