Last updated: February 13, 2026
What Is the Current Market Status for Perphenazine?
Perphenazine is an antipsychotic medication primarily used to treat schizophrenia, severe nausea, and other mental health disorders. It belongs to the phenothiazine class, with a long history of use dating back to the 1950s. Despite its age, it maintains a niche role in psychiatry, particularly in regions with limited access to newer drugs.
The global antipsychotic market was valued at approximately $15.7 billion in 2022. While newer atypical antipsychotics dominate the market, perphenazine retains relevance in specific segments due to its lower cost and established efficacy.
How Is the Market for Perphenazine Distributed Geographically?
The distribution of Perphenazine varies significantly across regions:
- North America: Low sales volume due to a preference for newer atypicals such as risperidone and olanzapine. Off-label use persists in specific cases, especially in Veterans Affairs hospitals.
- Europe: Similar to North America, used mainly in hospitals and clinics with limited prescriptions in outpatient settings.
- Asia-Pacific: Markets in India and China maintain use due to lower-cost generics, with a substantial demand in rural health settings.
No recent approvals of Perphenazine by major regulatory agencies (FDA, EMA) suggest declining official interest in new formulations, though generics remain accessible.
What Are the Key Drivers and Barriers in the Market?
Drivers
- Price sensitivity: Hospitals and healthcare providers in emerging markets favor low-cost generics.
- Established safety profile: Long-term clinical data support its use in specific populations.
- Generic availability: U.S. and European markets feature multiple manufacturers producing affordable versions.
Barriers
- Availability of atypical antipsychotics: Market preference shifts toward drugs with fewer side effects, such as quetiapine and aripiprazole.
- Side effect profile: Perphenazine frequently causes extrapyramidal symptoms, limiting its use.
- Regulatory restrictions: Use is limited in some countries due to adverse effects.
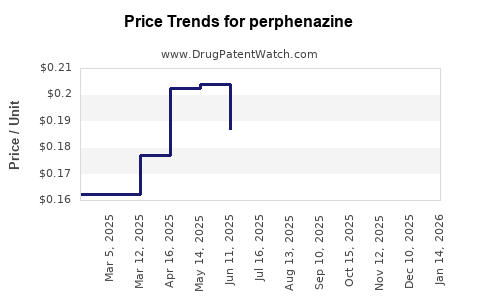
What Are the Price Trends and Projections?
Current Pricing Landscape
The average price of a 100 mg perphenazine tablet in the U.S. is approximately $0.10 to $0.20. Monthly treatment costs for typical doses (8-16 mg daily) are around $1 to $2 for generics. In low-income countries, prices can fall below $0.05 per tablet, driven by local manufacturing and regulatory policies.
Price Projections (2023-2028)
| Year |
Estimated Price Range per 100 mg Tablet |
Key Factors |
| 2023 |
$0.10 - $0.20 |
Stable generic supply |
| 2024 |
$0.09 - $0.18 |
Increased competition in generics |
| 2025 |
$0.09 - $0.17 |
Market saturation, commoditization |
| 2026 |
$0.08 - $0.16 |
Potential price pressure |
| 2027 |
$0.08 - $0.15 |
Regulatory changes in key markets |
Cost reductions may occur as patent expirations facilitate increased generic competition. No major "new" formulations or patents are anticipated, limiting price increases.
How Do Competitor Drugs and Alternative Therapies Impact Market Share?
- Atypical antipsychotics dominate new prescriptions, with risperidone, olanzapine, and quetiapine accounting for approximately 70% of the global market.
- Cost-focused healthcare systems sustain demand for older drugs like perphenazine, especially where affordability outweighs side effect concerns.
- The shift toward personalized medicine and combination therapies may marginalize perphenazine further.
What Are the Regulatory Outlooks and Patent Positions?
- No recent patents protect Perphenazine, as its original patents expired decades ago.
- Generic manufacturing is widespread, keeping prices low.
- Regulatory restrictions on use due to side effects are increasing, especially in high-income countries.
Summary of Market and Price Future Trends
The market for Perphenazine remains primarily driven by low-cost generic availability in emerging markets. Demand in developed regions declines due to side effect profiles and prescribing preferences for newer drugs. Price stability is expected for the near term, with slight decreases driven by escalating generic competition.
Key Takeaways
- Perphenazine’s market is shrinking in high-income countries but remains relevant in low-income and rural settings.
- Prices for generics are stable but tend to decline gradually due to increased competition.
- The drug faces stiff competition from newer atypicals offering better side-effect profiles.
- Regulatory restrictions in certain regions limit clinical use.
- No significant new developments or patents are anticipated to alter its market position.
FAQs
1. Is Perphenazine eligible for patent protection?
No. Existing patents have expired, and manufacturing is primarily by generic companies.
2. What are the primary side effects limiting Perphenazine's use?
Extrapyramidal symptoms, sedation, and metabolic disturbances.
3. Can Perphenazine be used in combination therapies?
Yes, but usage is limited by side-effect profile and clinical guidelines favoring newer drugs.
4. What are the prospects for new formulations of Perphenazine?
Unlikely, as most focus is on newer antipsychotics with improved tolerability.
5. How does regulatory environment affect Perphenazine availability?
Restrictive labeling and mandatory risk management programs limit use in some regions.
Citations
[1] Grand View Research. (2022). Antipsychotic Drugs Market Size, Share & Trends Analysis.
[2] IQVIA. (2023). Global Psychotropic Drug Sales Data.
[3] U.S. Food and Drug Administration. (2022). Drug Approvals and Clarifications.
[4] EMA. (2022). Management of Antipsychotic Drugs in Europe.