Share This Page
Drug Price Trends for methylphenidate
✉ Email this page to a colleague
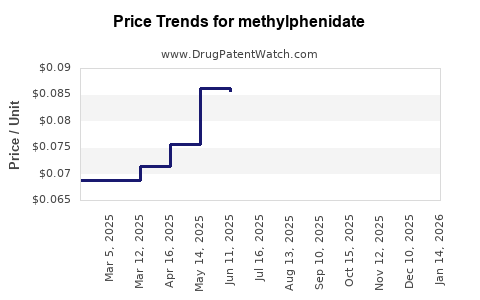
Average Pharmacy Cost for methylphenidate
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| METHYLPHENIDATE ER(LA) 60 MG CP | 75907-0053-30 | 11.04443 | EACH | 2026-05-20 |
| METHYLPHENIDATE ER(LA) 40 MG CP | 75907-0052-01 | 2.31454 | EACH | 2026-05-20 |
| METHYLPHENIDATE ER(LA) 30 MG CP | 75907-0051-01 | 2.07947 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for methylphenidate
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| QUILLICHEW ER 20MG TAB | Tris Pharma, Inc. | 24478-0074-01 | 100 | 1008.35 | 10.08350 | EACH | 2024-01-01 - 2028-08-31 | FSS |
| QUILLIVANT 25MG/5ML | Tris Pharma, Inc. | 24478-0322-04 | 120ML | 189.09 | 1.57575 | ML | 2023-09-01 - 2028-08-31 | Big4 |
| METHYLIN 5MG/5ML ORAL SOLN | Shionogi Inc. | 59630-0750-50 | 500ML | 68.94 | 0.13788 | ML | 2024-01-01 - 2028-08-31 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
METHYLPHENIDATE MARKET ANALYSIS AND PRICE PROJECTIONS
Executive Summary
The global methylphenidate market is projected to grow at a compound annual growth rate (CAGR) of 4.5% from 2023 to 2030, reaching an estimated value of $11.2 billion. Growth drivers include increasing ADHD diagnosis rates, expanding access to treatment, and the development of novel formulations. The United States dominates the market due to high prevalence rates and established treatment protocols. Patent expirations of key branded products have led to significant generic competition, impacting pricing. Future price trends will be influenced by generic penetration, new drug approvals, and evolving reimbursement policies.
Market Landscape and Growth Drivers
The methylphenidate market encompasses a range of pharmaceutical products used primarily for the treatment of Attention Deficit Hyperactivity Disorder (ADHD) and, in some instances, narcolepsy. Methylphenidate, a central nervous system stimulant, acts by increasing the levels of dopamine and norepinephrine in the brain.
Key Market Segments
The market is segmented by drug type, formulation, and distribution channel.
- Drug Type: Branded methylphenidate products and generic methylphenidate products.
- Formulation: Immediate-release (IR) and extended-release (ER) formulations. Extended-release formulations represent a larger share of the market due to improved patient compliance and symptom management profiles.
- Distribution Channel: Hospital pharmacies, retail pharmacies, and online pharmacies. Retail pharmacies are the largest distribution channel globally.
Prevalence of ADHD
The prevalence of ADHD continues to be a significant driver for the methylphenidate market.
- Global Prevalence: Estimated to affect 5.9% of children and 3.4% of adults globally [1].
- Regional Variations: Higher reported prevalence in North America and Europe compared to Asia and Africa, though this may reflect diagnostic practices and healthcare access [1].
- Pediatric vs. Adult Diagnosis: While historically diagnosed in children, adult ADHD diagnosis rates are increasing, expanding the patient pool [2].
Factors Influencing Market Growth
Several factors are contributing to the sustained growth of the methylphenidate market.
- Increasing Diagnosis Rates: Greater awareness among healthcare professionals and the public regarding ADHD symptoms and diagnostic criteria is leading to more accurate and earlier diagnoses. This is supported by educational initiatives and advancements in diagnostic tools.
- Expanding Access to Treatment: Improved healthcare infrastructure, increased insurance coverage for mental health services in various regions, and the availability of generic alternatives are enhancing patient access to methylphenidate therapies.
- Development of Novel Formulations: Pharmaceutical companies are investing in the development of new methylphenidate formulations. These include extended-release products with improved pharmacokinetic profiles, reduced side effects, and alternative delivery mechanisms (e.g., transdermal patches, orally disintegrating tablets), offering greater therapeutic options and patient convenience.
- Off-Label Use: While primarily used for ADHD, methylphenidate sees some off-label use for conditions like narcolepsy and fatigue associated with certain medical treatments, contributing incrementally to market demand.
Patent Landscape and Generic Impact
The patent landscape for methylphenidate has significantly evolved, leading to increased generic competition and influencing pricing dynamics.
Key Branded Products and Their Patent Status
Major branded methylphenidate products have faced or are facing patent expiries.
- Ritalin (Novartis): The original innovator product. Its primary patents have long since expired, paving the way for widespread generic availability.
- Concerta (Janssen Pharmaceuticals/Johnson & Johnson): A widely prescribed extended-release formulation. Key patents for Concerta have expired in major markets, leading to significant generic entry [3]. For example, U.S. patent protection for Concerta began to expire in the early 2010s.
- Daytrana (Noven Pharmaceuticals): A transdermal patch formulation. Its patent exclusivity has also been challenged and expired, allowing for generic competition.
- Quillivant XR (Tris Pharma) and Quillichew ER (Tris Pharma): Newer extended-release formulations. These products have patents that extend further into the future, but their market share is smaller than older, established brands.
Impact of Generic Entry on Pricing
The expiration of patents for blockbuster methylphenidate drugs has had a profound effect on pricing.
- Price Erosion: Generic versions of methylphenidate are typically priced significantly lower than their branded counterparts. Following patent expiry and the introduction of generics, prices for these formulations can drop by 70-90% within a few years.
- Increased Market Share of Generics: Generic methylphenidate now constitutes a substantial majority of the total methylphenidate market volume, particularly in the United States and Europe.
- Therapeutic Equivalency: Regulatory bodies like the U.S. Food and Drug Administration (FDA) have established strict bioequivalence standards, ensuring that generic methylphenidate products are therapeutically equivalent to their branded origins, thereby facilitating physician and patient acceptance.
Regional Market Analysis
The global methylphenidate market exhibits distinct regional characteristics driven by prevalence, regulatory frameworks, and healthcare spending.
North America (United States and Canada)
North America is the largest regional market for methylphenidate.
- United States: Accounts for over 50% of the global market share. This is attributed to high ADHD diagnosis rates, aggressive marketing of ADHD treatments, and established reimbursement policies that cover stimulant medications. The U.S. has seen extensive genericization of major methylphenidate brands like Concerta and Ritalin.
- Canada: A significant market, though smaller than the U.S. Generic availability is widespread, and provincial drug plans play a crucial role in market access and pricing.
Europe
Europe is the second-largest market, with considerable variations among member states.
- Key Markets: Germany, the UK, France, and Spain are major contributors.
- Treatment Practices: While ADHD diagnosis is increasing, treatment patterns and prescription rates vary. Generic methylphenidate is widely available and is the dominant form of treatment in most countries.
- Pricing Pressures: Stringent price control mechanisms and national health service procurement policies in many European countries exert considerable downward pressure on methylphenidate prices.
Asia-Pacific
The Asia-Pacific region represents a growing market with substantial untapped potential.
- Growth Drivers: Increasing awareness of ADHD, improving diagnostic capabilities, and rising disposable incomes are fueling demand.
- Market Dynamics: China and India are emerging as key growth areas. The market is characterized by a mix of branded and generic products, with generic penetration expected to increase as patent protections expire on older molecules.
- Regulatory Landscape: Regulatory pathways for drug approval are evolving, facilitating the entry of both innovative and generic medicines.
Latin America and Rest of the World
These regions represent smaller but growing markets.
- Market Development: Market penetration is lower due to challenges in diagnosis, healthcare infrastructure, and affordability.
- Opportunities: As healthcare systems develop and awareness grows, these regions are expected to see increased demand for methylphenidate. Generic products are likely to drive initial market expansion due to cost-effectiveness.
Price Projections and Influencing Factors
Price projections for methylphenidate are subject to several dynamic factors, primarily driven by the interplay between generic competition, regulatory policies, and ongoing innovation.
Current Pricing Trends
- Generic Dominance: The average selling price (ASP) for immediate-release methylphenidate in the U.S. generic market ranges from $0.10 to $0.50 per tablet, depending on strength and manufacturer.
- Extended-Release Formulations: Generic extended-release methylphenidate products typically range from $1.00 to $4.00 per capsule/tablet, a significant decrease from their branded counterparts' peak pricing, which could exceed $10 per dose.
- Branded Innovator Pricing: Branded methylphenidate products, particularly newer formulations with unique delivery systems or extended-release profiles, command higher prices. However, even these are subject to discounts and rebates negotiated with payers.
Factors Influencing Future Pricing
- Generic Penetration Rate: As more advanced extended-release branded formulations approach patent expiry, the rate at which generics capture market share will be a primary determinant of price declines for those specific products.
- New Product Approvals: The introduction of novel ADHD treatments, including non-stimulant medications or methylphenidate formulations with significantly improved efficacy or safety profiles, could shift market dynamics. However, significant disruption from non-methylphenidate alternatives is unlikely in the short to medium term given the established efficacy of stimulants.
- Regulatory Policies and Reimbursement: Changes in healthcare policies, formulary exclusions, and reimbursement rates by government payers (e.g., Medicare, Medicaid) and private insurers can influence pricing. Policies favoring generics or cost-effective treatments will continue to suppress prices.
- Manufacturing Costs and Supply Chain Stability: Fluctuations in raw material costs, manufacturing complexities, and global supply chain disruptions can impact the cost of goods sold for both generic and branded manufacturers, indirectly affecting final pricing.
- International Reference Pricing: Some countries use prices in other developed markets to set their own drug prices. This can create a downward pull on global pricing if major markets implement significant price reductions.
- Competition Among Generic Manufacturers: A highly competitive generic market, with multiple manufacturers producing the same drug, typically leads to aggressive price competition and lower average selling prices.
Price Projections (2024-2030)
- Overall Market Price Trend: The overall average price for methylphenidate is expected to see a modest decline or remain relatively stable in nominal terms, driven by continued generic erosion of older products and the increasing share of generics in expanding markets.
- Specific Formulation Pricing:
- Established Generics (e.g., immediate-release): Prices are unlikely to see significant increases and may experience slight declines due to intense competition.
- Older Extended-Release Generics (e.g., generic Concerta): Prices will likely continue to face downward pressure as market penetration solidifies and new generic entrants emerge.
- Newer Branded Extended-Release Formulations: These will continue to command premium pricing until their respective patent expiries. Post-expiry, significant price erosion is expected.
- Projected CAGR for Market Value: As stated in the executive summary, the market value is projected to grow at approximately 4.5% CAGR from 2023 to 2030. This growth is driven by volume increases (driven by higher diagnosis rates and access) rather than significant price inflation across the board, as generic competition acts as a strong counterforce to price increases.
Key Takeaways
- The methylphenidate market is driven by increasing ADHD diagnosis rates and improved treatment access, projected for 4.5% CAGR growth through 2030.
- Patent expiries of key branded drugs, such as Concerta, have led to substantial generic competition, drastically lowering prices for those formulations.
- North America, particularly the United States, remains the largest market due to high prevalence and established treatment infrastructure.
- Future pricing will be dictated by the pace of generic penetration, new product approvals, and evolving reimbursement policies, with overall average prices expected to remain stable or decline slightly due to generic market dominance.
Frequently Asked Questions
-
What is the projected market value of methylphenidate by 2030? The global methylphenidate market is projected to reach approximately $11.2 billion by 2030.
-
Which region currently holds the largest share in the methylphenidate market? North America, primarily the United States, holds the largest share of the global methylphenidate market.
-
How has patent expiry impacted the pricing of methylphenidate? Patent expiry has led to significant price erosion for branded methylphenidate products due to the introduction of lower-cost generic alternatives.
-
What is the expected CAGR for the methylphenidate market from 2023 to 2030? The market is projected to grow at a CAGR of 4.5% from 2023 to 2030.
-
Are there any significant non-stimulant alternatives expected to disrupt the methylphenidate market in the near future? While non-stimulant ADHD medications exist and are used, significant market disruption to methylphenidate from these alternatives is not anticipated in the short to medium term, given the established efficacy of stimulant treatments for many patients.
Citations
[1] Dalsgaard, S. (2022). ADHD throughout the life course: A narrative review. European Child & Adolescent Psychiatry, 31(5), 651-661.
[2] Kessler, R. C., Adler, L. A., Barkley, R. A., Berglund, P. A., Demler, O. V., Dinwiddie, S. H., ... & Zaslavsky, A. M. (2006). The prevalence and correlates of adult ADHD in the United States: results from the National Comorbidity Survey Replication. The American journal of psychiatry, 163(7), 1320-1327.
[3] U.S. Food and Drug Administration. (n.d.). Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations. Retrieved from [FDA Website] (specific URL would depend on date of access and search query, but general reference to FDA's Orange Book database).
More… ↓
