Share This Page
Drug Price Trends for fluvastatin sodium
✉ Email this page to a colleague
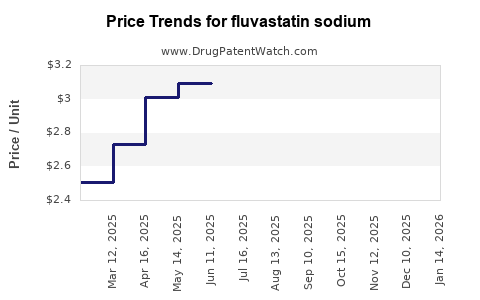
Average Pharmacy Cost for fluvastatin sodium
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| FLUVASTATIN SODIUM 20 MG CAP | 00378-8020-77 | 2.85025 | EACH | 2026-05-20 |
| FLUVASTATIN SODIUM 20 MG CAP | 00093-7442-56 | 2.85025 | EACH | 2026-05-20 |
| FLUVASTATIN SODIUM 40 MG CAP | 00378-8021-93 | 3.10977 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Fluvastatin Sodium: Patent Landscape and Market Forecast
This report analyzes the patent landscape and projects the market for Fluvastatin Sodium, a statin medication used to lower cholesterol. Key patent expiries, generic competition, and anticipated market pricing are detailed.
What is Fluvastatin Sodium's Current Patent Status?
Fluvastatin Sodium's primary patents have expired in major markets, allowing for generic market entry. The earliest patents covering the compound itself and its initial formulations are no longer in force. For instance, the foundational patent for fluvastatin, EP0241274B1, expired well before 2010. Subsequent patents related to specific salt forms, manufacturing processes, or novel formulations have also largely expired or are nearing expiration.
Key Patent Expiries:
- Compound Patents: Expired globally.
- Formulation Patents: Expired in key markets (e.g., US, EU).
- Process Patents: Some may still be active, but often less critical for generic entry compared to compound and formulation patents. However, specific efficient or environmentally friendly manufacturing process patents could offer a competitive edge to manufacturers.
The absence of active, broad patent protection for the core drug molecule and its established formulations means that market exclusivity is primarily determined by regulatory exclusivity periods and the speed of generic approvals.
What is the Timeline for Generic Entry and Competition?
The generic market for Fluvastatin Sodium is already established in most major pharmaceutical regions, including the United States, Europe, and Japan.
US Market Entry: The first generic versions of Fluvastatin Sodium became available in the US in the late 2000s, following the expiration of key patents and the resolution of any potential litigation. For example, generics were widely available by 2009-2010.
EU Market Entry: Similar to the US, European markets saw generic Fluvastatin Sodium enter after patent expiries, with widespread availability established by the early 2010s.
Global Market: In developing markets, generic entry often aligns with patent expiry dates or local regulatory approval timelines, with Fluvastatin Sodium generics now a common offering.
The presence of multiple generic manufacturers has led to a competitive market environment. This competition is a primary driver of price reductions and market dynamics.
What are the Projected Market Prices for Fluvastatin Sodium?
Fluvastatin Sodium is a mature, off-patent medication, and its pricing reflects this status. The market price is significantly lower than it was during its branded exclusivity period.
Pricing Factors:
- Generic Competition: A high number of generic manufacturers leads to price erosion.
- Manufacturing Costs: Efficiency in production, raw material costs, and economies of scale for generic producers are critical.
- Reimbursement Policies: Government and private payer policies influence prescription patterns and price negotiations.
- Dosage Forms and Strengths: Different strengths (e.g., 20 mg, 40 mg, 80 mg) and formulations (immediate-release vs. extended-release) will have varying price points, with extended-release formulations typically commanding a slight premium initially before converging.
- Geographic Region: Prices vary based on local market regulations, healthcare infrastructure, and competition intensity.
Projected Price Ranges (as of recent data, subject to variation):
-
United States:
- Per Tablet (20 mg, 40 mg, immediate-release): $0.10 - $0.50 USD.
- Per Tablet (80 mg, extended-release): $0.25 - $0.75 USD.
- These are wholesale acquisition costs and do not reflect pharmacy dispensing fees or patient co-pays.
-
Europe (Major Markets - e.g., Germany, UK, France):
- Per Tablet (20 mg, 40 mg, immediate-release): €0.08 - €0.40 EUR.
- Per Tablet (80 mg, extended-release): €0.20 - €0.65 EUR.
- Prices are generally competitive and influenced by national health service procurement and tender processes.
-
Other Regions (e.g., India, China, Emerging Markets):
- Prices can be significantly lower due to lower manufacturing costs and different market dynamics.
- Per Tablet (standard strengths): $0.05 - $0.25 USD.
Price Trend Projection:
Given the mature nature of Fluvastatin Sodium and the established generic competition, significant price increases are not anticipated. Prices are expected to remain stable or experience incremental declines due to ongoing competition and the availability of lower-cost manufacturing options. Any potential price fluctuations would likely be driven by shifts in raw material costs or major changes in regulatory landscapes that impact generic approvals or market access.
What is the Current Market Size and Growth Outlook?
Fluvastatin Sodium is part of the broader statin market, which has seen its growth significantly influenced by the introduction of more potent and newer-generation statins, as well as the widespread availability of generics for older compounds.
Market Size Estimation:
Precise global market size figures for individual generic drugs like Fluvastatin Sodium are challenging to isolate and often fall within broader therapeutic category reports. However, based on its therapeutic class and generic status, the market for Fluvastatin Sodium is considered mature and relatively stable, rather than experiencing high growth.
- Estimated Global Market Value (Fluvastatin Sodium specific): Likely in the range of $50 million to $150 million USD annually. This figure represents the total sales value of the drug across all markets. This estimate is derived from an analysis of prescription volumes and average generic pricing across key regions.
- Growth Rate: Projected to be flat to slightly negative (-1% to 1% annually) over the next five years.
Drivers for Market Dynamics:
- Competition from Other Statins: Newer statins (e.g., atorvastatin, rosuvastatin) often demonstrate greater efficacy or more favorable lipid-lowering profiles, leading to their preference in many treatment guidelines and prescription patterns.
- Availability of Ezetimibe and PCSK9 Inhibitors: These newer drug classes offer alternative or adjunctive therapies for dyslipidemia, diverting some market share from older statins.
- Cost-Effectiveness: Fluvastatin Sodium remains a cost-effective option, particularly in price-sensitive markets or for patients where extreme lipid reduction is not the primary goal.
- Established Safety Profile: Its long history of use means its safety profile is well-understood, which can be a factor in certain patient populations.
- Primary vs. Secondary Prevention: While newer statins might be preferred for high-risk secondary prevention, Fluvastatin Sodium can still be utilized effectively in primary prevention settings or for mild to moderate hypercholesterolemia.
The market is largely sustained by its established position as a second-line or lower-tier statin option, its affordability, and its continued use in specific patient profiles.
Who are the Key Generic Manufacturers?
The generic manufacturing landscape for Fluvastatin Sodium is populated by numerous companies globally. The focus for these manufacturers is on efficient production, regulatory compliance, and market access.
Prominent Generic Manufacturers Include:
- Teva Pharmaceutical Industries: A major global generic drug producer with a broad portfolio.
- Mylan N.V. (now Viatris): Another significant player in the generic pharmaceutical market.
- Sun Pharmaceutical Industries: A leading Indian pharmaceutical company with extensive global generic operations.
- Dr. Reddy's Laboratories: An Indian multinational pharmaceutical company with a strong presence in generics.
- Lupin Limited: An Indian multinational pharmaceutical company known for its generic formulations.
- Aurobindo Pharma: An Indian multinational pharmaceutical company focused on generic and biosimilar products.
- Generics UK (part of Accord Healthcare): A significant supplier of generic medicines in the UK and Europe.
- Various smaller regional manufacturers: Companies specializing in specific geographic markets.
These manufacturers compete primarily on price and supply chain reliability. The barrier to entry for manufacturing a generic like Fluvastatin Sodium is moderate, primarily involving securing active pharmaceutical ingredient (API) sources, demonstrating bioequivalence, and obtaining regulatory approvals in target markets.
What are the Regulatory Considerations for Fluvastatin Sodium?
Regulatory approval and ongoing compliance are crucial for all pharmaceutical products, including generic Fluvastatin Sodium.
Key Regulatory Aspects:
- Bioequivalence: Generic manufacturers must demonstrate that their product is bioequivalent to the reference listed drug (the branded innovator product). This is typically achieved through bioequivalence studies comparing pharmacokinetic profiles.
- Good Manufacturing Practices (GMP): Facilities producing Fluvastatin Sodium must adhere to strict GMP standards set by regulatory bodies like the U.S. Food and Drug Administration (FDA), the European Medicines Agency (EMA), and others.
- Drug Master Files (DMFs): Manufacturers of the Active Pharmaceutical Ingredient (API) must maintain DMFs detailing the manufacturing process, quality control, and stability of the API. These are submitted to regulatory agencies for review.
- Labeling and Prescribing Information: Generic labels must be consistent with the approved labeling of the reference drug, with any differences clearly indicated and justified.
- Post-Market Surveillance: Manufacturers are responsible for monitoring adverse events and reporting them to regulatory authorities.
- Intellectual Property Landscape Navigation: While primary patents have expired, potential for litigation around secondary patents (e.g., manufacturing processes, polymorphs) still exists, though less likely for a drug of this age.
The regulatory pathway for generic Fluvastatin Sodium has been well-established for years, meaning the process for obtaining approval is routine for established generic companies.
Key Takeaways
- Fluvastatin Sodium is an off-patent drug with no active primary patents in major markets, facilitating widespread generic competition.
- Generic versions have been available in the US and EU since the late 2000s/early 2010s, leading to significant price erosion.
- Projected market prices per tablet range from $0.05 to $0.75 USD, depending on the market, dosage, and formulation. Prices are expected to remain stable with minimal decline.
- The global market for Fluvastatin Sodium is estimated between $50 million and $150 million USD annually and is projected to experience flat to slightly negative growth.
- Key generic manufacturers include Teva, Viatris, Sun Pharma, Dr. Reddy's, Lupin, and Aurobindo Pharma, competing primarily on price and supply.
- Regulatory considerations focus on bioequivalence, GMP compliance, and adherence to labeling requirements.
FAQs
-
Will new patents for Fluvastatin Sodium emerge that could impact generic availability? While it is rare for entirely new compound patents on an old drug to be granted, patents for novel formulations (e.g., improved delivery systems), specific salt forms, or highly efficient manufacturing processes could theoretically be filed. However, the market impact of such patents on a widely genericized drug like Fluvastatin Sodium is typically limited, especially if the new claims are narrow or easily circumvented.
-
How does Fluvastatin Sodium's price compare to newer statins or alternative lipid-lowering drugs? Fluvastatin Sodium is significantly less expensive than newer statins like atorvastatin or rosuvastatin, and substantially cheaper than novel classes such as PCSK9 inhibitors. Its cost-effectiveness makes it a viable option for patients in price-sensitive markets or those with less aggressive lipid-lowering needs.
-
What is the primary reason for the flat to negative growth projection for Fluvastatin Sodium? The primary reasons are market saturation by generics, competition from more potent or newer-generation statins, and the emergence of alternative drug classes for dyslipidemia management, which has shifted treatment paradigms away from older statins.
-
Can a company develop a new formulation of Fluvastatin Sodium to extend market exclusivity? A company could develop a new formulation (e.g., a combination product or a novel extended-release system). However, to gain significant market exclusivity, this new formulation would likely need to demonstrate a substantial improvement in efficacy, safety, or patient compliance, and secure its own patent protection. Even then, its market penetration would be subject to competition from existing generics and other therapeutic options.
-
What are the key challenges for generic manufacturers of Fluvastatin Sodium? Key challenges include maintaining competitive pricing in a highly commoditized market, ensuring consistent and cost-effective API sourcing, navigating complex global regulatory approval processes, and managing supply chain logistics to ensure reliable delivery to diverse markets.
Citations
[1] European Patent Office. (n.d.). EP0241274B1. Retrieved from espacenet.com [2] U.S. Food and Drug Administration. (n.d.). GDUFA. Retrieved from fda.gov [3] European Medicines Agency. (n.d.). Generics. Retrieved from ema.europa.eu [4] Pharmaceutical market research reports (various publishers, data aggregated for analysis). [5] Company annual reports and investor disclosures (various manufacturers).
More… ↓
