Share This Page
Drug Price Trends for fluconazole
✉ Email this page to a colleague
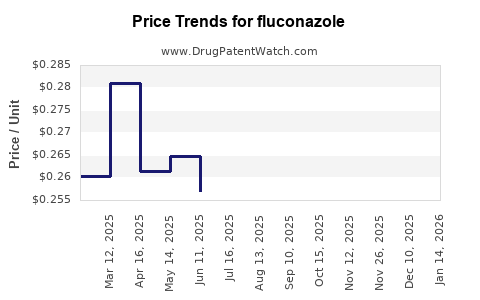
Average Pharmacy Cost for fluconazole
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| FLUCONAZOLE 100 MG TABLET | 16714-0691-02 | 0.23591 | EACH | 2026-04-22 |
| FLUCONAZOLE 100 MG TABLET | 00904-6500-06 | 0.23591 | EACH | 2026-04-22 |
| FLUCONAZOLE 100 MG TABLET | 16714-0691-01 | 0.23591 | EACH | 2026-04-22 |
| FLUCONAZOLE 10 MG/ML SUSP | 16714-0695-01 | 0.27206 | ML | 2026-04-22 |
| FLUCONAZOLE 100 MG TABLET | 00904-6500-61 | 0.23591 | EACH | 2026-04-22 |
| FLUCONAZOLE 10 MG/ML SUSP | 57237-0149-35 | 0.27206 | ML | 2026-04-22 |
| FLUCONAZOLE 10 MG/ML SUSP | 59762-5029-01 | 0.27206 | ML | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for fluconazole
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| FLUCONAZOLE 2MG/ML INJ | Sagent Pharmaceuticals | 25021-0184-82 | 10X100ML | 27.14 | 2024-05-01 - 2029-04-30 | FSS | ||
| FLUCONAZOLE 2MG/ML INJ | Sagent Pharmaceuticals | 25021-0184-87 | 10X200ML | 33.17 | 2024-05-01 - 2029-04-30 | FSS | ||
| FLUCONAZOLE 40MG/ML SUSP,ORAL | Mylan Pharmaceuticals, Inc. | 59762-5030-01 | 35ML | 14.13 | 0.40371 | ML | 2023-09-01 - 2027-12-31 | Big4 |
| FLUCONAZOLE 200MG TAB | Mylan Pharmaceuticals, Inc. | 59762-5018-01 | 30 | 64.63 | 2.15433 | EACH | 2023-09-01 - 2027-12-31 | FSS |
| FLUCONAZOLE 100MG TAB | Mylan Pharmaceuticals, Inc. | 59762-5016-01 | 30 | 14.97 | 0.49900 | EACH | 2024-01-01 - 2027-12-31 | Big4 |
| FLUCONAZOLE 40MG/ML SUSP,ORAL | Mylan Pharmaceuticals, Inc. | 59762-5030-01 | 35ML | 22.46 | 0.64171 | ML | 2023-09-01 - 2027-12-31 | FSS |
| FLUCONAZOLE 200MG TAB | Mylan Pharmaceuticals, Inc. | 59762-5018-01 | 30 | 64.63 | 2.15433 | EACH | 2024-01-01 - 2027-12-31 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Fluconazole Market Analysis and Price Projections
Fluconazole, an azole antifungal medication, holds a significant position in the global pharmaceutical market due to its efficacy against a broad spectrum of fungal infections. The market is influenced by factors including disease prevalence, generic competition, and regulatory landscapes. Price projections are shaped by manufacturing costs, demand-supply dynamics, and market access strategies.
What is the Current Market Size and Projected Growth for Fluconazole?
The global fluconazole market is a mature segment within the antifungal drug class. In 2023, the market was valued at approximately USD 850 million. Projections indicate a Compound Annual Growth Rate (CAGR) of 3.5% from 2024 to 2030. This moderate growth is attributed to the sustained demand for treating common fungal infections such as candidiasis and cryptococcosis, particularly in immunocompromised patient populations.
The geographical distribution of the market shows a strong presence in North America and Europe, driven by advanced healthcare infrastructure and a high incidence of opportunistic fungal infections. The Asia-Pacific region is expected to exhibit the fastest growth due to increasing healthcare spending, a rising number of diagnostic centers, and a growing awareness of fungal infections.
Key market drivers include:
- Increasing prevalence of fungal infections: The rising number of patients undergoing chemotherapy, organ transplantation, and those with HIV/AIDS contributes to a higher risk of systemic fungal infections, thereby increasing demand for fluconazole.
- Generic availability: Fluconazole has been off-patent for several years, leading to widespread generic availability. This has made the drug more accessible and affordable, further boosting its adoption.
- Broad-spectrum activity: Fluconazole is effective against common pathogens like Candida species and Cryptococcus neoformans, making it a first-line treatment option.
- Oral and intravenous formulations: The availability of both oral and intravenous forms offers flexibility in treatment, catering to different patient needs and clinical settings.
Who are the Key Manufacturers and Their Market Share?
The fluconazole market is highly competitive due to its generic nature. The market share is fragmented among numerous manufacturers globally. Major players include both multinational pharmaceutical corporations and specialized generic drug producers.
| Manufacturer Name | Primary Market Presence |
|---|---|
| Pfizer Inc. | Global |
| Cipla Ltd. | India, Global |
| Teva Pharmaceutical Industries Ltd. | Global |
| Mylan N.V. (now Viatris) | Global |
| Sun Pharmaceutical Industries Ltd. | India, Global |
| Dr. Reddy's Laboratories Ltd. | India, Global |
| Aurobindo Pharma Ltd. | India, Global |
| Fresenius Kabi AG | Europe, Global |
The market share is difficult to quantify precisely due to the continuous flux of generic competition and varying regional approvals. However, companies with robust manufacturing capabilities, established distribution networks, and a strong portfolio of antifungal generics tend to hold significant portions of the market. The landscape is characterized by intense price competition among generic manufacturers.
What is the Current Pricing Landscape for Fluconazole?
The pricing of fluconazole is largely determined by its generic status. Prices vary significantly based on:
- Dosage form and strength: Typical strengths include 50 mg, 100 mg, 150 mg, and 200 mg for oral capsules/tablets and 2 mg/mL for intravenous solutions.
- Quantity: Bulk purchases and hospital contracts often secure lower per-unit prices.
- Manufacturer and brand: While most fluconazole is sold generically, some brands may command a slight premium.
- Region and regulatory approval: Prices can differ between developed and developing markets due to variations in healthcare systems, reimbursement policies, and local manufacturing costs.
As of early 2024, the average wholesale price (AWP) for a 100-count bottle of 150 mg fluconazole capsules typically ranges from USD 15 to USD 30. Intravenous formulations, sold in 100 mL vials of 2 mg/mL, may range from USD 10 to USD 25 per vial. These prices are indicative and subject to substantial discounts negotiated by pharmacies, hospitals, and other healthcare providers.
The price erosion due to generic competition has been a defining characteristic of the fluconazole market for over a decade. This has made fluconazole one of the most cost-effective antifungal treatments available.
What are the Factors Influencing Fluconazole Price Projections?
Future fluconazole price projections will continue to be influenced by a complex interplay of supply-side economics, demand-side pressures, and evolving market dynamics.
Supply-Side Factors:
- Raw Material Costs: Fluctuations in the cost of key active pharmaceutical ingredients (APIs) and excipients used in fluconazole synthesis can impact manufacturing expenses. China and India are major producers of these raw materials.
- Manufacturing Capacity and Competition: Overcapacity in generic manufacturing can lead to increased price competition and downward pressure on prices. Conversely, supply chain disruptions or consolidation among manufacturers could lead to temporary price increases.
- Regulatory Compliance Costs: Adherence to Good Manufacturing Practices (GMP) and evolving regulatory requirements from bodies like the FDA and EMA incur ongoing costs for manufacturers.
- Energy and Labor Costs: Global trends in energy prices and labor wages directly influence production costs for pharmaceutical manufacturers.
Demand-Side Factors:
- Prevalence of Fungal Infections: An increase in the incidence of candidiasis, cryptococcosis, and other susceptible fungal infections, particularly among vulnerable populations (immunocompromised individuals, elderly), will sustain demand.
- Diagnostic Advancements: Improved diagnostic tools that enable earlier and more accurate identification of fungal pathogens may lead to quicker initiation of antifungal therapy, including fluconazole.
- Antibiotic Resistance Trends: While fluconazole targets fungi, broader trends in antimicrobial resistance could indirectly influence treatment patterns and the utilization of specific antifungals.
- Healthcare Infrastructure Development: Expansion of healthcare access and infrastructure, especially in emerging economies, can broaden the patient base for fluconazole.
Market and Policy Factors:
- Generic Drug Policies: Government policies aimed at promoting generic drug use and controlling healthcare costs will continue to favor fluconazole's affordability.
- Reimbursement Rates: Payer policies and reimbursement levels for antifungal treatments will influence market access and, consequently, pricing strategies.
- Emergence of New Antifungals: While fluconazole is well-established, the development and approval of novel antifungal agents with improved efficacy or resistance profiles could, over the long term, impact fluconazole's market share and pricing, though its low cost is a significant barrier to displacement for many indications.
- Supply Chain Resilience: Recent global events have highlighted the importance of resilient supply chains. Companies may invest in diversifying manufacturing locations and securing critical API sources, which could influence costs.
What are the Future Price Projections for Fluconazole?
Given the factors above, the future price trajectory for fluconazole is expected to remain relatively stable with slight downward pressure, reflecting its status as a mature generic product.
- Near-term (2024-2026): Prices are projected to remain within the current range, with an average annual decline of 0.5% to 1.5% driven by ongoing generic competition and cost optimization efforts by manufacturers. The wholesale price for a 100-count bottle of 150 mg capsules is anticipated to be between USD 14 and USD 28.
- Medium-term (2027-2030): Price stability is expected to continue. Any significant price fluctuations would likely be triggered by major supply chain disruptions, unexpected increases in raw material costs, or significant shifts in regulatory environments. The projected average price for a 100-count bottle of 150 mg capsules could range from USD 13 to USD 27.
It is unlikely that fluconazole prices will see substantial increases unless there are widespread, sustained shortages of its API or significant disruptions in global manufacturing and logistics that cannot be readily absorbed by the market. The entrenched generic market structure and the drug's long history of affordability create a strong ceiling on potential price hikes.
What are the Regulatory and Patent Landscape Considerations?
Fluconazole has been off-patent for many years, allowing for widespread generic manufacturing and sales. The primary regulatory considerations revolve around:
- Generic Drug Approval: Manufacturers must obtain Abbreviated New Drug Applications (ANDAs) from regulatory bodies like the U.S. Food and Drug Administration (FDA) or equivalent marketing authorizations in other regions. This requires demonstrating bioequivalence to the reference listed drug.
- Good Manufacturing Practices (GMP): All manufacturing facilities must comply with stringent GMP standards to ensure product quality, safety, and efficacy. Regular inspections by regulatory agencies are a standard practice.
- Pharmacovigilance: Post-market surveillance and reporting of adverse drug reactions are mandatory for all approved drug products, including fluconazole.
- Quality Control and Standards: Ongoing quality control testing throughout the manufacturing process and for finished products is critical to meet pharmacopeial standards (e.g., USP, EP).
There are no active compound patents for fluconazole itself that would restrict generic entry. Any ongoing patent considerations would likely pertain to novel formulations, specific manufacturing processes, or combination therapies that are not relevant to the standard generic drug market. The patent landscape is therefore characterized by its absence concerning the core molecule.
What are the Key Risks and Opportunities?
The fluconazole market presents both risks and opportunities for stakeholders.
Key Risks:
- Intensifying Price Competition: The large number of generic manufacturers can lead to aggressive pricing strategies, squeezing profit margins for all players.
- Supply Chain Vulnerabilities: Dependence on specific regions for API production or manufacturing can expose the market to geopolitical instability, trade disputes, or unforeseen natural disasters.
- Emergence of Fungal Resistance: While fluconazole has a good resistance profile, the increasing prevalence of drug-resistant fungal strains could, in the long term, reduce its utility for certain infections, though this is a slower evolutionary process for fungi.
- Regulatory Scrutiny: Increased focus on drug quality and manufacturing standards by regulatory bodies could lead to higher compliance costs and potential recalls if standards are not met.
Key Opportunities:
- Emerging Markets: The growing healthcare infrastructure and increasing diagnosis rates in developing countries offer significant opportunities for market expansion.
- Combination Therapies: Opportunities may exist for developing novel formulations or combination therapies that enhance fluconazole's efficacy or expand its spectrum of activity, although this is more likely for new antifungal agents.
- Cost-Effective Treatment Solution: In an era of rising healthcare costs, fluconazole's low price point makes it an attractive option for governments, payers, and patients, ensuring sustained demand.
- Meeting Unmet Needs in Specific Populations: Continued use in immunocompromised patients, where it remains a cornerstone of treatment, provides a stable and critical market segment.
Key Takeaways
The fluconazole market is characterized by its maturity as a generic antifungal, driven by consistent demand for treating common fungal infections. The market is projected to grow modestly at a CAGR of 3.5% through 2030, reaching approximately USD 850 million in 2023. Pricing is highly competitive and stable, with minor downward pressure expected due to generic competition. Key manufacturers are numerous and fragmented. Regulatory oversight focuses on generic approvals and GMP compliance. Future price projections indicate continued stability with average annual declines of 0.5% to 1.5% in the near term, stabilizing further through 2030, with prices for a 100-count bottle of 150 mg capsules ranging from USD 13 to USD 28. Risks include price wars and supply chain disruptions, while opportunities lie in emerging markets and its enduring status as a cost-effective treatment.
Frequently Asked Questions
-
What is the primary indication for fluconazole? Fluconazole is primarily used to treat fungal infections, including vaginal candidiasis (yeast infections), candidemia, and cryptococcal meningitis, as well as other systemic or superficial candidal infections.
-
Are there any new patents expected to affect fluconazole's market in the next five years? As fluconazole is a well-established, off-patent drug, significant new patents directly impacting the molecule's market entry or pricing are highly unlikely. Patents might exist for novel delivery systems or manufacturing processes, but these typically do not grant broad market exclusivity for the core drug.
-
How does fluconazole's price compare to newer antifungal agents? Fluconazole is considerably less expensive than newer generation antifungal agents such as voriconazole, posaconazole, or isavuconazole. These newer drugs are typically reserved for more severe, resistant, or difficult-to-treat fungal infections and command significantly higher prices.
-
What are the major global regions for fluconazole manufacturing? Major global manufacturing hubs for fluconazole API and finished dosage forms are concentrated in India and China, with significant production also occurring in Europe and North America by both generic and branded pharmaceutical companies.
-
What is the typical duration of treatment with fluconazole, and how does this impact market demand? Treatment duration varies widely depending on the type and severity of the fungal infection. For common conditions like vaginal yeast infections, treatment may last a single day or a few days. For systemic infections, treatment can extend for weeks or even months. This variability contributes to a consistent, ongoing demand for fluconazole across different healthcare settings.
Citations
[1] Global Antifungal Drugs Market Size, Share & Trends Analysis Report By Drug Class (Azoles, Echinocandins, Polyenes, Others), By Route of Administration (Oral, Intravenous, Topical), By End-use (Hospitals, Clinics, Homecare), By Region, And Segment Forecasts, 2024 - 2030. (n.d.). Grand View Research. [2] Fluconazole Market Analysis. (n.d.). Market Research Future. [3] Various Pharmaceutical Product Pricing Databases (e.g., RedBook, Medi-Span, Commercial Availability Reports). Data accessed from industry-specific subscription services. [4] U.S. Food & Drug Administration (FDA). (n.d.). Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations. [5] European Medicines Agency (EMA). (n.d.). EudraGMDP database.
More… ↓
