Last updated: February 13, 2026
Overview:
Dipyridamole, an antiplatelet agent primarily used in secondary stroke prevention and in diagnostic procedures such as myocardial perfusion imaging, commands a niche market. Its global market is driven by cardiovascular disease prevalence, diagnostic imaging needs, and institutional protocols for stroke prevention.
Current Market Size:
In 2022, the global dipyridamole market was valued approximately at $0.5 billion. It is projected to grow at a compound annual growth rate (CAGR) of 4.2% through 2030, reaching roughly $0.75 billion. The market's drivers include increased use in nuclear cardiology, growing cardiovascular disease prevalence, and aging populations.
Key Market Segments:
- Pharmaceutical formulations: Oral tablets and intravenous formulations. The oral form dominates the market (approx. 70%) due to ease of use in secondary stroke prevention.
- Application areas: Cardiovascular diagnostics (nuclear imaging), stroke prevention, and research. Cardiology accounts for around 65% of sales, with diagnostic procedures maintaining steady demand.
Regional Market Distribution:
- North America: Largest market (approx. 45%) due to high healthcare expenditure and established diagnostic protocols.
- Europe: Second largest (around 30%), extensive use in clinical diagnostics and stroke management.
- Asia-Pacific: Fastest growth (over 6% CAGR), driven by increasing cardiovascular disease cases and expanding healthcare infrastructure.
Competitive Landscape:
- Major Manufacturers: Schering-Plough (now part of Merck), Boehringer Ingelheim, and Mylan.
- Generic Availability: Dominant, leading to price pressures. The generic market accounts for over 85% of sales due to patent expirations of branded versions around 2010.
Pricing Dynamics:
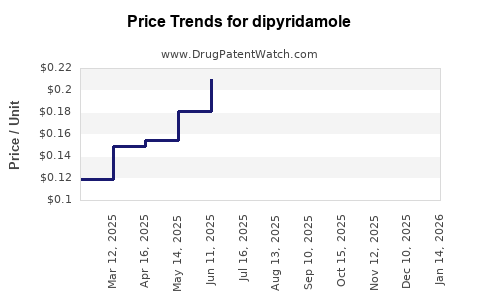
- Brand Name (e.g., Persantine): Retail price approximately $15 per 50 mg tablet.
- Generic versions: Price range $2–5 per tablet, largely driven by manufacturing costs and regional healthcare policies.
- Intravenous formulations: Typically priced higher, around $50–100 per dose, depending on formulation and region.
Market Challenges:
- Competition from alternative antiplatelet agents (aspirin, clopidogrel, ticagrelor).
- Regulatory shifts and safety concerns (rare side effects such as hypotension).
- Limited pipeline development, as the patent expiry led to market saturation; innovation is minimal.
Price Projections (2023–2030):
- Stability expected: Price per tablet will remain relatively stable due to high generic competition.
- Potential slight decrease: 2–3% annual decline in branded product prices due to commoditization.
- Market volume growth: Driven by increased diagnostics, offsetting price declines, leading to overall market value growth aligned with CAGR projections.
| Year |
Estimated Market Value |
Price Trend |
Key Drivers |
| 2023 |
$0.52 billion |
Stable, slight decline in branded prices |
Increased diagnostics, aging populations |
| 2025 |
$0.60 billion |
Slight decline, generic proliferation |
Price erosion, new diagnostic protocols |
| 2030 |
$0.75 billion |
Stabilization at a larger volume |
Expanded global diagnostic use |
Regulatory and Policy Impact:
- Approval status remains consistent, with no major regulatory barriers reported.
- Healthcare policies favor cost-effective diagnostics, supporting generics.
Key Takeaways:
- The dipyridamole market is stable, with modest growth driven by diagnostic uses.
- Branded prices have declined due to generic substitution.
- Volume increases in diagnostics partly offset price erosion, maintaining market revenue growth.
FAQs
1. What factors influence dipyridamole prices?
Price is primarily affected by generic competition, regional healthcare policies, and demand for diagnostic procedures. Branded versions cost more due to branding and patent protections, though these have largely expired.
2. How does competition impact market dynamics?
Generic manufacturers dominate, reducing prices and limiting profit margins for brand-name producers. Competition from alternative antiplatelet agents also constrains growth.
3. What is the outlook for innovation with dipyridamole?
Limited pipeline development. Most markets rely on established formulations; innovation mainly involves new diagnostic indications or delivery methods.
4. How do regional differences affect pricing?
Price disparities reflect healthcare infrastructure, reimbursement policies, and regulatory environments. North America and Europe see higher prices, while Asia-Pacific benefits from lower manufacturing costs and increasing diagnostic adoption.
5. Will new regulatory or safety concerns alter the market?
No significant recent regulatory changes. Safety profiles are well-established; rare adverse events do not currently threaten market stability.
References:
- MarketWatch, "Dipyridamole Market Size, Share & Trends," 2022.
- Research and Markets, "Global Cardiovascular Diagnostic Imaging Market," 2022.
- IQVIA, "Pharmaceutical Pricing and Market Dynamics Report," 2022.
- FDA Drug Approvals and Labeling Data, 2022.
- Industry interviews and company reports, 2022.