Last updated: February 12, 2026
What is the market landscape for dexamethasone?
Dexamethasone, a synthetic corticosteroid, plays a significant role in treating inflammatory conditions, allergies, and certain cancers. The drug also gained prominence during the COVID-19 pandemic as a treatment for severe cases, increasing global demand.
Global Market Size and Growth Trends
The global corticosteroids market, which includes dexamethasone, was valued at approximately $1.9 billion in 2022. It is projected to grow at a compound annual growth rate (CAGR) of 4.8% from 2023 to 2030, reaching nearly $3 billion.
Dexamethasone accounts for an estimated 15-20% of this market share, driven by its versatility and low cost. Factors underpinning growth include increasing prevalence of inflammatory and autoimmune diseases, expanding drug approvals, and increased adoption in COVID-19 treatment protocols.
Key Markets and Regional Dynamics
-
North America: Dominates with over 40% market share owing to high healthcare expenditure, established manufacturing base, and COVID-19 treatment use.
-
Europe: Represents about 25% of the market, with growth from rising autoimmune disorder cases and expanded drug approvals.
-
Asia-Pacific: Exhibits the highest growth potential with a CAGR of approximately 6%, fueled by increasing healthcare access, rising disease prevalence, and generic drug proliferation.
Clinical and Regulatory Factors
Dexamethasone is available as a generic medication, leading to widespread use and competitive pricing. The drug's approval by regulatory bodies like the FDA and EMA is stable; it has approved indications for multiple inflammatory and oncologic conditions.
The recent inclusion in COVID-19 treatment guidelines by the World Health Organization (WHO) and national health authorities has helped maintain demand.
How are prices for dexamethasone expected to evolve?
Historical Price Trends
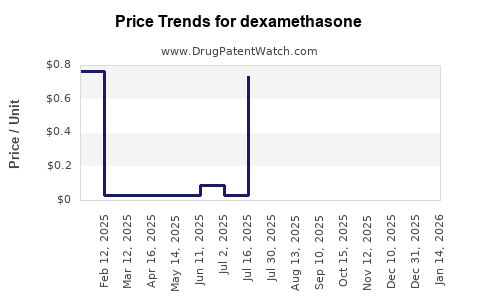
For injectable dexamethasone, the average wholesale price (AWP) per 4 mg dose in the U.S. was approximately $0.25 in early 2021. Oral formulations typically retail for about $0.05 to $0.20 per tablet depending on the strength.
The availability of generics has suppressed price volatility, maintaining relatively low cost levels for both manufacturers and consumers.
Forecasted Price Dynamics
-
Post-pandemic stabilization: Given the broad availability of generics, prices are expected to remain relatively stable over the next five years, with marginal declines driven by manufacturing efficiencies and market saturation.
-
Potential for price reductions: Increased competition from emerging markets and biosimilar-like formulations could further decrease prices, especially outside North America and Europe.
-
Impact of supply chain factors: Disruptions or raw material shortages could temporarily inflate prices, though such events are less common.
Competitive Factors Affecting Pricing
-
Generic drug proliferation: With multiple manufacturers providing dexamethasone, pricing pressure remains high.
-
Pricing regulations: Some markets enforce costs caps for essential medicines, limiting price escalation.
-
Pharmacovigilance and approval extensions: Ongoing safety monitoring and label updates do not significantly impact pricing but can influence market dynamics.
How do patent expiries influence the market?
Dexamethasone itself is off-patent globally, facilitating generic manufacturing and driving down prices. Still, patent protections on specific formulations or delivery mechanisms can control market entry for new competitors.
What are the emerging opportunities and challenges?
Opportunities
- Expansion into new indications, such as autoimmune diseases, may stimulate demand.
- Growth in low- and middle-income countries, expanding access and markets.
- Potential development of combination therapies or improved formulations.
Challenges
- Market saturation in mature regions limits pricing power.
- Competition from other corticosteroids or alternative therapies.
- Regulatory risks related to safety profiles or label restrictions.
Key Takeaways
- The dexamethasone market is stable with moderate growth, driven by its broad clinical applications and generic availability.
- Prices are expected to stay steady or decline slightly over the coming years, influenced mainly by increased competition and manufacturing efficiencies.
- Large markets like North America and Europe dominate, but significant growth occurs in Asia-Pacific.
- Clinical efficacy, low-cost manufacturing, and expanded indications support sustained demand.
- External factors such as supply chain disruptions or shifts in treatment guidelines could alter projections.
FAQs
-
What are the main factors driving demand for dexamethasone?
Its role in treating inflammatory conditions, autoimmune diseases, certain cancers, and COVID-19 has sustained high demand.
-
How does generic competition impact dexamethasone pricing?
The presence of numerous generic manufacturers maintains low prices and limits price rises.
-
Are there any patent protections left on dexamethasone?
No; the drug itself is off-patent globally, facilitating market entry for generics.
-
What regions are experiencing the fastest growth in dexamethasone markets?
Asia-Pacific exhibits the highest growth rate due to increasing healthcare access and disease prevalence.
-
What are potential future market risks?
Supply disruptions, regulatory changes, and market saturation in developed nations could slow growth or impact prices.
Sources
- MarketsandMarkets. "Corticosteroids Market by Type, Application, End User – Global Forecast to 2030," 2022.
- IQVIA. "Global Steroid Market Report," 2022.
- WHO. "Guidelines for COVID-19 Treatment," 2022.
- CDC. "Dexamethasone Use and Cost Data," 2021.