Last updated: February 19, 2026
Carbromal, an older sedative and hypnotic agent, faces a declining market due to the development of safer and more effective alternatives. Its therapeutic niche is narrow, primarily limited to specific off-label or historical uses where newer agents are unsuitable or unavailable. The global market for carbromal is small and projected to contract further.
What is Carbromal and Its Current Therapeutic Landscape?
Carbromal (acetylcarbromal) is a brominated ureide derivative. Historically, it was used for its sedative and hypnotic properties. Its mechanism of action is not fully elucidated but is believed to involve central nervous system depression, similar to barbiturates.
Current therapeutic use of carbromal is significantly limited. Regulatory bodies in major pharmaceutical markets, including the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA), do not list carbromal as an approved drug for any indication. Its availability is largely restricted to certain niche markets, primarily for compounding pharmacies or as a component in historical formulations.
Key Characteristics:
- Chemical Class: Brominated ureide.
- Primary Historical Use: Sedative, hypnotic.
- Current Regulatory Status: Not approved for marketing in major markets (e.g., U.S., EU).
- Availability: Limited, primarily through compounding or specialized distributors.
The lack of formal approval in major markets stems from concerns regarding its safety profile, including potential for toxicity, dependence, and the availability of superior alternatives with better efficacy and safety margins.
What is the Current Market Size and Demand for Carbromal?
The global market for carbromal is not actively tracked by major market research firms due to its limited and fragmented nature. Available data suggests a market size in the low millions of U.S. dollars annually. Demand is driven by a small number of specialized compounding pharmacies and research institutions.
Estimated Market Size:
- Global Annual Revenue: Estimated to be between $1 million and $3 million USD.
- Key Demand Drivers:
- Compounding of specialized formulations.
- Research and development activities requiring historical reference compounds.
- Limited use in regions with less stringent pharmaceutical regulations.
The demand is highly inelastic concerning price due to the specialized nature of its application. Users requiring carbromal for specific purposes often have few, if any, alternatives. However, the absolute volume of demand remains very low.
What are the Primary Applications and Uses of Carbromal?
Carbromal's primary historical application was as a sedative and hypnotic. However, due to safety concerns and the advent of benzodiazepines and newer sleep aids, its use in mainstream clinical practice has ceased in most developed nations.
Current and Historical Applications:
- Sedation and Hypnosis (Historical): Used for the short-term management of insomnia and anxiety. This application is largely obsolete in contemporary medicine.
- Compounding Pharmacies: Some compounding pharmacies may still produce formulations containing carbromal for specific patient needs where conventional medications are not suitable or tolerated. This is often for very niche, individualized prescriptions.
- Research and Analytical Standards: Carbromal may be used as a reference standard in forensic toxicology, analytical chemistry, and pharmaceutical research to identify or quantify its presence in samples or to study its properties.
- Veterinary Medicine (Limited/Historical): Some older veterinary texts might mention its use, but this is not a current or significant application.
The vast majority of current demand originates from compounding and research sectors, representing a fraction of its historical pharmaceutical market.
What are the Key Factors Influencing Carbromal Pricing?
Carbromal pricing is influenced by several factors, primarily related to its limited production volume, specialized supply chain, and regulatory status. Unlike high-volume, widely approved pharmaceuticals, carbromal does not benefit from economies of scale.
Pricing Influencers:
- Manufacturing Costs: Small-batch synthesis and purification processes for niche compounds are inherently more expensive per unit than large-scale pharmaceutical production.
- Supply Chain Complexity: Sourcing raw materials and distributing a low-volume specialty chemical can incur higher logistical costs.
- Regulatory Compliance (for distributors/compounders): While carbromal itself may not be approved for general marketing, companies handling it must adhere to regulations concerning controlled substances, hazardous materials, and good manufacturing practices if applicable.
- Lack of Competition: With very few manufacturers or suppliers globally, competition is minimal, allowing for higher price points.
- Demand from Niche Markets: The inelastic demand from compounding pharmacies and research entities, who require the substance regardless of cost for specific applications, supports higher pricing.
- Purity and Grade: The price will vary based on the required purity for analytical standards versus raw material for compounding.
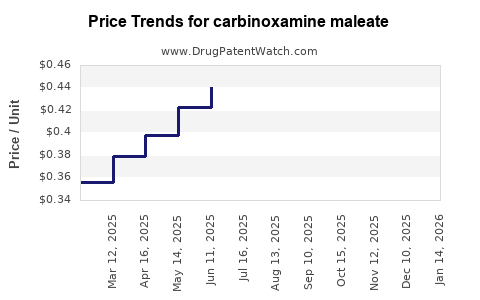
Typical Price Range (per gram, estimated):
- Bulk Pharmaceutical Grade (rarely traded): $50 - $150 USD
- Analytical Standard Grade: $100 - $300+ USD
These prices are indicative and can fluctuate based on specific supplier, quantity, and purity requirements.
What are the Future Market Trends and Price Projections for Carbromal?
The future market for carbromal is projected to remain small and is likely to experience a gradual decline. The absence of new therapeutic indications, coupled with ongoing regulatory scrutiny and the continued development of superior drug classes, limits any potential for market expansion.
Market Trends:
- Continued Decline in Therapeutic Use: As newer, safer sedatives and hypnotics become more prevalent and accessible, the rationale for using carbromal diminishes.
- Stable but Low Demand from Compounding/Research: While the therapeutic market shrinks, demand from compounding pharmacies and research institutions is expected to remain relatively stable at low levels. These sectors may continue to require carbromal for specific, unchanging needs.
- Potential for Increased Regulatory Oversight: Although already not widely approved, any resurgence in adverse event reporting or quality concerns could lead to further restrictions on its availability.
- Consolidation of Suppliers: The low market volume might lead to consolidation among the few existing suppliers, potentially impacting price stability.
Price Projections:
Given the existing market dynamics, significant price increases are unlikely. The price is more likely to remain stable or experience a slight upward trend due to incremental increases in manufacturing and regulatory compliance costs. A substantial decrease in price is improbable without a significant shift in production or demand.
- Short-Term (1-3 years): Stable to a slight increase (0-5% annually) driven by rising production costs.
- Long-Term (3-5+ years): Continued stability with potential for slight increases. The overall market volume will likely continue its slow contraction.
The market's trajectory is dictated by its obsolescence as a mainstream pharmaceutical agent and its sustained, albeit minuscule, utility in specialized applications.
What are the Regulatory Considerations for Carbromal?
Regulatory considerations for carbromal are significant, primarily due to its historical classification and the safety profile associated with older sedative-hypnotic drugs. In major pharmaceutical markets, it is not an approved drug, meaning it cannot be marketed for therapeutic purposes through standard regulatory pathways.
Key Regulatory Aspects:
- FDA (U.S.): Carbromal is not listed on the FDA's approved drug products list (Orange Book). It is not permitted for marketing as a new drug or as a generic equivalent for any indication. Any U.S. availability would likely be through compounding pharmacies operating under specific state and federal regulations, or for research purposes via regulated channels.
- EMA (Europe): Similar to the U.S., carbromal is not an authorized medicinal product within the European Union. Its use would be highly restricted, possibly confined to investigational purposes or specific national exemptions for compounded preparations where permitted by member state legislation.
- Controlled Substance Status: While not universally scheduled as a controlled substance globally, its sedative properties mean that some jurisdictions may impose specific controls on its handling, distribution, and prescription, particularly if it were ever to be considered for therapeutic use or if adverse events triggered scrutiny. For example, its classification might fall under broader categories of regulated chemicals or drug precursors depending on the country.
- Compounding Regulations: Pharmacies compounding carbromal must adhere to Good Compounding Practices (GCP), relevant state and federal regulations (e.g., U.S. Pharmacopeia <795> and <797> standards if applicable to the preparation), and may require specific licenses for handling certain chemical ingredients.
- International Regulations: Regulations vary significantly by country. In regions with less developed pharmaceutical regulatory frameworks, availability might be less restricted, but this also carries higher risks of substandard quality or unsafe use.
The lack of formal approval and the associated safety concerns mean that any entity involved in the manufacture, distribution, or use of carbromal must navigate a complex and often restrictive regulatory landscape.
Key Takeaways
- Carbromal, an older sedative-hypnotic, has a minimal global market characterized by declining therapeutic relevance.
- Its current demand is predominantly driven by specialized compounding pharmacies and research institutions, not mainstream healthcare.
- Pricing is high per unit due to small-scale production, limited competition, and niche demand, with typical prices ranging from $50 to $300+ per gram depending on purity.
- Future market trends indicate continued contraction, with stable but low demand from its niche applications. Price projections suggest stability with potential slight annual increases due to rising production costs.
- Carbromal is not approved for marketing as a drug in major markets like the U.S. and EU, and its handling is subject to strict compounding and chemical regulations.
Frequently Asked Questions
-
Is carbromal currently approved for any medical indications in the United States or Europe?
No, carbromal is not approved for marketing as a drug for any indication by the U.S. Food and Drug Administration (FDA) or the European Medicines Agency (EMA).
-
What is the primary reason for the limited market of carbromal?
The limited market is primarily due to the availability of safer and more effective sedative and hypnotic alternatives, coupled with safety concerns associated with carbromal's historical use.
-
Where can one typically source carbromal for research or compounding purposes?
Carbromal is typically sourced from specialized chemical suppliers or compounding ingredient distributors that cater to research laboratories and compounding pharmacies.
-
Are there any significant growth prospects for the carbromal market?
No, significant growth prospects for the carbromal market are unlikely. The market is expected to remain niche and likely contract slowly over time.
-
What are the potential risks associated with handling or using carbromal?
Potential risks include its toxicity, potential for dependence, and the lack of robust clinical data supporting its safety and efficacy compared to modern pharmaceuticals. Regulatory non-compliance in handling and distribution also poses significant risks.
Citations
[1] U.S. Food and Drug Administration. (n.d.). Approved Drug Products. Retrieved from https://www.accessdata.fda.gov/scripts/cder/ob/
[2] European Medicines Agency. (n.d.). Medicines. Retrieved from https://www.ema.europa.eu/en/medicines
[3] U.S. Pharmacopeial Convention. (n.d.). USP Standards. Retrieved from https://www.usp.org/compounding/standards