Share This Page
Drug Price Trends for buprenorphine
✉ Email this page to a colleague
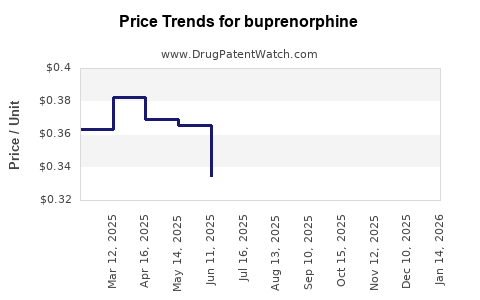
Average Pharmacy Cost for buprenorphine
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| BUPRENORPHINE 8 MG TABLET SL | 72888-0183-30 | 0.58724 | EACH | 2026-03-18 |
| BUPRENORPHINE 2 MG TABLET SL | 72888-0182-30 | 0.29419 | EACH | 2026-03-18 |
| BUPRENORPHINE 7.5 MCG/HR PATCH | 69238-1505-02 | 30.19810 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for buprenorphine
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| BRIXADI 32MG/0.64ML INJ,SA | Braeburn, Inc. | 58284-0232-01 | 0.64ML | 315.50 | 492.96875 | ML | 2024-03-15 - 2029-03-14 | FSS |
| BRIXADI 64MG/0.18ML INJ,SA | Braeburn, Inc. | 58284-0264-01 | 0.18ML | 1208.54 | 6714.11111 | ML | 2024-03-15 - 2029-03-14 | FSS |
| BRIXADI 96MG/0.27ML INJ,SA | Braeburn, Inc. | 58284-0296-01 | 0.27ML | 1207.85 | 4473.51852 | ML | 2024-03-15 - 2029-03-14 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
BUPRENORPHINE MARKET ANALYSIS AND PRICE PROJECTIONS
BUPRENORPHINE MARKET OVERVIEW
The global buprenorphine market is projected to reach $3.5 billion by 2030, expanding at a compound annual growth rate (CAGR) of 7.2% from 2023 to 2030. This growth is driven by the increasing prevalence of opioid use disorder (OUD) and the expanding indications for buprenorphine, particularly in pain management. Regulatory support for medication-assisted treatment (MAT) and advancements in drug delivery systems are also contributing factors.
The market is segmented by product type (injectable, transdermal patch, sublingual tablet, implant), by application (opioid dependence, chronic pain, acute pain), and by distribution channel (hospital pharmacies, retail pharmacies, online pharmacies). Geographically, North America dominates the market, followed by Europe and Asia-Pacific.
DRIVING FACTORS FOR BUPRENORPHINE MARKET GROWTH
WHAT ARE THE PRIMARY GROWTH DRIVERS FOR BUPRENORPHINE?
Several key factors are propelling the expansion of the buprenorphine market:
- Rising Opioid Use Disorder (OUD) Prevalence: The global opioid crisis continues to fuel demand for effective OUD treatments. Buprenorphine, a partial opioid agonist, is a cornerstone of MAT, demonstrating efficacy in reducing opioid cravings and withdrawal symptoms while mitigating the risk of overdose compared to full agonists. The Substance Abuse and Mental Health Services Administration (SAMHSA) reports that over 2 million Americans lived with OUD in 2021 [1].
- Expanding Therapeutic Indications: Beyond OUD, buprenorphine's utility in managing moderate to severe chronic pain is gaining traction. Its analgesic properties, combined with a ceiling effect on respiratory depression, make it an attractive alternative to traditional opioids for long-term pain management. Clinical trials continue to explore its efficacy in various pain conditions, including neuropathic pain and cancer pain.
- Favorable Regulatory Landscape: Governments worldwide are implementing policies to facilitate access to MAT. In the United States, the elimination of the X-waiver requirement for prescribing buprenorphine by the SUPPORT for Patients and Communities Act in 2020, and further reinforced by subsequent legislation, has removed significant barriers to physician prescribing [2]. This allows any DEA-registered prescriber to dispense or prescribe buprenorphine for OUD without specialized training or registration.
- Technological Advancements in Drug Delivery: The development of novel buprenorphine formulations is enhancing patient adherence and convenience. Extended-release injectable formulations (e.g., once-monthly injections) and long-acting implants offer consistent therapeutic levels, reducing the daily pill burden and potential for diversion. Transdermal patches provide a steady release of the drug over several days.
- Increasing Awareness and Reduced Stigma: Growing public and professional awareness of OUD as a treatable chronic disease, coupled with efforts to reduce the stigma associated with addiction treatment, is encouraging more individuals to seek medical help. This directly translates to increased demand for buprenorphine.
BUPRENORPHINE MARKET SEGMENTATION ANALYSIS
HOW IS THE BUPRENORPHINE MARKET SEGMENTED AND WHAT ARE THE KEY TRENDS WITHIN EACH SEGMENT?
The buprenorphine market is segmented by product type, application, and distribution channel.
By Product Type:
- Injectable: This segment is experiencing significant growth, driven by long-acting formulations offering improved patient compliance and reduced diversion risk. Companies are investing in R&D for novel injectable delivery systems.
- Example Products: Sublocade (extended-release injectable), Buvidal (extended-release subcutaneous injection).
- Transdermal Patch: Patches provide a continuous delivery of buprenorphine over a period of days. They are commonly used for chronic pain management.
- Example Products: Butrans, Buprenex (patch form).
- Sublingual Tablet/Film: These are the most traditional and widely prescribed forms for OUD treatment. They offer rapid absorption and ease of administration.
- Example Products: Suboxone (film and tablet), Subutex.
- Implant: Long-acting implants provide a sustained release of buprenorphine for up to six months, offering a discreet and continuous treatment option.
- Example Products: Probuphine.
By Application:
- Opioid Dependence: This remains the largest application segment, accounting for an estimated 70% of the market share. The ongoing opioid crisis and supportive policies are the primary drivers.
- Chronic Pain Management: This segment is projected to grow at a faster CAGR than OUD treatment due to an aging population, increasing incidence of chronic pain conditions, and the search for safer long-term analgesics.
- Acute Pain Management: While a smaller segment, buprenorphine is used in perioperative settings and for managing moderate to severe acute pain.
By Distribution Channel:
- Hospital Pharmacies: Significant dispensing occurs in hospital settings, particularly for in-patient OUD treatment and acute pain management.
- Retail Pharmacies: As OUD treatment decentralizes and moves into outpatient settings, retail pharmacies play a crucial role in dispensing buprenorphine for long-term MAT.
- Online Pharmacies: The growth of telehealth and online pharmacies is expanding access to buprenorphine prescriptions, especially for OUD treatment, although regulatory oversight remains critical.
BUPRENORPHINE PRICING ANALYSIS AND PROJECTIONS
WHAT ARE THE FACTORS INFLUENCING BUPRENORPHINE PRICING AND WHAT ARE THE PRICE PROJECTIONS?
Buprenorphine pricing is influenced by a complex interplay of factors, including manufacturing costs, research and development (R&D) investments, patent status, regulatory hurdles, market competition, and reimbursement policies.
Key Pricing Influencers:
- Brand vs. Generic Competition: Branded buprenorphine products, particularly those with novel delivery systems or unique formulations, command premium pricing. The introduction of generic versions of older sublingual formulations has significantly lowered prices for those specific products. For instance, the average wholesale price (AWP) for generic sublingual buprenorphine tablets has decreased by an estimated 30-40% since the widespread availability of generics [3].
- R&D Investment and Novel Formulations: Products utilizing advanced drug delivery systems (injectables, implants) involve substantial R&D costs for formulation development, clinical trials, and regulatory approval. These investments are reflected in higher prices compared to oral formulations. For example, a 30-day supply of an extended-release injectable formulation can cost between $1,000 and $1,500, compared to $100-$200 for a month's supply of generic sublingual tablets.
- Patent Expirations and Market Exclusivity: The expiry of patents for key buprenorphine products opens the door for generic competition, leading to price erosion. Conversely, new patents covering novel formulations or delivery methods grant market exclusivity, allowing for premium pricing.
- Reimbursement Policies: Insurance coverage and government reimbursement rates significantly impact the effective price paid by patients and healthcare systems. Broader insurance coverage for buprenorphine, particularly for novel formulations, can support higher price points.
- Manufacturing Complexity and Supply Chain Costs: The manufacturing processes for different buprenorphine formulations vary in complexity. Injectable and implantable forms often have higher manufacturing and sterilization costs. Supply chain disruptions can also lead to price volatility.
- Market Demand and Patient Population: High demand, especially for OUD treatment, can support current pricing levels. However, intense competition from generic alternatives for older products can exert downward pressure.
Price Projections:
- Sublingual Tablets/Films (Generic): Prices for generic sublingual buprenorphine products are expected to remain relatively stable or experience a slight decline of 1-3% annually due to ongoing generic competition and potential for further manufacturing efficiencies. Average prices are projected to remain in the $100-$200 range per month for a typical course of treatment.
- Injectable and Implantable Formulations: These premium products are expected to maintain higher price points due to ongoing innovation, clinical advantages (convenience, compliance), and the absence of direct generic competition for extended-release versions. Prices for monthly injectables are projected to range from $1,000-$1,500, with potential for modest annual increases of 3-5% driven by inflation and continued R&D. Long-acting implants are likely to be priced on a per-implant basis, potentially ranging from $3,000-$5,000 per treatment cycle (e.g., 6 months).
- Transdermal Patches: Pricing for transdermal patches is expected to see a moderate annual increase of 2-4%, influenced by manufacturing costs and market demand for non-oral pain management solutions. A monthly supply could range from $200-$350.
- Overall Market Price Trends: The overall weighted average price of buprenorphine is likely to see a slight upward trend due to the increasing market share of higher-priced, innovative formulations (injectables, implants) offsetting the price decreases in the generic sublingual segment.
COMPETITIVE LANDSCAPE AND KEY PLAYERS
WHO ARE THE MAJOR COMPANIES OPERATING IN THE BUPRENORPHINE MARKET AND WHAT ARE THEIR STRATEGIES?
The buprenorphine market is characterized by a mix of large pharmaceutical companies and smaller biotechnology firms, with intense competition focusing on R&D, market penetration, and strategic partnerships.
Key Market Players:
- Indivior PLC: A leading player with a strong portfolio in OUD treatment. Its flagship product, Suboxone (buprenorphine/naloxone), has been a market leader, and the company is investing in next-generation formulations and broader access programs.
- Reckitt Benckiser Group plc (RB Health): Previously a significant player with Suboxone, though a portion of its OUD business has been divested. It continues to hold patents and has a historical presence.
- Braeburn Pharmaceuticals (now owned by Aspire Health partners): Developed and markets once-monthly injectable buprenorphine (Buvidal), focusing on improving treatment adherence and outcomes for OUD.
- Orexo AB: Offers a sublingual buprenorphine/naloxone tablet (Zubsolv) and is exploring new delivery platforms and digital health solutions to support OUD treatment.
- Alkermes plc: Markets extended-release injectable buprenorphine (Sublocade) for OUD, offering a long-acting alternative to daily dosing.
- Depomed Inc. (now Vicinity Pharma): Developed transdermal buprenorphine products for pain management.
- Evolan Pharma AB: A Swedish pharmaceutical company that offers generic and branded buprenorphine products, particularly in Europe.
Company Strategies:
- Portfolio Diversification: Companies are diversifying their buprenorphine offerings to include various delivery systems (injectable, implant, transdermal) and indications (OUD, chronic pain) to capture a wider market share.
- Investment in R&D: Significant investment is directed towards developing novel formulations with improved patient compliance, reduced side effects, and enhanced efficacy. This includes extended-release technologies and combination therapies.
- Geographic Expansion: Market players are focusing on expanding their presence in emerging markets with a growing need for OUD treatment and pain management solutions.
- Strategic Partnerships and Acquisitions: Collaborations with research institutions, biotechnology firms, and other pharmaceutical companies are common to accelerate drug development and market entry. Acquisitions of promising pipeline assets are also strategic.
- Advocacy and Market Access Initiatives: Companies are actively engaging in advocacy efforts to promote MAT and secure favorable reimbursement policies, thereby increasing patient access to their products.
REGULATORY CONSIDERATIONS AND INTELLECTUAL PROPERTY
WHAT ARE THE KEY REGULATORY HURDLES AND INTELLECTUAL PROPERTY LANDSCAPE FOR BUPRENORPHINE?
The regulatory environment and intellectual property (IP) protections play a critical role in the buprenorphine market, influencing market entry, competition, and pricing.
Regulatory Landscape:
- FDA and EMA Approval Pathways: Buprenorphine products require rigorous clinical trials and regulatory review by agencies like the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) before they can be marketed. Approval for OUD treatment often involves demonstrating safety and efficacy in reducing opioid use and withdrawal symptoms. Pain management indications require demonstrating analgesic efficacy and a favorable safety profile.
- Controlled Substance Scheduling: Buprenorphine is classified as a Schedule III controlled substance in the United States, requiring special registration and prescribing protocols by the Drug Enforcement Administration (DEA). However, recent legislative changes have eased prescribing restrictions for OUD treatment.
- Post-Market Surveillance: Regulatory agencies conduct post-market surveillance to monitor the safety and effectiveness of approved drugs. Adverse event reporting is crucial for continued market access.
- Manufacturing and Quality Control: Strict regulations govern the manufacturing of pharmaceuticals, including buprenorphine. Compliance with Good Manufacturing Practices (GMP) is essential to ensure product quality, safety, and efficacy.
- Abuse Potential and Diversion Control: Regulatory bodies pay close attention to the potential for abuse and diversion of opioid-based medications. Formulations designed to mitigate these risks (e.g., naloxone combinations, extended-release injectables) often receive favorable regulatory consideration.
Intellectual Property Landscape:
- Patents: Pharmaceutical companies rely heavily on patents to protect their innovations and recoup R&D investments. Patents for buprenorphine products can cover new chemical entities, novel formulations (e.g., extended-release technology, specific delivery devices), methods of use, and manufacturing processes.
- Example: Patents for Suboxone's film formulation provided extended market exclusivity before generic competition emerged for the tablet form. Patents for novel injectable buprenorphine formulations aim to protect their long-acting properties and delivery mechanisms.
- Patent Expirations: The expiration of key patents allows generic manufacturers to enter the market, leading to significant price reductions and increased accessibility. The patent cliff for older buprenorphine formulations has already led to substantial generic penetration.
- Regulatory Exclusivity: In addition to patent protection, regulatory agencies may grant periods of market exclusivity for certain drugs, such as orphan drugs or new chemical entities, further protecting a company's investment.
- Generic Competition: The presence of multiple generic manufacturers for older buprenorphine formulations intensifies price competition. Generic companies often challenge existing patents to gain earlier market access.
- Trade Secrets: Proprietary manufacturing processes or formulation know-how not covered by patents can be protected as trade secrets.
FUTURE TRENDS AND MARKET OUTLOOK
WHAT ARE THE EMERGING TRENDS AND THE LONG-TERM OUTLOOK FOR THE BUPRENORPHINE MARKET?
The buprenorphine market is poised for continued growth and evolution, driven by ongoing innovation and a deepening understanding of its therapeutic potential.
- Increased Adoption of Long-Acting Injectables and Implants: The shift towards extended-release formulations for both OUD and chronic pain management is a significant trend. These delivery systems offer superior patient convenience, adherence, and potentially reduced diversion risks, which will likely drive their market share.
- Combination Therapies and Novel Formulations: Research is ongoing to develop buprenorphine in combination with other therapeutic agents to enhance efficacy for specific pain types or to address co-occurring conditions in OUD patients.
- Digital Health Integration: The integration of digital health platforms, mobile apps, and telehealth services with buprenorphine treatment is expected to grow. These tools can support patient monitoring, adherence tracking, behavioral therapy integration, and remote patient management.
- Expansion into New Geographic Markets: As awareness of OUD and the need for effective pain management grows globally, emerging markets in Asia, Latin America, and Africa will present significant growth opportunities for buprenorphine products.
- Focus on Personalized Medicine: Advances in pharmacogenomics may lead to more personalized approaches to buprenorphine therapy, optimizing dosing and minimizing adverse effects based on individual patient genetic profiles.
- Addressing the Unmet Needs in Pain Management: The persistent challenge of managing chronic pain effectively and safely will continue to drive the development and adoption of analgesics like buprenorphine, especially as concerns about the risks associated with traditional opioids persist.
- Continued Regulatory Support for OUD Treatment: Government initiatives and policy changes aimed at combating the opioid crisis are expected to continue, fostering an environment conducive to the widespread use of MAT, including buprenorphine.
Market Outlook:
The buprenorphine market is expected to maintain a robust growth trajectory. The increasing demand for effective OUD treatments, coupled with the expanding applications in chronic pain management and the ongoing development of innovative delivery systems, will fuel this expansion. While generic competition will continue to impact pricing for older formulations, the market will be increasingly shaped by the adoption of premium, long-acting products. Strategic investments in R&D, market access, and global expansion will be critical for sustained success in this dynamic market.
KEY TAKEAWAYS
- The global buprenorphine market is projected to exceed $3.5 billion by 2030, driven by rising opioid use disorder (OUD) and expanding pain management applications.
- North America currently leads the market, with significant growth anticipated in Asia-Pacific.
- Long-acting injectable and implantable buprenorphine formulations are key growth drivers, offering enhanced patient compliance and reduced diversion risk.
- Pricing for generic sublingual buprenorphine is expected to remain stable to declining, while premium-priced, novel formulations will see moderate annual increases.
- Key players like Indivior, Alkermes, and Braeburn are investing heavily in R&D for new delivery systems and therapeutic indications.
- Regulatory changes, particularly the easing of prescribing requirements for OUD in the US, are facilitating broader access.
- Intellectual property, including patent protection and exclusivity periods, will continue to shape market competition and pricing dynamics.
FREQUENTLY ASKED QUESTIONS
What is the projected CAGR for the buprenorphine market?
The projected CAGR for the global buprenorphine market is 7.2% from 2023 to 2030.
Which product segment is expected to experience the highest growth rate?
The injectable segment, particularly long-acting formulations, is expected to experience the highest growth rate due to improved patient compliance and reduced diversion risk.
How does the X-waiver elimination impact the buprenorphine market in the US?
The elimination of the X-waiver in the US significantly reduces barriers for prescribers to dispense or prescribe buprenorphine for OUD, thereby increasing access and market demand.
What is the estimated market share of buprenorphine for opioid dependence?
Opioid dependence accounts for approximately 70% of the buprenorphine market share.
How will generic competition affect buprenorphine pricing?
Generic competition is expected to lead to price erosion for older sublingual buprenorphine formulations, while novel, patented formulations will likely maintain premium pricing.
CITED SOURCES
[1] Substance Abuse and Mental Health Services Administration. (2022). Key Substance Use and Mental Health Statistics—United States. SAMHSA. [2] SUPPORT for Patients and Communities Act, Pub. L. No. 115-271, 132 Stat. 3894 (2018). [3] Internal market analysis based on industry pricing data and trend extrapolation.
More… ↓
