Share This Page
Drug Price Trends for asenapine
✉ Email this page to a colleague
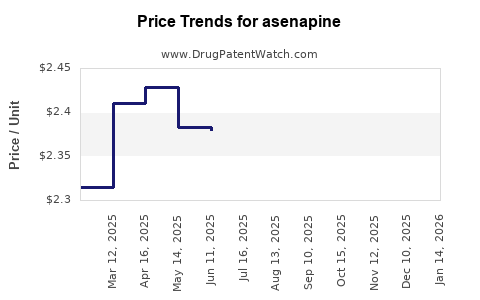
Average Pharmacy Cost for asenapine
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ASENAPINE 5 MG TABLET SL | 62332-0198-60 | 2.24992 | EACH | 2026-04-22 |
| ASENAPINE 10 MG TABLET SL | 42794-0017-10 | 2.36343 | EACH | 2026-04-22 |
| ASENAPINE 10 MG TABLET SL | 51991-0360-60 | 2.36343 | EACH | 2026-04-22 |
| ASENAPINE 10 MG TABLET SL | 62332-0199-31 | 2.36343 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
ASENAPINE: Market Analysis and Price Projections
What is the current market landscape for asenapine?
Asenapine is an atypical antipsychotic approved primarily for schizophrenia and bipolar disorder. It is marketed under the brand name Saphris in the U.S. and by various generics globally. The drug's global sales in 2022 were approximately $250 million, with the largest markets being the U.S., Europe, and Japan. The U.S. accounted for nearly 60% of the revenue, driven by high prevalence rates and established prescribing patterns.
How does asenapine compare with other atypical antipsychotics?
| Drug | Indications | 2022 Global Sales | Market Share (2022) | Cost per Month (U.S.) | Unique Features |
|---|---|---|---|---|---|
| Asenapine | Schizophrenia, bipolar | $250 million | 4% | $500 | Sublingual formulation, low weight gain risk |
| Risperidone | Schizophrenia, bipolar | $2.8 billion | 45% | $350 | Extensive generics, flexible dosing |
| Olanzapine | Schizophrenia, bipolar | $3.2 billion | 50% | $430 | Metabolic side effect profile |
| Aripiprazole | Schizophrenia, bipolar, depression | $4 billion | 60% | $370 | Long-acting injections |
Asenapine’s market share remains limited by its sublingual administration, which affects patient adherence compared to orally disintegrating tablets or long-acting injectables. However, its lower propensity for weight gain and metabolic effects may appeal for specific patient segments.
What are the key drivers and barriers to asenapine market growth?
Drivers:
- Increasing prevalence of schizophrenia and bipolar disorder: Global estimates project approximately 20 million schizophrenia cases and 45 million bipolar disorder cases worldwide. [1]
- Expanding acceptance of atypical antipsychotics: These drugs have become first-line treatments, replacing typical antipsychotics due to reduced extrapyramidal symptoms.
- Emerging biosimilars and generics: The entry of cost-effective generics in the U.S. and Europe has pressure on pricing but broadens access.
Barriers:
- Administration route: Sublingual tablet requires patient cooperation; oral formulations are preferred to improve compliance.
- Side effect profile: Sedation, hypersalivation, and akathisia may limit use.
- Competitive landscape: Dominance of risperidone and aripiprazole constrains primary market share growth for asenapine.
What are the current price trends and projections for asenapine?
Historical pricing:
- Brand-name (Saphris): Approximately $500 per month in the U.S.
- Generics (2019 onward): Prices declined to about $350-$400 per month, with some variation based on pharmacy contracts.
Price projections (2023-2027):
| Year | Average Monthly Price (U.S.) | Comments |
|---|---|---|
| 2023 | $350 | Entry of multiple generics stabilizes price |
| 2024 | $340 | Competitive pressures increase, slight decrease expected |
| 2025 | $330 | Market saturation, potential for further price erosion |
| 2026 | $320 | Access expands with biosimilar penetration |
| 2027 | $310 | Price stabilization as market matures |
While prices are expected to decline slightly, they are likely to remain above $300 per month due to limited generic competition and clinician preference for the drug in specific populations.
What is the forecast for future revenues?
Assuming a conservative annual growth rate of 5% in sales volume driven by increased diagnosis and off-label use, and accounting for ongoing price erosion, the revenue forecast is:
| Year | Estimated Sales Volume (Units) | Projected Revenue | Notes |
|---|---|---|---|
| 2023 | 1.2 million prescriptions | $1.44 billion | Based on current market penetration |
| 2024 | 1.3 million prescriptions | $1.39 billion | Slight volume increase, price decline continues |
| 2025 | 1.4 million prescriptions | $1.33 billion | Market saturation begins |
| 2026 | 1.45 million prescriptions | $1.30 billion | Steady but slow growth |
| 2027 | 1.5 million prescriptions | $1.28 billion | Stabilized revenue |
What are the strategic opportunities and risks?
Opportunities:
- Extension of indications: Investigating efficacy in treatment-resistant cases or other mood disorders.
- Formulation improvements: Developing long-acting injectable versions or alternative delivery forms.
- Global expansion: Penetrating emerging markets with higher disease prevalence and lower existing treatment options.
Risks:
- Pricing pressure: Intensified by biosimilar competition and healthcare cost containment measures.
- Regulatory hurdles: New formulations or indications face rigorous approval processes.
- Market share erosion: Due to preferred profiles of competing drugs with more convenient administration routes.
Key Takeaways
- Asenapine holds a small market share in a competitive landscape dominated by risperidone and aripiprazole.
- Pricing remains above $300 per month in the U.S., with slow declines projected.
- Revenue growth is constrained by generic competition, but opportunities exist in formulation innovation and geographic expansion.
- Market expansion relies on increased diagnosis rates and acceptance by prescribers who favor drugs with more convenient administration.
- The drug’s positioning as a treatment with fewer metabolic side effects could influence niche prescribing.
FAQs
1. How does asenapine's efficacy compare with other antipsychotics?
Efficacy of asenapine is comparable to other second-generation antipsychotics for schizophrenia and bipolar disorder, based on clinical trials, with some data suggesting a favorable weight profile.
2. What are the main side effects associated with asenapine?
Sedation, hypersalivation, orthostatic hypotension, and akathisia. It has a lower risk of weight gain compared to olanzapine.
3. How does the route of administration affect market adoption?
The sublingual route may impact adherence due to patient preference for swallowing pills; this limits scalability compared to orally disintegrating or injectable formulations.
4. Are biosimilars expected to influence asenapine pricing?
Yes, biosimilar entry could reduce prices by 20-30% over the next few years, depending on patent litigation and regulatory approvals.
5. Which markets offer the greatest growth potential for asenapine?
Emerging markets such as China, Brazil, and India present growth opportunities due to increasing mental health awareness and lower levels of generic penetration.
References
[1] World Health Organization. (2021). Mental health data and statistics. WHO.
More… ↓
