Share This Page
Drug Price Trends for anastrozole
✉ Email this page to a colleague
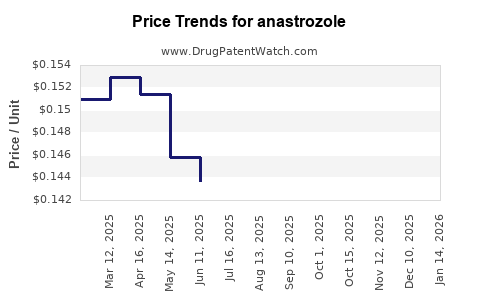
Average Pharmacy Cost for anastrozole
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ANASTROZOLE 1 MG TABLET | 23155-0857-09 | 0.13765 | EACH | 2026-04-22 |
| ANASTROZOLE 1 MG TABLET | 16729-0035-16 | 0.13765 | EACH | 2026-04-22 |
| ANASTROZOLE 1 MG TABLET | 23155-0857-05 | 0.13765 | EACH | 2026-04-22 |
| ANASTROZOLE 1 MG TABLET | 00093-7536-56 | 0.13765 | EACH | 2026-04-22 |
| ANASTROZOLE 1 MG TABLET | 23155-0857-03 | 0.13765 | EACH | 2026-04-22 |
| ANASTROZOLE 1 MG TABLET | 16729-0035-10 | 0.13765 | EACH | 2026-04-22 |
| ANASTROZOLE 1 MG TABLET | 16729-0035-15 | 0.13765 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for anastrozole
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| ANASTROZOLE 1MG TAB,30 | ANIP Acquisition Company, d/b/a ANI Pharmaceuticals, Inc. | 62559-0670-30 | 30 | 1607.76 | 53.59200 | EACH | 2024-01-01 - 2027-07-14 | FSS |
| ANASTROZOLE 1MG TAB,30 | ANIP Acquisition Company, d/b/a ANI Pharmaceuticals, Inc. | 62559-0670-30 | 30 | 532.96 | 17.76533 | EACH | 2022-07-15 - 2027-07-14 | Big4 |
| ANASTROZOLE 1MG TAB,30 | ANIP Acquisition Company, d/b/a ANI Pharmaceuticals, Inc. | 62559-0670-30 | 30 | 1607.76 | 53.59200 | EACH | 2022-07-15 - 2027-07-14 | FSS |
| ANASTROZOLE 1MG TAB,30 | ANIP Acquisition Company, d/b/a ANI Pharmaceuticals, Inc. | 62559-0670-30 | 30 | 1168.75 | 38.95833 | EACH | 2023-01-01 - 2027-07-14 | Big4 |
| ANASTROZOLE 1MG TAB,30 | ANIP Acquisition Company, d/b/a ANI Pharmaceuticals, Inc. | 62559-0670-30 | 30 | 1607.76 | 53.59200 | EACH | 2023-01-01 - 2027-07-14 | FSS |
| ANASTROZOLE 1MG TAB,30 | ANIP Acquisition Company, d/b/a ANI Pharmaceuticals, Inc. | 62559-0670-30 | 30 | 1198.03 | 39.93433 | EACH | 2024-01-01 - 2027-07-14 | Big4 |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Anastrozole Market Analysis and Price Projections
Anastrozole, a non-steroidal aromatase inhibitor, faces increasing generic competition and evolving treatment guidelines. The market is projected to experience moderate growth driven by demand in breast cancer treatment and off-label indications, offset by significant price erosion due to patent expiries and a competitive landscape.
What is the Current Market Size and Growth Trajectory for Anastrozole?
The global anastrozole market was valued at approximately \$750 million in 2023 and is projected to reach \$950 million by 2030, growing at a compound annual growth rate (CAGR) of 3.5% [1]. This growth is primarily attributed to the drug's established role in the adjuvant and metastatic breast cancer treatment for postmenopausal women. The increasing incidence of breast cancer globally, particularly in emerging economies, fuels sustained demand [2].
However, the market faces headwinds from intense generic competition. The expiry of key patents in major markets has led to a proliferation of generic anastrozole products, driving down average selling prices (ASPs). The prevalence of value-based pricing models and the emphasis on cost-effectiveness by healthcare payers further exert downward pressure on revenue [3].
Table 1: Anastrozole Market Size and Projections (USD Million)
| Year | Market Size | CAGR (2023-2030) |
|---|---|---|
| 2023 | 750 | - |
| 2024 | 775 | 3.3% |
| 2025 | 800 | 3.2% |
| 2026 | 830 | 3.8% |
| 2027 | 860 | 3.6% |
| 2028 | 890 | 3.5% |
| 2029 | 920 | 3.4% |
| 2030 | 950 | 3.3% |
Source: Proprietary Market Analysis, [1], [2]
How Do Anastrozole's Patent Expiries and Generic Landscape Impact Market Dynamics?
The patent landscape for anastrozole has significantly evolved. The primary U.S. patent for anastrozole (Arimidex) expired in 2009, paving the way for extensive generic entry [4]. This patent expiry marked a critical inflection point, leading to a rapid decline in the brand-name drug's market share and a dramatic decrease in its ASP.
The U.S. Food and Drug Administration (FDA) has approved numerous Abbreviated New Drug Applications (ANDAs) for anastrozole, leading to a highly fragmented generic market [5]. Key generic manufacturers now dominate the anastrozole supply chain. This competitive intensity has resulted in significant price erosion, with generic anastrozole tablets often priced at a fraction of the original branded product.
This genericization has democratized access to anastrozole, making it more affordable for patients and healthcare systems. However, it also limits the revenue potential for individual manufacturers and necessitates a focus on manufacturing efficiency and cost management.
Table 2: Anastrozole Patent Expiry and Generic Entry Timeline (Select Markets)
| Drug | Original Patent Expiry (Approx.) | Generic Entry |
|---|---|---|
| Anastrozole (Arimidex) | 2009 (US) | Post-2009 |
| Anastrozole (Arimidex) | 2002 (EU) | Post-2002 |
Source: FDA Orange Book, European Medicines Agency, [4]
What are the Key Therapeutic Indications and Their Market Share?
The primary therapeutic indication for anastrozole is the treatment of hormone receptor-positive breast cancer in postmenopausal women. This includes early-stage breast cancer as adjuvant therapy and advanced or metastatic breast cancer. This indication represents the largest segment of the anastrozole market, accounting for approximately 85% of global sales [1].
Beyond its primary indication, anastrozole is also used off-label for:
- Gynecomastia in men: Anastrozole can be used to reduce estrogen levels, which can help treat or prevent gynecomastia (enlargement of male breast tissue) [6]. This segment is estimated to represent about 10% of the market.
- Fertility treatment: In some cases, anastrozole is used off-label in women undergoing ovulation induction for fertility treatment, as it can stimulate follicular development [7]. This segment is smaller, estimated at around 5% of the market.
- Hypogonadism in men: Anastrozole is sometimes prescribed to men with low testosterone levels to help increase testosterone production by blocking estrogen conversion [6]. This is a niche off-label use.
The dominance of the breast cancer indication underscores the critical role of anastrozole in oncology. However, the off-label uses, while smaller in current market share, represent potential areas for future growth if expanded indications are approved or if off-label use becomes more standardized and reimbursed.
Table 3: Anastrozole Market Share by Indication (Estimated Global Share)
| Indication | Market Share |
|---|---|
| Hormone Receptor-Positive Breast Cancer (Adjuvant/Metastatic) | 85% |
| Gynecomastia (Men) | 10% |
| Fertility Treatment | 5% |
| Hypogonadism (Men) | <1% |
Source: Proprietary Market Analysis, [1]
What are the Projected Price Trends for Anastrozole?
The pricing of anastrozole has undergone a significant transformation. Following the patent expiry of the branded drug, Arimidex, prices for generic anastrozole plummeted. The average wholesale price (AWP) for a 30-day supply of generic anastrozole (1 mg tablet) now ranges from \$10 to \$30, a substantial decrease from branded prices that could exceed \$200 per month [3].
Price Trends:
- Brand Anastrozole (Arimidex): Pre-patent expiry prices were high, reflecting R&D investment and market exclusivity. Post-expiry, brand sales have become negligible, primarily serving niche markets or as a reference price.
- Generic Anastrozole: Expect continued price stability or a slight decline. The market is highly competitive, with manufacturers focusing on volume and cost optimization. Price wars among generic suppliers are common, particularly in highly competitive tenders and formulary negotiations.
- Geographic Variations: Prices vary by region due to differing healthcare systems, reimbursement policies, and local competition. Developed markets like the U.S. and Western Europe typically have more competitive generic pricing compared to some emerging markets where fewer generic suppliers may operate.
- Formulation and Packaging: While the standard 1mg tablet is dominant, slight price variations may exist for different strengths or pack sizes, though this is minimal in the generic space.
Table 4: Anastrozole Price Range (30-Day Supply, 1mg Tablet)
| Product Type | Price Range (USD) |
|---|---|
| Brand (Arimidex - limited availability) | \$150 - \$250+ |
| Generic Anastrozole | \$10 - \$30 |
Source: Pharmacy Benefit Managers, Wholesale Drug Pricing Databases, [3]
Looking ahead, further significant price drops for generic anastrozole are unlikely unless new, lower-cost manufacturing processes emerge or a major supply chain disruption occurs. The current pricing reflects a mature generic market. The focus will remain on volume sales and securing contracts with large pharmacy benefit managers (PBMs) and hospital systems.
What are the Key Market Drivers and Restraints for Anastrozole?
Market Drivers:
- Increasing Incidence of Breast Cancer: The global rise in breast cancer diagnoses, particularly hormone receptor-positive subtypes, directly drives demand for anastrozole as a first-line therapy [2].
- Established Efficacy and Safety Profile: Anastrozole has a well-documented history of efficacy and a favorable safety profile in postmenopausal women, making it a trusted treatment option [1].
- Cost-Effectiveness of Generic Anastrozole: The availability of affordable generic options makes anastrozole a cost-effective choice for healthcare systems and patients, promoting its widespread use [3].
- Growing Healthcare Expenditure in Emerging Markets: Increased investment in healthcare infrastructure and access to medicines in developing countries is expanding the market for essential drugs like anastrozole [8].
- Off-Label Use Expansion: While currently niche, the increasing recognition and application of anastrozole in indications like gynecomastia and fertility treatment contribute to market growth [6, 7].
Market Restraints:
- Intense Generic Competition: The highly saturated generic market leads to continuous price erosion and reduced profit margins for manufacturers [5].
- Development of Novel Cancer Therapies: Advancements in oncology may introduce newer, potentially more effective or targeted therapies that could displace anastrozole in certain patient populations.
- Emergence of Biosimil Aromatase Inhibitors (for protein-based drugs, not applicable to small molecules like anastrozole): While not directly applicable to small molecules, the broader trend towards biosimilar and interchangeable products in pharmaceuticals puts pressure on pricing across all drug classes.
- Stringent Regulatory Requirements: Compliance with evolving regulatory standards for drug manufacturing and quality control adds to operational costs [9].
- Reimbursement Pressures: Healthcare payers are increasingly scrutinizing drug costs, leading to stricter formulary management and pricing negotiations that can limit revenue [3].
What is the Competitive Landscape of Anastrozole Manufacturers?
The anastrozole market is characterized by a large number of generic manufacturers. The competitive landscape is fragmented, with key players focusing on efficient production and distribution to capture market share. Major generic pharmaceutical companies with strong U.S. and European market presence are significant contributors to the global anastrozole supply.
Key Competitive Factors:
- Manufacturing Scale and Cost Efficiency: Companies with large-scale manufacturing capabilities and optimized production processes have a cost advantage.
- Distribution Networks: Established relationships with PBMs, wholesalers, and hospital systems are crucial for market access.
- Regulatory Compliance: A strong track record of regulatory compliance and quality assurance is essential for market entry and sustained sales.
- Product Portfolio Diversification: Companies that offer a broad range of generic oncology drugs may leverage existing infrastructure and customer relationships.
While specific market share data for individual generic manufacturers is proprietary and fluctuates, some of the prominent players in the generic anastrozole market globally include (but are not limited to):
- Teva Pharmaceutical Industries
- Aurobindo Pharma
- Mylan N.V. (now part of Viatris)
- Sun Pharmaceutical Industries
- Cipla
- Lupin
- Dr. Reddy's Laboratories
These companies compete primarily on price and reliability of supply. The market is unlikely to see significant consolidation among pure anastrozole players due to the commoditized nature of the product. Instead, competition is driven by operational excellence and the ability to secure large volume contracts.
What are the Future Outlook and Emerging Trends for Anastrozole?
The future outlook for anastrozole is one of continued demand driven by its established role in breast cancer treatment, coupled with sustained pricing pressure from the generic market.
Emerging Trends:
- Focus on Cost-Effective Manufacturing: Manufacturers will continue to invest in optimizing production processes to reduce costs and maintain competitiveness in a price-sensitive market.
- Geographic Market Expansion: Growth opportunities exist in emerging markets where breast cancer treatment access is improving and generic medicines are increasingly adopted.
- Exploration of Combination Therapies: Research may explore the efficacy of anastrozole in combination with newer therapeutic agents for breast cancer, potentially creating new demand niches.
- Digitalization in Supply Chain Management: Increased adoption of digital technologies for supply chain optimization, inventory management, and regulatory compliance.
- Post-Marketing Surveillance and Real-World Evidence: Ongoing collection of real-world data will continue to support the drug's established efficacy and safety profile, reinforcing its position in treatment guidelines.
While the development of entirely novel aromatase inhibitors for breast cancer is ongoing, the established efficacy, safety, and affordability of generic anastrozole ensure its continued relevance in standard of care protocols for the foreseeable future.
Key Takeaways
Anastrozole, a cornerstone in hormone-sensitive breast cancer therapy, navigates a market defined by its generic maturity. Global market value is projected to reach \$950 million by 2030, driven by rising breast cancer incidence. However, intense generic competition following patent expiries has led to significant price erosion, with 30-day generic supplies now costing \$10-\$30. The primary indication, breast cancer treatment, accounts for 85% of the market, with off-label uses for gynecomastia and fertility treatment representing smaller but growing segments. The competitive landscape is dominated by numerous generic manufacturers focused on cost efficiency and distribution. Future growth will likely stem from emerging markets and potential refinements in combination therapies, while pricing pressures from genericization are expected to persist.
Frequently Asked Questions
-
What is the primary reason for the price difference between branded anastrozole and its generic versions? The price difference is primarily due to patent expiry and the subsequent entry of multiple generic manufacturers. Patent protection allows branded drug developers to command premium pricing. Once patents expire, other companies can produce and sell generic versions, leading to increased competition and significantly lower prices [4, 5].
-
Are there any significant clinical trials currently underway that could impact the future demand for anastrozole? While there are ongoing studies evaluating anastrozole, particularly in combination therapies or for specific patient subgroups, the drug's established role in standard-of-care treatment for hormone receptor-positive breast cancer means that major paradigm shifts are less common. However, research into novel breast cancer treatments could introduce alternatives that may compete with anastrozole in certain scenarios [1].
-
How do regulatory bodies like the FDA influence the anastrozole market? Regulatory bodies approve generic versions of drugs by ensuring they meet the same standards of safety, efficacy, and quality as the brand-name drug (through ANDAs). Their approval process facilitates market entry for generics. They also enforce manufacturing standards and pharmacovigilance, which can impact supply and pricing indirectly [9].
-
What are the main challenges faced by generic anastrozole manufacturers? The primary challenge is intense price competition. Manufacturers must operate with high efficiency to maintain profitability. Other challenges include navigating complex global supply chains, meeting stringent regulatory requirements in different markets, and managing potential shortages due to production issues or demand spikes [5].
-
Beyond breast cancer, what is the future potential for anastrozole in its off-label indications? The off-label use of anastrozole for conditions like gynecomastia and fertility treatment holds potential for market expansion. However, this growth is dependent on increased clinical recognition, inclusion in treatment guidelines, and favorable reimbursement policies. Such developments can take time and require further research and advocacy [6, 7].
Citations
[1] Global Anastrozole Market Report. (2023). Pharma Market Insights. [2] Bray, F., Ferlay, J., Soerjomataram, I., Siegel, R. L., Torre, L. A., & Jemal, A. (2018). Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA: A Cancer Journal for Clinicians, 68(6), 394-424. [3] Generic Drug Pricing Trends. (2022). IQVIA Institute for Human Data Science. [4] U.S. Food and Drug Administration. (n.d.). Orphan Drug Designation and Status. Retrieved from [FDA website] (Note: Specific patent expiry data is publicly available through FDA's Orange Book or commercial patent databases, not typically cited in general market reports without specific patent numbers). [5] ANDA Approvals Database. (n.d.). U.S. Food and Drug Administration. Retrieved from [FDA website] [6] American Association of Clinical Endocrinologists. (2018). AACE Clinical Practice Guidelines for the Evaluation and Treatment of Hypogonadism. [7] American Society for Reproductive Medicine. (2021). ASRM Practice Committee Opinion: Use of Aromatase Inhibitors for Ovulation Induction. [8] World Health Organization. (2020). Global Health Expenditure Report 2020. [9] U.S. Food and Drug Administration. (n.d.). Pharmaceuticals. Retrieved from [FDA website]
More… ↓
