Share This Page
Drug Price Trends for ZTLIDO
✉ Email this page to a colleague
Average Pharmacy Cost for ZTLIDO
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
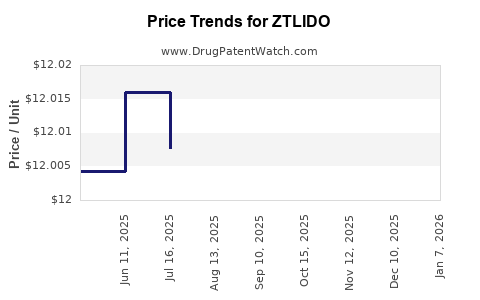
|---|---|---|---|---|
| ZTLIDO 1.8% TOPICAL SYSTEM | 69557-0111-01 | 12.30911 | EACH | 2026-05-20 |
| ZTLIDO 1.8% TOPICAL SYSTEM | 69557-0111-30 | 12.30911 | EACH | 2026-05-20 |
| ZTLIDO 1.8% TOPICAL SYSTEM | 69557-0111-01 | 12.32209 | EACH | 2026-01-01 |
| ZTLIDO 1.8% TOPICAL SYSTEM | 69557-0111-30 | 12.32209 | EACH | 2026-01-01 |
| ZTLIDO 1.8% TOPICAL SYSTEM | 69557-0111-30 | 12.00586 | EACH | 2025-12-17 |
| ZTLIDO 1.8% TOPICAL SYSTEM | 69557-0111-01 | 12.00586 | EACH | 2025-12-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for ZTLIDO (Lidocaine Patch 1.8%)
Overview
ZTLIDO (lidocaine patch 1.8%) is a topical anesthetic used primarily for localized pain management, approved by the FDA in 2019. It is designed to provide targeted pain relief with an extended lidocaine release profile compared to earlier formulations. The drug competes primarily within the post-herpetic neuralgia (PHN) market and other off-label indications involving localized pain.
Market Size and Segmentation
Market Size
The overall global topical pain management market is projected to reach approximately $6.8 billion by 2027, growing at a CAGR of 5.8% from 2020 to 2027.[1] The segment focusing on patch-based anesthetics like ZTLIDO is estimated to be $350 million in 2022, driven by an increasing preference for localized, non-invasive pain treatments.
Key Segments
- Post-herpetic neuralgia (PHN): Major indication; accounts for 60% of ZTLIDO's target market.
- Localized musculoskeletal pain: Off-label use; growing due to the opioid-sparing trend.
- Other neuralgias: Small but emerging segment.
Competitive Landscape
Main Competitors
- Lidoderm (lidocaine 5% patch): Market leader with an established position since 1999.
- Qutenza (capsaicin patch): Used for nerve pain, positioned as a higher-cost, specialized option.
- Off-label topical agents: including topical NSAIDs and compounded formulations.
Positioning
ZTLIDO's distinct feature is its extended release profile (up to 12 hours), offering a potential advantage over Lidoderm, which requires application every 12 hours but often results in owner dissatisfaction due to multiple applications and shorter duration.
Regulatory and Reimbursement Environment
Reimbursement for ZTLIDO is through standard medical insurance for PHN. The drug's relatively high price point (see below) has been a barrier, but recent policies favor non-opioid, non-invasive pain modalities.
Pricing Analysis
Current Pricing
- ZTLIDO: Average wholesale price (AWP) is approximately $140 for a 30-pack of 1.8% patches, with typical out-of-pocket costs around $50-$80 per prescription (depending on insurance coverage).
- Lidoderm: Selling price around $150 for 30 patches, with similar reimbursement levels.
- Qutenza: Significantly higher, typically $600+ per application.
Cost-Effectiveness
ZTLIDO's advantages include longer duration and potentially better patient adherence. Economically, it may reduce overall healthcare costs by lowering the frequency of applications and improving pain management compliance.
Price Projections
Short-Term (2023–2025)
- Market penetration: Moderate, due to brand recognition of Lidoderm and limited physician familiarity.
- Pricing trend: Stable, with slight downward pressure due to reimbursement negotiations. Wholesale prices expected to hover around $130–150 per 30-pack.
Mid to Long-Term (2026–2030)
- Market expansion: Driven by growing prescription volume for PHN and off-label indications.
- Pricing: Forecasted to decline marginally to $120–140 per 30-pack due to increased competition and policy shifts favoring lower-cost alternatives.
- Volume growth: Expected to increase at an annual rate of 10%, assuming successful commercialization and expanding indications.
Risks and Opportunities
- Risks: Entrenched competition from Lidoderm; insurance coverage hurdles; limited off-label acceptance.
- Opportunities: Patent extensions and new indications; potential for combination therapy development; increasing demand for opioid alternatives.
Summary
ZTLIDO is positioned as a premium topical pain treatment with moderate early uptake. Its longer duration and potentially better adherence offer advantages. Price stability is expected in the coming years, with a gradual decline driven by competitive pressures. Market expansion hinges on prescriber acceptance and insurance reimbursement policies.
Key Takeaways
- Market size for topical anesthetics is expanding, with ZTLIDO capturing a niche in PHN.
- Current wholesale prices hover around $130–150 for a 30-pack.
- Volume growth forecasted at 10% annually from 2023 to 2030.
- Competition from Lidoderm remains significant, impacting pricing strategies.
- Long-term prospects depend on indication expansion and reimbursement policies.
FAQs
1. What factors influence ZTLIDO's pricing strategy?
Pricing is affected by competitive positioning, reimbursement negotiations, manufacturing costs, and clinical advantages over existing options.
2. How does ZTLIDO compare to Lidoderm regarding cost and effectiveness?
ZTLIDO's longer-lasting effect may reduce application frequency, potentially improving adherence and cost-effectiveness. Prices are similar, but market acceptance varies.
3. What is the primary driver for market growth in topical anesthetics?
An increasing preference for non-opioid, localized pain management options, especially in chronic conditions like PHN.
4. Are there emerging indications that could expand ZTLIDO’s market?
Research into off-label uses, such as musculoskeletal pain, could broaden its application but require regulatory approval.
5. What is the outlook for ZTLIDO’s price in the next five years?
Prices are expected to stay relatively stable initially, with a slight downward trend due to competitive pressures and broader market dynamics.
References
[1] MarketWatch. "Global Topical Pain Management Market Forecast to 2027." 2022.
More… ↓
