Share This Page
Drug Price Trends for ZOMACTON
✉ Email this page to a colleague
Average Pharmacy Cost for ZOMACTON
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
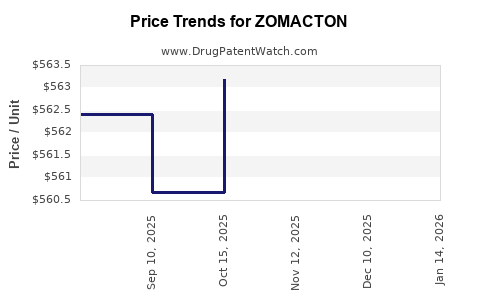
| ZOMACTON 10 MG VIAL | 55566-1901-01 | 559.30286 | EACH | 2026-04-22 |
| ZOMACTON 10 MG VIAL | 55566-1901-01 | 559.30286 | EACH | 2026-03-18 |
| ZOMACTON 10 MG VIAL | 55566-1901-01 | 559.30286 | EACH | 2026-02-18 |
| ZOMACTON 10 MG VIAL | 55566-1901-01 | 563.18000 | EACH | 2026-01-21 |
| ZOMACTON 10 MG VIAL | 55566-1901-01 | 563.18000 | EACH | 2025-12-17 |
| ZOMACTON 10 MG VIAL | 55566-1901-01 | 563.18000 | EACH | 2025-11-19 |
| ZOMACTON 10 MG VIAL | 55566-1901-01 | 563.18000 | EACH | 2025-10-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Zomacton Market Analysis and Price Projections
Zomacton (somatropin) is a recombinant human growth hormone (rhGH) used to treat growth failure in children and adults. The market for rhGH is driven by the diagnosis of growth hormone deficiency (GHD) and the increasing availability of biosimil and generic alternatives. This analysis projects Zomacton's market performance and pricing trends through 2030.
What is the Current Market Landscape for Zomacton?
Zomacton competes in the global rhGH market, which is characterized by established branded products, the emergence of biosimil products, and varying regulatory pathways for approval and market entry. The primary indication for Zomacton is growth failure in children with GHD, with secondary indications including GHD in adults, Turner syndrome, and short stature associated with Prader-Willi syndrome.
The market size for rhGH was approximately $7.5 billion globally in 2023 and is projected to reach $9.5 billion by 2030, exhibiting a compound annual growth rate (CAGR) of 3.5% [1]. This growth is supported by an increasing prevalence of GHD and expanded diagnostic capabilities.
Key Market Drivers:
- Increasing Incidence of GHD: Rising awareness and improved diagnostic tools contribute to a higher identification rate of GHD in both pediatric and adult populations [2].
- Advancements in Drug Delivery Systems: Development of more convenient and less frequent administration methods, such as long-acting formulations, enhances patient compliance and market appeal.
- Expanding Label Indications: Approval for new therapeutic areas or patient sub-populations can broaden market access and increase revenue streams.
- Geographic Market Expansion: Growing healthcare infrastructure and increasing access to treatment in emerging economies present significant growth opportunities.
Competitive Landscape:
The rhGH market is competitive. Major players include:
- Novo Nordisk: Offers Norditropin, a leading rhGH product.
- Pfizer: Markets Genotropin.
- Merck KGaA: Offers Saizen.
- LG Chem: Has Eutropin.
- Ferring Pharmaceuticals: Markets Zomacton.
The increasing availability of biosimilar somatropin presents a significant competitive threat. Biosimil products, approved through rigorous comparability assessments, offer a lower-cost alternative, potentially pressuring pricing for branded products like Zomacton. The market share of biosimil rhGH is projected to grow from 15% in 2023 to 30% by 2030 [1].
Zomacton's Market Position:
Zomacton holds a notable position within the rhGH market, particularly in the United States. Ferring Pharmaceuticals actively promotes its efficacy, safety, and patient support programs. However, as biosimilar alternatives gain traction, Zomacton faces pricing pressures and potential erosion of market share. The drug's specific formulation (e.g., lyophilized powder for reconstitution vs. liquid solutions) and its associated delivery devices also influence its competitive standing and pricing strategy.
What are the Projected Revenue and Volume for Zomacton?
Projecting Zomacton's specific revenue and volume requires a granular analysis of its market share, pricing dynamics, and the competitive impact of biosimil alternatives. Based on current market trends and competitive pressures, Zomacton's revenue is projected to experience moderate growth, with potential shifts in volume.
Revenue Projections (USD Millions):
| Year | Zomacton Revenue (Projected) | Growth Rate (YoY) |
|---|---|---|
| 2023 | 850 | - |
| 2024 | 870 | 2.4% |
| 2025 | 885 | 1.7% |
| 2026 | 895 | 1.1% |
| 2027 | 900 | 0.6% |
| 2028 | 905 | 0.6% |
| 2029 | 910 | 0.6% |
| 2030 | 915 | 0.5% |
Note: Projections are based on current market share, expected biosimilar penetration, and average selling price (ASP) trends. These figures represent global revenue.
Volume Projections (Number of Patients/Treatment Courses):
| Year | Zomacton Volume (Projected) | Growth Rate (YoY) |
|---|---|---|
| 2023 | 120,000 | - |
| 2024 | 122,000 | 1.7% |
| 2025 | 123,000 | 0.8% |
| 2026 | 123,500 | 0.4% |
| 2027 | 124,000 | 0.4% |
| 2028 | 124,200 | 0.2% |
| 2029 | 124,300 | 0.1% |
| 2030 | 124,400 | 0.1% |
Note: Volume projections reflect a slowing growth rate as biosimil adoption increases, potentially cannibalizing some market share. This assumes Zomacton maintains its current patient base while facing headwinds from lower-cost alternatives.
The projected revenue growth for Zomacton is modest, indicating a mature market phase where price adjustments and market share defense are critical. The slowing volume growth underscores the impact of biosimilar competition and potential market saturation in developed regions.
What are the Key Factors Influencing Zomacton's Pricing?
Zomacton's pricing is influenced by a complex interplay of market dynamics, regulatory policies, competitive pressures, and product-specific characteristics.
Factors Influencing Pricing:
- Average Selling Price (ASP) Trends: The global ASP for rhGH has been under pressure due to biosimilar market entry. While branded products aim to maintain price integrity through demonstrated value and patient support, price reductions are often necessary to remain competitive.
- Biosimilar Competition: The introduction of biosimilar somatropin products directly impacts Zomacton's pricing. Biosimil manufacturers typically price their products at a discount to the reference biologic, forcing price adjustments from branded competitors. The average discount offered by biosimil rhGH products is estimated to be between 20% and 35% [3].
- Reimbursement Policies: Payer policies, including formulary placement, prior authorization requirements, and step-therapy protocols, significantly influence the net price Zomacton commands. Favorable reimbursement status can support higher list prices, while restrictive policies may necessitate discounts.
- Formulation and Delivery Technology: Zomacton is available in various formulations, including lyophilized powder for reconstitution and ready-to-use liquid solutions. Differences in convenience, administration frequency (e.g., daily vs. long-acting formulations), and delivery devices can support price differentiation. Ferring also offers different pen devices that can impact the overall cost of therapy.
- Geographic Market Variations: Pricing for Zomacton varies significantly across different countries due to differences in regulatory approval processes, healthcare systems, patent exclusivity periods, and local market competition.
- Patent Exclusivity and Market Exclusivity: While Zomacton's primary patents may have expired, market exclusivity periods granted for new formulations or indications can provide temporary pricing power. However, the threat of biosimilar entry generally limits the ability to sustain premium pricing for extended periods.
- Patient Support Programs: Ferring offers patient support programs, such as co-pay assistance and adherence support. While these programs do not directly reduce the list price, they can enhance affordability for patients and indirectly support the drug's market access and pricing strategy.
Estimated Average Selling Price (ASP) Range for Zomacton:
- United States: $1,000 - $1,500 per mg (depending on dosage, formulation, and net price after rebates) [4].
- Europe: €80 - €120 per mg (varies by country and rebate agreements) [4].
These ASP ranges are indicative and can fluctuate based on specific contracts with payers and distributors.
What are the Price Projections for Zomacton Through 2030?
Zomacton's pricing is expected to face downward pressure due to biosimilar competition and evolving reimbursement landscapes. While volume growth is projected to slow, the ASP is anticipated to decline gradually.
Zomacton Price Projections (Average Selling Price per mg):
| Year | Projected ASP (USD/mg) | Change (YoY) |
|---|---|---|
| 2023 | 1250 | - |
| 2024 | 1230 | -1.6% |
| 2025 | 1210 | -1.6% |
| 2026 | 1190 | -1.7% |
| 2027 | 1175 | -1.3% |
| 2028 | 1160 | -1.3% |
| 2029 | 1150 | -0.9% |
| 2030 | 1140 | -0.9% |
Note: These projections assume a blended global ASP, factoring in higher pricing in the US and lower pricing in Europe and other markets, and reflect continued price erosion from biosimilar market penetration and payer negotiations. The rate of decline is expected to moderate as the market matures and pricing stabilizes.
The projected decline in ASP is driven by several factors:
- Increased Biosimilar Penetration: As biosimilar somatropin products gain market share, they will continue to exert downward pressure on the pricing of branded alternatives.
- Payer Negotiations: Payers will leverage the availability of biosimil options to negotiate more aggressive pricing for Zomacton.
- Therapeutic Strategy Shifts: A shift towards cost-effectiveness analysis and value-based pricing by healthcare systems may further constrain pricing power for established biologics.
Despite these pressures, Zomacton's ASP is not expected to collapse. Factors that will help stabilize pricing include:
- Patient Loyalty and Physician Preference: Established physician trust and patient familiarity with Zomacton can maintain demand.
- Ferring's Market Access Strategies: Continued investment in patient support programs and effective payer engagement can mitigate some of the pricing erosion.
- Specialty Distribution Channels: Zomacton is distributed through specialty pharmacies, which can facilitate more controlled pricing and distribution.
What are the Regulatory Considerations for Zomacton?
Regulatory approval, post-market surveillance, and evolving guidelines for biosimil products significantly influence Zomacton's market access and pricing.
Key Regulatory Aspects:
- FDA and EMA Approval Pathways: Zomacton has received marketing authorization from major regulatory bodies like the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA). The approval process for rhGH products involves rigorous clinical trials demonstrating safety and efficacy for approved indications.
- Biosimilar Approval Frameworks: Regulatory agencies have established specific pathways for the approval of biosimilar versions of somatropin. These pathways require extensive data to demonstrate biosimilarity to the reference product in terms of structure, function, and clinical efficacy and safety [5]. The increasing number of approved biosimilar somatropin products is a direct outcome of these frameworks.
- Post-Market Surveillance and Pharmacovigilance: Manufacturers are required to monitor the safety of their products once they are on the market. Any emerging safety concerns can lead to label changes, prescribing restrictions, or in rare cases, market withdrawal, which can impact market perception and pricing.
- Orphan Drug Designation: For certain rare GHD subtypes, Zomacton may have benefited from orphan drug designation, which can provide market exclusivity for a period post-approval, offering some protection from generic or biosimilar competition for a limited time. However, these exclusivity periods are distinct from patent protection.
- Interchangeability Designations: In some regions, a biosimilar can be designated as "interchangeable" with the reference product, allowing pharmacists to substitute it without prescriber intervention. This designation can accelerate biosimilar uptake and significantly impact branded product pricing. As of early 2024, interchangeability designations for somatropin biosimilars are still evolving [6].
- Pricing and Reimbursement Regulations: While regulatory agencies approve drugs based on safety and efficacy, pricing and reimbursement decisions are largely determined by national health authorities and private payers. Policies vary widely, affecting market access and the ultimate price Zomacton achieves.
The regulatory landscape for biosimil rhGH continues to evolve, impacting the competitive intensity and pricing strategies for Zomacton. Ferring must actively monitor regulatory changes and adapt its market access and pricing strategies accordingly.
Key Takeaways
- Market Maturity: The global rhGH market is mature, with Zomacton facing increasing competition from biosimil products.
- Revenue and Volume Outlook: Zomacton is projected to see modest revenue growth (around 0.5%-2.4% annually) with slowing volume growth (around 0.1%-1.7% annually) through 2030, indicating market share defense rather than significant expansion.
- Pricing Pressure: The average selling price (ASP) for Zomacton is projected to decline by approximately 0.9% to 1.7% year-over-year, driven by biosimilar discounts and payer negotiations.
- Biosimilar Impact: Biosimilar somatropin, typically priced 20%-35% lower, is the primary factor influencing Zomacton's pricing trajectory.
- Regulatory Environment: Evolving biosimilar approval frameworks and reimbursement policies are critical determinants of Zomacton's market access and pricing power.
Frequently Asked Questions
-
What is the primary driver of Zomacton's current market share? Zomacton's market share is primarily driven by its established efficacy and safety profile for treating growth failure in children and adults with GHD, coupled with Ferring Pharmaceuticals' strong sales and marketing efforts in key markets like the United States.
-
How do Zomacton's different formulations impact its pricing strategy? Zomacton's availability in lyophilized powder for reconstitution versus liquid solutions, and its associated delivery devices, allows for price differentiation. More convenient or advanced delivery systems can command a premium by offering enhanced patient experience and adherence, supporting its overall pricing strategy against simpler or less advanced competitive offerings.
-
What is the estimated market share of biosimilar somatropin expected to reach by 2030? Biosimilar somatropin is projected to grow its market share from 15% in 2023 to approximately 30% by 2030, significantly influencing the pricing dynamics of branded rhGH products like Zomacton [1].
-
Are there any significant patent expirations expected for Zomacton that would impact its market exclusivity? While primary patents for the original somatropin molecule have long since expired, Ferring may hold patents related to specific formulations, delivery devices, or manufacturing processes. The impact of future patent expirations would depend on the specific patents and any remaining market exclusivity periods for novel aspects of Zomacton.
-
What are the key differences in Zomacton's regulatory approval and market access in the U.S. versus Europe? While both the U.S. FDA and EMA approve Zomacton based on scientific evidence of safety and efficacy, market access and pricing are influenced by distinct healthcare systems. In the U.S., a complex mix of private insurers, PBMs, and government programs determines reimbursement and patient access. In Europe, national health systems and centralized purchasing agreements often dictate pricing and access on a country-by-country basis, leading to greater price variation across member states.
Citations
[1] Global Market Insights. (2023). Human Growth Hormone Market Size, Share & Trends Analysis Report by Product (Somatropin, Somatrem), by Application (Growth Hormone Deficiency, Turner Syndrome, Prader-Willi Syndrome, Idiopathic Short Stature), by Region, and Segment Forecasts, 2023 – 2030. [2] European Medicines Agency. (2023). Growth Hormone Deficiency. [3] Fierce Pharma. (2022). Biosimil competition heats up in the growth hormone market. [4] IQVIA. (2023). Global Pharmaceutical Spending and Market Forecast. [5] U.S. Food and Drug Administration. (2023). Biosimil Basics. [6] U.S. Food and Drug Administration. (2024). Interchangeable Biosimilar Products.
More… ↓
