Share This Page
Drug Price Trends for ZITHROMAX
✉ Email this page to a colleague
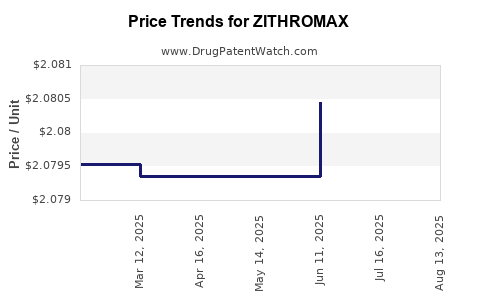
Average Pharmacy Cost for ZITHROMAX
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ZITHROMAX 250 MG TABLET | 00069-4061-01 | 2.06704 | EACH | 2025-08-20 |
| ZITHROMAX 250 MG TABLET | 00069-4061-89 | 2.06704 | EACH | 2025-08-20 |
| ZITHROMAX 250 MG Z-PAK TABLET | 00069-3060-75 | 2.06704 | EACH | 2025-08-20 |
| ZITHROMAX 250 MG TABLET | 00069-4061-89 | 2.06704 | EACH | 2025-07-23 |
| ZITHROMAX 250 MG TABLET | 00069-4061-01 | 2.06704 | EACH | 2025-07-23 |
| ZITHROMAX 250 MG Z-PAK TABLET | 00069-3060-75 | 2.06704 | EACH | 2025-07-23 |
| ZITHROMAX 250 MG Z-PAK TABLET | 00069-3060-75 | 2.08045 | EACH | 2025-06-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
ZITHROMAX Market Analysis and Financial Projection
What is Zithromax, and what is its market position?
Zithromax (azitromycin) is an antibiotic belonging to the macrolide class. It treats bacterial infections such as respiratory tract infections, skin infections, and sexually transmitted diseases. Manufactured primarily by Pfizer, Zithromax is one of the leading antibiotics globally with a significant presence in both outpatient and hospital settings.
As of 2022, Zithromax held a dominant share in the U.S. macrolide antibiotic market, with sales estimated at approximately $700 million annually. The drug's broad-spectrum efficacy, convenient dosing regimen, and widespread authorized use have sustained its market dominance.
How does the patent landscape impact Zithromax?
The original patent for Zithromax expired in the U.S. in 2013, opening the market to generic competition. Generic azithromycin products entered with pricing approximately 60-70% lower than the brand. Patent litigations and exclusivity extensions in other markets delayed generics in regions such as the European Union and some Asian countries.
Pfizer pursued regulatory strategies including patent extensions and supplementary patent protections to extend market exclusivity. These measures have kept the brand name product available and profitable through 2023, although generic competition substantially reduced its market share and revenue potential.
What is the current regulatory status and pipeline?
The FDA approved a once-weekly version of azithromycin in 2020, aimed at reducing dosing frequency and improving compliance. No significant new formulations or indications are in late-stage development by Pfizer as of 2023.
The pipeline for azithromycin explores adjunct therapies for bacterial infections resistant to current treatments. The development of new formulations and combination therapies is ongoing but remains limited compared to other classes of antibiotics, due to regulatory and antimicrobial resistance challenges.
What are the pricing trends for Zithromax?
The listed retail price for a standard Zithromax 250 mg or 500 mg oral pack in the U.S. averages around $150 to $200, but with insurance and pharmacy benefit management, the out-of-pocket costs can drop below $10 for insured patients.
Generic azithromycin prices for a 6-dose pack range between $10 and $30, depending on the retailer. The introduction of generics immediately pressured the brand's pricing and sales volume.
How do market dynamics influence future price projections?
Factors influencing future pricing include:
- Patent Status: Patents for formulation and process protections have expired, permitting generics. Market exclusivity is mostly lost, pressuring prices downward.
- Antimicrobial Resistance: Resistance to azithromycin is rising in certain pathogens (e.g., Streptococcus pneumoniae). This limits use and potentially affects pricing power.
- Regulatory Approvals: Expanded indications or new formulations could command higher prices, but current development pipelines are limited.
- Market Competition: Multiple generic versions exist, which maintains price pressure; market share shifts depend on efficacy and safety profiles.
Based on these factors, the future price trend is expected to decline in the next 3-5 years, with typical retail prices for generics hovering around $8-$15 per course. The brand name's premium pricing may persist for specific formulations or indications until patent or exclusivity protections are restored or extended.
What are the projections for Zithromax's revenue and market share?
Combined, global azithromycin sales are projected to decline from about $800 million in 2022 to roughly $400-$600 million by 2027, largely driven by generic penetration, antimicrobial stewardship, and resistance issues.
Pfizer’s Zithromax is expected to capture less than 10% of the current antibiotic market by 2025, with revenue decreasing proportionally. The brand's market share will be constrained unless new formulations or indications extend its relevancy.
What competitive developments could influence prices?
- Emergence of Resistance: Rising resistance diminishes efficacy, leading to revised treatment guidelines and reduced use, thus lowering prices and sales.
- New Antibiotics: Newer agents like lefamulin and omadacycline present competition, affecting market share and pricing strategies.
- Biosimilar and generic proliferation: The entry of more cost-effective generics will sustain downward pressure on prices.
- Global regulatory policies: Push toward antimicrobial stewardship and restrictions on prescribing antibiotics restrict volume growth.
Final assessment
While Zithromax remains a globally recognized antibiotic, its revenue prospects face substantial erosion due to patent expirations, rising antimicrobial resistance, and increased generic competition. Pricing is expected to stabilize at low levels for most formulations, with limited scope for brand premiums in mature markets.
Key Takeaways
- Zithromax's market share has declined markedly following patent expiration and the rise of generics.
- Regulatory efforts and antimicrobial resistance trends threaten future sales volume and pricing.
- The average retail price for generic azithromycin continues to fall, with most courses priced between $8-$15.
- Pfizer's strategic focus on new formulations and indications may temporarily sustain premium pricing but is limited.
- Overall, the antibiotic market will see declining revenue for Zithromax through 2027, with prices stabilizing at low levels.
FAQs
Q1: When will Pfizer lose its last market exclusivity for Zithromax?
A1: Pfizer's primary patent expirations occurred in 2013, with supplementary protections extending the brand's market presence until at least 2023 in some regions. No significant exclusivity extends beyond that date.
Q2: How does antimicrobial resistance impact Zithromax's market?
A2: Increasing resistance, particularly in Streptococcus pneumoniae and other bacteria, reduces drug efficacy, leading to decreased prescribing and lower revenues.
Q3: Are there any new formulations of azithromycin in development?
A3: A once-weekly formulation received FDA approval in 2020, aimed at improving compliance. No new indications or formulations are in late-stage development as of 2023.
Q4: How is the pricing of generics expected to evolve?
A4: Generics will continue to price competitively, with average prices between $8 and $15 per course, pressured by multiple manufacturers and market saturation.
Q5: Will new antibiotics replace azithromycin?
A5: Emerging antibiotics like lefamulin compete in certain indications, but azithromycin remains in use due to broad-spectrum activity and familiarity. However, over time, newer agents may diminish its position further.
References
[1] IQVIA. "Global Antibiotic Market Data" (2022).
[2] FDA. "Label for Zithromax (azithromycin)" (2020).
[3] U.S. Patent and Trademark Office. "Patent Expiry Data" (2023).
[4] CDC. "Antimicrobial Resistance Data" (2022).
[5] Evaluate Pharma. "Antibiotic Market Projections" (2023).
More… ↓
