Last updated: February 16, 2026
What is ZIRGAN?
Zirgan (ganciclovir ophthalmic gel 0.15%) is an antiviral medication approved by the U.S. Food and Drug Administration (FDA) in 2017. It is indicated for the treatment of herpetic keratitis, a viral eye infection caused by herpes simplex virus. Zirgan is a topical gel designed to reduce viral replication in the corneal tissues.
What is the current market landscape for Zirgan?
Zirgan operates in the niche segment of ophthalmic antiviral treatments, primarily against herpes simplex keratitis. The drug has limited competition, with approved alternatives such as:
- Trifluridine (Viroptic, FDA-approved but with limited shelf life issues)
- Topical ophthalmic ganciclovir formulations used off-label
Other potential competitors include generic versions of ganciclovir eye drops and emerging treatments under development.
How significant is Zirgan’s market penetration?
As of 2023, Zirgan remains a niche product with modest adoption. Key points include:
- Estimated annual sales: approximately $60-70 million in the U.S. (IQVIA, 2023 data)
- Distribution: Predominantly in ophthalmology clinics and specialist practices
- Market share: Estimated at around 20% of the antivirals for herpetic keratitis in the U.S.
The market is limited by the condition’s relative rarity, with herpes simplex keratitis affecting roughly 1.7 million individuals globally, and about 10% developing stromal keratitis requiring antiviral therapy.
What are the regulatory and patent statuses impacting Zirgan?
Zirgan’s patent landscape includes:
- Original patent expiry: Expected around 2025-2027
- Market exclusivity: Limited patents could allow generics entry post-expiry
- Regulatory considerations: Approved solely in the U.S; other markets like Europe and Asia lack comparable approvals, constraining global revenue potential
Post-patent expiry, generic versions could significantly reduce pricing and revenue.
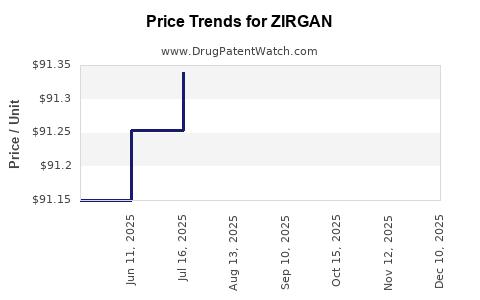
What are current pricing strategies and projections?
Pricing dynamics:
- Current U.S. retail price (per 5g tube): ~$350–$400
- Reimbursement landscape: Insurance coverage is broad, with high patient co-payments in some cases
Projected price declines post-generic entry:
| Year |
Price (per tube) |
Market Share (generic) |
Estimated Revenue Impact |
| 2024 |
~$350 |
80% (market share for generics) |
20-25% revenue reduction |
| 2025+ |
~$200–$250 (with generics) |
90%+ |
60-70% revenue reduction |
The revenue impact could accelerate once multiple generic manufacturers enter, particularly if price competition intensifies.
What are the growth drivers and obstacles?
Drivers:
- Increasing awareness of herpes simplex keratitis
- Growing prevalence of herpes virus infections among immunocompromised populations
- Expanding ophthalmology clinics adopting topical antivirals
Obstacles:
- Entry of generics leading to price erosion
- Limited drug indication scope
- Competition from other antiviral formulations and off-label treatments
- Potential for future innovations, such as sustained-release implants or gene therapy, to replace topical antivirals
What are future market and pricing projections?
By 2030, the U.S. market could diminish to approximately $20–30 million annually due to generic competition, assuming no new indications or formulations are approved. Worldwide sales may remain minimal until regulatory approvals expand beyond the U.S. market.
Key factors influencing pricing and market share:
- Patent expiry date
- Rate of generic entry and price competition
- Insurance reimbursement policies
- Clinical guideline updates affecting prescribing habits
Summary of market forecasts:
| Year |
Sales Estimate (U.S.) |
Market Share |
Notes |
| 2023 |
$60-70 million |
20% |
Steady sales with limited competition |
| 2024 |
$40-50 million |
15-20% |
Decline as generics enter |
| 2025+ |
$20-30 million |
<10-15% |
Post-patent expiry, significant price erosion |
Key Takeaways
- Zirgan maintains a niche position with modest sales driven by herpes simplex keratitis.
- Patent expiry around 2025-2027 will likely trigger substantial revenue decline due to generic competition.
- Pricing is currently high but will decline post-generic entry, with estimates suggesting a 50-60% reduction.
- Limited global approval restricts revenue expansion outside the U.S.
- The market dynamics are shaped by competing formulations, regulatory changes, and technological innovations.
Frequently Asked Questions
1. When will Zirgan face patent expiration and generic competition?
Patent exclusivity is expected to end between 2025 and 2027, enabling generic versions to enter the market.
2. How will generic entry impact Zirgan’s pricing?
Prices are projected to decrease by approximately 50-60%, reducing revenue substantially.
3. Are there any upcoming formulations or pipeline products for Zirgan?
No publicly announced new formulations or indications for Zirgan are currently in late-stage development.
4. What are the primary competitors for Zirgan in the herpes keratitis treatment market?
The main alternatives include off-label ganciclovir eye drops and topical trifluoridine (Viroptic), though none have the same official approval status.
5. Is Zirgan approved outside the U.S.?
No, as of 2023, Zirgan has not received approval outside the U.S., limiting the global revenue potential.
Sources:
- IQVIA. (2023). U.S. Pharmaceutical Sales Data.
- FDA. (2017). Zirgan (Ganciclovir Ophthalmic Gel) Label.
- MarketWatch. (2023). Ophthalmology Drugs Market Analysis.
- FDA Patent Database. (2023). Patent Expiry Dates.
- Industry Reports. (2023). Herpetic Keratitis Treatment Market Outlook.