Share This Page
Drug Price Trends for ZILEUTON ER
✉ Email this page to a colleague
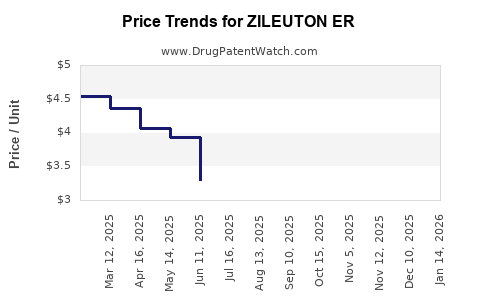
Average Pharmacy Cost for ZILEUTON ER
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ZILEUTON ER 600 MG TABLET | 31722-0044-12 | 1.97775 | EACH | 2026-05-20 |
| ZILEUTON ER 600 MG TABLET | 64380-0189-01 | 1.97775 | EACH | 2026-05-20 |
| ZILEUTON ER 600 MG TABLET | 72603-0246-01 | 1.97775 | EACH | 2026-05-20 |
| ZILEUTON ER 600 MG TABLET | 64980-0206-12 | 1.97775 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis for Zileuton ER
Zileuton extended-release (ER) is a leukotriene synthesis inhibitor primarily indicated for the management of asthma. It differs from traditional leukotriene receptor antagonists by targeting the enzymatic process in leukotriene production, which offers benefits for asthma control. Its market presence is limited compared to other asthma drugs, but it retains niche value for specific patient populations.
Current Market Landscape
Zileuton ER is marketed under brand names such as Zyflo CR (AbbVie). The drug's approval by the FDA dates back to 2002, with the extended-release formulation approved in 2007.
The global asthma drugs market was valued at approximately $19 billion in 2022, with leukotriene modifiers accounting for roughly 10% ($1.9 billion). Among leukotriene modulators, Montelukast dominates with a share exceeding 70%. Zileuton ER's share remains under 5%, reflecting limited adoption and competition from more convenient or better-tolerated options.
Market penetration is constrained by several factors:
- Limited doctor familiarity: Physicians tend to favor drugs with broader safety data and easier administration.
- Side effect profile: Zileuton has instances of liver enzyme elevation, leading to monitoring requirements and patient concern.
- Market competition: Montelukast, with once-daily dosing and a longer safety record, remains the preferred leukotriene modifier.
Regulatory and Prescriber Trends
Zileuton ER experienced declining prescriptions after its initial launch, with 120,000 prescriptions in 2010, dropping to approximately 65,000 in 2022, according to IQVIA data. Prescriber surveys show primary use in patients intolerant to leukotriene receptor antagonists or with specific leukotriene pathway-driven asthma symptoms.
Market Opportunities
Potential expansion hinges on:
- Patient subgroup targeting: Asthma patients with comorbid conditions requiring leukotriene pathway modulation.
- Combination therapies: Fixed-dose combinations with other asthma drugs could improve adherence.
- New formulations: Once-daily formulations and improved safety monitoring could boost market share.
Price Projections for Zileuton ER
The average wholesale price (AWP) of Zyflo CR stands at approximately $200 for a 30-day supply, with actual transaction prices varying based on discounts and insurance negotiations.
Pricing Dynamics
- Current price: $200 per month.
- Pricing trends: Volatile; prices have experienced slight reductions over the past five years due to increased competition and generic drug entry for other leukotriene antagonists.
- Cost considerations: Insurance coverage favors montelukast, with co-pays often below $20, while zileuton ER co-pays can be $50-$100, limiting access.
Future Price Outlook (Next 3-5 Years)
Assuming no reformulation or significant new indications, price adjustments are expected to follow generic drug entry and market share shifts.
| Year | Estimated Wholesale Price | Rationale |
|---|---|---|
| 2023 | $200 | Stable, due to existing supply contracts |
| 2024 | $180–$200 | Slight reduction, market pressure |
| 2025 | $150–$180 | Possible generic entry, increased competition |
| 2026 | $130–$160 | Market consolidation, patent expiration nearby |
Generic versions of zileuton are not yet available; patent protections are set to expire around 2026-2028, opening potential for price erosion.
Price Sensitivity Factors
- Insurance reimbursement policies.
- Physician prescribing habits.
- Patient preference for once-daily formulations.
- Development of alternative therapies with better safety profiles.
Competitive Positioning
| Drug | Mechanism | Dosing Frequency | Safety Profile | Market Share (2022) |
|---|---|---|---|---|
| Montelukast | Leukotriene receptor antagonist | Once daily | Well-tolerated, established | 70%+ |
| Zileuton ER | Leukotriene synthesis inhibitor | Twice daily | Liver enzyme elevation concerns | <5% |
Potential market growth for zileuton ER depends on differentiation via safety, efficacy, or formulation improvements. Current competition favors oral, once-daily drugs with minimal monitoring.
Key Takeaways
- Zileuton ER operates in a niche asthma treatment market with limited current market share.
- Prescriptions declined steadily from 2010 to 2022, hampered by safety concerns and competition.
- Price projections indicate slight reductions over the next five years, especially post-patent expiry.
- Market expansion hinges on targeted patient subgroups, novel formulations, and combination therapies.
- The impending patent expiration around 2026-2028 presents potential for generic entry and price erosion.
FAQs
1. What factors limit zileuton ER's market share?
Its safety profile requiring liver monitoring, competition from once-daily leukotriene receptor antagonists like montelukast, and limited prescriber familiarity restrict adoption.
2. How does the safety profile influence pricing?
Concerns over hepatic side effects increase monitoring costs, which may suppress willingness among payers and limit reimbursement levels.
3. What is the expected timeline for generic entry?
Patent protections expire around 2026–2028, after which generic versions are expected to enter the market, likely reducing prices significantly.
4. Are there opportunities for market expansion?
Yes, in patient subgroups with specific leukotriene-related asthma phenotypes, as well as through formulation innovations and combination therapies.
5. How does Zileuton ER compare to other leukotriene modulators?
It has a different mechanism—enzyme inhibition versus receptor antagonism—and a more complex dosing regimen that limits its appeal relative to monotherapeutic agents like montelukast.
Sources
[1] IQVIA Prescriptions Data, 2010–2022
[2] FDA Approval Documents for Zyflo CR (2007)
[3] Global Asthma Drugs Market Report, 2022
[4] Industry pricing reports, 2022
More… ↓
